Advanced Catalytic Cyclization for High-Purity S-Pregabalin Lactam Manufacturing
Advanced Catalytic Cyclization for High-Purity S-Pregabalin Lactam Manufacturing
The pharmaceutical industry's relentless pursuit of purity and regulatory compliance has placed significant emphasis on the synthesis of critical impurity standards, particularly for high-volume drugs like pregabalin. Patent CN109851543B introduces a groundbreaking methodology for the preparation of S-pregabalin lactam, a key degradation product that serves as an essential reference standard for quality control. This patent details a robust catalytic system that utilizes inexpensive acidic metal oxides to facilitate the intramolecular cyclization of pregabalin, achieving exceptional yields exceeding 94% and purity levels greater than 99%. For R&D directors and procurement specialists, this technology represents a paradigm shift away from costly, toxic reagents toward a sustainable, scalable, and economically superior manufacturing process.
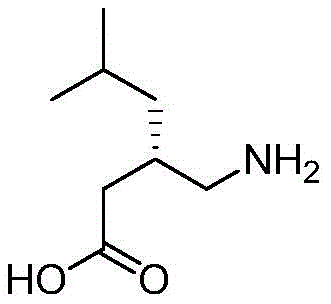
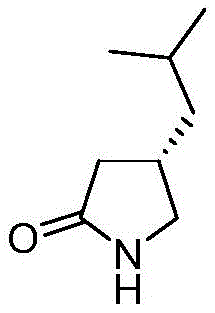
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of S-pregabalin lactam has been fraught with significant technical and economic challenges that hinder efficient large-scale production. Traditional routes described in literature, such as those utilizing azo methyl trimethyl silane as a cyclization agent, suffer from prohibitive costs and severe toxicity profiles, making them unsuitable for modern green chemistry standards. Other methods involving the activation of carboxyl groups with oxalyl chloride or thionyl chloride generate hazardous acidic gases and require stringent safety controls, increasing operational expenditures. Furthermore, earlier acid-catalyzed approaches using hydrochloric or acetic acid often struggled with reversible hydrolysis; without effective water removal, the equilibrium favored the starting material, resulting in low conversion rates and difficult purification processes that compromised overall yield and product integrity.
The Novel Approach
The methodology disclosed in patent CN109851543B elegantly circumvents these historical bottlenecks by employing a heterogeneous catalytic system driven by thermodynamic control. By utilizing cheap and readily available acidic promoters such as acidic alumina, acidic silica gel, or acidic kaolin, the process eliminates the need for expensive homogeneous catalysts or toxic activating agents. The core innovation lies in the integration of a water separator during the reflux stage in a water-immiscible solvent like toluene or xylene. This setup continuously removes the water byproduct generated during the lactamization, effectively driving the reaction to completion according to Le Chatelier's principle. This strategic modification ensures that the reaction substrate, pregabalin, is thoroughly converted into the target S-pregabalin lactam, minimizing residual starting material and simplifying downstream purification.
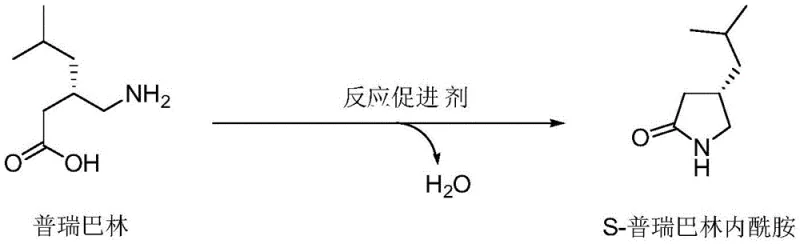
Mechanistic Insights into Acidic Oxide-Catalyzed Cyclization
The mechanistic pathway of this novel synthesis relies on the surface acidity of the metal oxide promoters to activate the carboxylic acid moiety of pregabalin for nucleophilic attack by the pendant amine group. The acidic sites on the alumina or silica gel surface act as Lewis or Brønsted acids, protonating the carbonyl oxygen or coordinating with the carboxyl group, thereby increasing its electrophilicity. This activation lowers the energy barrier for the intramolecular nucleophilic attack by the gamma-amino group, facilitating the formation of the five-membered lactam ring. Unlike homogeneous acid catalysis where the acid remains in solution and can promote reverse hydrolysis, the heterogeneous nature of the solid acidic oxides allows for easy separation post-reaction, reducing the risk of product degradation during workup and ensuring a cleaner reaction profile.
Crucially, the control of impurities in this process is governed by the efficient management of the reaction equilibrium. In conventional acid-catalyzed methods, the accumulation of water in the reaction medium promotes the hydrolysis of the newly formed lactam bond, regenerating the starting pregabalin and creating a dynamic equilibrium that limits yield. The patented process addresses this by employing a Dean-Stark apparatus or similar water-separating equipment. As the reaction mixture refluxes, the azeotrope formed between the solvent and water is condensed, and the denser water phase is separated and removed while the dry solvent returns to the flask. This continuous dehydration prevents the reverse hydrolysis reaction, ensuring that the impurity profile remains dominated by the target lactam with minimal unreacted pregabalin, thus achieving the reported purity of over 99% without complex chromatographic separation.
