Advanced Pseudoionone Manufacturing: Achieving Ultra-High Purity Through Precise Raw Material Control
The chemical industry is constantly evolving, driven by the relentless demand for higher purity intermediates that serve as the foundational building blocks for vitamins, fragrances, and pharmaceuticals. A pivotal advancement in this domain is detailed in Patent CN113548952A, which discloses a sophisticated preparation method for high-quality pseudoionone. This patent addresses a long-standing bottleneck in the synthesis of this critical C13 ketone, specifically targeting the elusive goal of achieving purity levels greater than or equal to 98.5% through a novel control strategy. Unlike traditional methods that rely heavily on aggressive post-reaction purification to salvage yield, this innovation shifts the focus upstream to the quality of the raw material, specifically the citral feedstock. By rigorously controlling the index of prenol (isopentenol) within the citral, the process fundamentally alters the reaction pathway to suppress the formation of stubborn impurities. This technical breakthrough offers a compelling value proposition for global supply chains, promising not only superior product specifications but also a more streamlined, cost-effective manufacturing protocol that minimizes waste and energy consumption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of pseudoionone has been plagued by significant technical and environmental challenges that hinder the attainment of ultra-high purity standards required by top-tier downstream applications. Conventional alkaline catalysis methods, such as those reported in earlier patents like CN109096073A, typically involve the condensation of citral and acetone followed by extensive washing and distillation. While these methods can achieve purities in the range of 90% to 95%, they invariably generate massive quantities of salt-containing wastewater, creating a heavy burden on environmental treatment facilities and inflating operational expenditures. Furthermore, alternative high-temperature and high-pressure synthesis routes, while avoiding some catalyst issues, introduce severe risks of thermal degradation. Under such harsh conditions (230-300°C), both the citral reactant and the pseudoionone product are prone to decomposition, leading to the accumulation of heavy components and a consequent reduction in overall reaction yield. These legacy processes struggle to break the 95% purity ceiling, leaving a gap for high-end applications in vitamin A and E synthesis where trace impurities can compromise the stability and efficacy of the final active pharmaceutical ingredients.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology outlined in Patent CN113548952A introduces a paradigm shift by prioritizing feedstock specification over brute-force purification. The core innovation lies in the precise regulation of prenol content within the citral raw material, maintaining it within a narrow window of 0.001 to 0.2 wt%. This strategic control effectively starves the side reactions that lead to the formation of the notorious "pre-isomer," an impurity that has historically been the bane of pseudoionone manufacturers due to its similar boiling point. By mitigating the formation of this specific byproduct at the source, the subsequent separation process becomes remarkably efficient, requiring only simple distillation after light and heavy component removal to achieve the target purity. The reaction conditions are notably mild, operating between 20°C and 100°C with dilute alkali metal hydroxide solutions, which preserves the integrity of the delicate conjugated system in the molecule. This approach not only elevates the purity to ≥98.5% but also ensures a robust separation yield of over 97%, demonstrating that intelligent process design can outperform aggressive physical separation.
Mechanistic Insights into Prenol-Controlled Aldol Condensation
To fully appreciate the technical depth of this invention, one must understand the intricate relationship between raw material impurities and reaction selectivity in aldol condensation chemistry. The synthesis of citral itself is a complex multi-step process that inevitably introduces various impurities, with prenol being a persistent contaminant even after rectification. Research underlying this patent reveals that the presence of prenol is not merely a passive spectator but an active participant that drives the formation of pseudoionone pre-isomers. These pre-isomers are structurally analogous to the desired product, possessing very close boiling points and thermodynamic properties that render them virtually inseparable by standard fractional distillation. When the prenol content in the citral exceeds optimal thresholds, the reaction selectivity shifts unfavorably, generating significant quantities of these isomers which then co-distill with the product, capping the achievable purity. By reducing the prenol content to the specified range of 0.01-0.06 wt%, the kinetic pathway favoring pre-isomer formation is suppressed, allowing the main condensation reaction between acetone and citral to proceed with high fidelity.
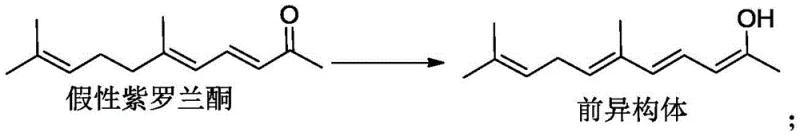
The structural nuance between the target pseudoionone and the pre-isomer is critical for R&D teams evaluating process feasibility. As illustrated in the reaction scheme, the pre-isomer represents a reduced or isomerized variant that complicates the purification landscape. In traditional scenarios where prenol is uncontrolled, the reaction mixture becomes a complex soup of conjugated enones and their saturated or rearranged counterparts. The patent data highlights that when prenol levels are kept low (e.g., 0.0013 wt% in Example 1), the selectivity for the pre-isomer drops to a negligible 0.2%, whereas higher prenol content (e.g., 0.2923 wt% in Comparative Example 1) causes the pre-isomer selectivity to spike to 1.8%. This seemingly small percentage difference is magnified during the concentration steps, where the pre-isomer accumulates and drags down the final assay. The use of acidic resin for catalyst removal further refines the stream, ensuring that no residual base remains to catalyze post-reaction isomerization during the distillation phase. This dual strategy of feedstock control and gentle workup creates a robust mechanism for impurity management that is far superior to attempting to strip these impurities out after they have already formed.