How to Synthesize S-Pregabalin Lactam Efficiently
The operational protocol for this synthesis is designed for simplicity and reproducibility, making it highly attractive for both laboratory reference standard preparation and potential commercial scale-up. The process involves suspending pregabalin in a suitable water-immiscible organic solvent, such as toluene, at a volume-to-mass ratio optimized for efficient mixing and heat transfer. Following the addition of the acidic promoter, the mixture is subjected to prolonged heating under reflux conditions. The detailed standardized synthetic steps, including specific molar ratios, temperature gradients, and workup procedures derived from the patent examples, are outlined in the guide below.
- Suspend pregabalin in a water-immiscible organic solvent such as toluene or xylene within a reaction vessel equipped with a Dean-Stark water separator.
- Add a catalytic amount of acidic metal oxide promoter, specifically acidic alumina, acidic silica gel, or acidic kaolin, to the reaction mixture.
- Heat the mixture to reflux for 4 to 20 hours, continuously removing generated water to drive the equilibrium toward the lactam product, followed by filtration and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic cyclization method offers substantial strategic advantages regarding cost structure and supply reliability. The substitution of expensive, specialized reagents like azo silanes with commodity chemicals such as acidic alumina drastically reduces the raw material cost per kilogram of the final product. Additionally, the elimination of toxic reagents simplifies waste disposal protocols and reduces the environmental compliance burden, translating into lower operational overheads. The robustness of the reaction conditions, which tolerate standard reflux temperatures and atmospheric pressure, minimizes the need for specialized high-pressure or cryogenic equipment, further enhancing the economic feasibility of the process.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the utilization of low-cost, commercially abundant catalysts. By avoiding the procurement of high-priced silane reagents or corrosive acid chlorides, manufacturers can significantly lower the direct material costs associated with production. Furthermore, the high conversion rate (>94% yield) implies that less raw material is wasted, maximizing the atom economy of the process. The simplified purification strategy, which relies on basic filtration and recrystallization rather than resource-intensive chromatography, also contributes to reduced processing time and lower utility consumption, collectively driving down the total cost of goods sold.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on generic, widely available chemical inputs. Acidic alumina, silica gel, and solvents like toluene are commodity products with stable global supply chains, reducing the risk of production stoppages due to raw material shortages. Unlike proprietary reagents that may have single-source suppliers and long lead times, the materials required for this synthesis can be sourced from multiple vendors, ensuring continuity of supply. The operational simplicity also means that the process can be easily transferred between different manufacturing sites or contract manufacturing organizations without requiring extensive retraining or specialized infrastructure.
- Scalability and Environmental Compliance: From a scalability perspective, the heterogeneous nature of the catalyst and the use of standard reflux equipment make this process highly amenable to scale-up from kilogram to multi-ton production. The absence of hazardous byproducts and the use of recyclable solvents align with modern green chemistry principles, facilitating easier regulatory approval and environmental permitting. The efficient removal of water not only drives the reaction but also simplifies the solvent recovery process, allowing for the recycling of toluene or xylene, which further enhances the sustainability profile and reduces the environmental footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of S-pregabalin lactam. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the method's efficacy and practical implementation for industry stakeholders.
Q: Why is S-pregabalin lactam critical for pregabalin quality control?
A: S-pregabalin lactam is the primary degradation impurity formed during the storage of pregabalin bulk drug and tablets. Regulatory guidelines require strict monitoring of this impurity, often limiting it to below 0.20%, necessitating high-purity reference standards for accurate HPLC quantification.
Q: What are the advantages of using acidic alumina over traditional cyclization reagents?
A: Traditional methods often employ expensive and toxic reagents like azo methyl trimethyl silane or require harsh activation with oxalyl chloride. Acidic alumina is inexpensive, non-toxic, easily available, and facilitates the reaction under mild reflux conditions without generating hazardous byproducts.
Q: How does the new method prevent the reverse hydrolysis reaction?
A: The process utilizes a water-immiscible solvent system coupled with a water separator (Dean-Stark trap). By continuously removing the water produced during the intramolecular amidation, the thermodynamic equilibrium is shifted decisively toward the lactam product, preventing the reverse hydrolysis that plagues acid-catalyzed methods in closed systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-Pregabalin Lactam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity impurity standards play in the pharmaceutical quality assurance ecosystem. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your demand for S-pregabalin lactam with consistent quality and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for HPLC calibration and stability testing, supporting your regulatory compliance efforts globally.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your R&D and commercial objectives efficiently.