How to Synthesize High-Purity Pseudoionone Efficiently
Implementing this advanced synthesis route requires a disciplined approach to raw material sourcing and reaction monitoring to ensure consistent output that meets the rigorous demands of the fine chemical market. The process begins with the selection of citral that has been specifically rectified or treated to meet the stringent prenol specification, followed by a controlled addition of acetone and dilute alkali catalyst under moderate thermal conditions. Detailed standardized synthetic steps, including specific mass ratios, temperature ramps, and resin treatment protocols, are essential for replicating the high yields and purity reported in the patent examples. Operators must pay close attention to the gas phase monitoring of the reaction to determine the exact endpoint, ensuring maximum conversion of citral while minimizing residence time that could lead to degradation. For a comprehensive guide on executing this protocol in a pilot or production setting, please refer to the standardized operating procedures below.
- Prepare the reaction mixture by combining industrial grade citral (with controlled prenol content of 0.001-0.2 wt%) and acetone in a mass ratio of 1: 1 to 1:10 within a reactor.
- Introduce an aqueous alkali metal hydroxide solution (e.g., NaOH or KOH, 0.01-20% concentration) at a mass ratio of 0.0001-0.01 relative to citral, and maintain the temperature between 20°C and 100°C for 1 to 10 hours.
- Treat the resulting reaction liquid with acidic resin (such as sulfonic acid resin) to remove the catalyst, followed by simple distillation to remove light and heavy components, yielding high-purity pseudoionone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic advantages that extend far beyond simple chemical specifications. The shift towards a process that relies on feedstock quality rather than complex, energy-intensive purification infrastructure offers a clear path to cost optimization and supply resilience. By eliminating the need for high-pressure reactors and extreme temperature conditions, the capital expenditure (CAPEX) required for manufacturing equipment is significantly reduced, allowing for more flexible production scaling. Furthermore, the reduction in salt-containing wastewater simplifies environmental compliance, removing a major bottleneck that often limits production capacity in regions with strict discharge regulations. This streamlined workflow ensures that the supply of high-purity pseudoionone can be maintained with greater continuity, reducing the risk of stockouts that can disrupt the production schedules of downstream vitamin and fragrance manufacturers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the downstream processing units and the reduction in utility consumption. Because the reaction generates significantly fewer hard-to-separate impurities, the reliance on complex, multi-stage distillation columns or solvent extraction processes is minimized, leading to lower energy costs and reduced solvent loss. Additionally, the use of mild alkaline catalysts that can be easily neutralized and removed by resin avoids the expensive disposal costs associated with heavy metal catalysts or large volumes of saline effluent. This holistic reduction in operational complexity allows for a leaner manufacturing model, where the cost per kilogram of high-purity product is drastically lowered without compromising on quality standards.
- Enhanced Supply Chain Reliability: From a logistics and sourcing perspective, this method enhances reliability by decoupling production success from the limitations of specialized high-pressure equipment. The raw materials, citral and acetone, are commodity chemicals available from multiple global sources, and the specific requirement for low-prenol citral can be met through established rectification partnerships rather than relying on exotic reagents. This flexibility means that supply chains are less vulnerable to single-point failures or equipment downtime. The robustness of the mild reaction conditions also implies a longer lifespan for production vessels and a lower frequency of maintenance shutdowns, ensuring a steady and predictable flow of material to customers who depend on just-in-time delivery for their own formulation lines.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new variables that can compromise yield, but the linear nature of this condensation reaction makes it highly amenable to commercial scale-up. The absence of exothermic runaway risks associated with high-concentration catalysts or extreme temperatures allows for safer operation in larger reactors, facilitating a smooth transition from pilot batches to multi-ton production. Moreover, the environmental footprint of the process is substantially smaller, aligning with the increasing corporate sustainability goals of major pharmaceutical and flavor houses. By generating less hazardous waste and consuming less energy, manufacturers can secure long-term operating licenses more easily and position themselves as preferred green suppliers in a market that increasingly values eco-friendly production methodologies.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this technology into their existing portfolios, we have compiled a set of frequently asked questions based on the specific technical disclosures and experimental data found within the patent literature. These inquiries address the critical parameters regarding raw material specifications, expected purity outcomes, and the practical implications of the catalyst removal system. Understanding these nuances is vital for R&D directors planning tech transfers and for quality assurance teams establishing incoming inspection criteria for the new process. The answers provided below are derived directly from the empirical results of the patent examples, ensuring that the information is grounded in verified experimental reality rather than theoretical speculation.
Q: Why is controlling prenol content in citral critical for pseudoionone purity?
A: Research indicates that prenol impurities in the citral feedstock directly correlate with the formation of 'pre-isomers' during the condensation reaction. These pre-isomers possess boiling points and thermodynamic properties extremely close to the target pseudoionone, making them nearly impossible to separate via standard rectification. By restricting prenol levels to below 0.2 wt%, the generation of these difficult-to-remove impurities is drastically minimized.
Q: What purity levels can be achieved with this new preparation method?
A: By implementing the specific raw material controls and mild reaction conditions outlined in Patent CN113548952A, manufacturers can consistently achieve pseudoionone with a purity of ≥98.5%. In optimized examples, purities reaching 99.1% have been demonstrated with separation yields exceeding 97%, significantly outperforming conventional market standards which typically hover around 94%.
Q: How does this method improve environmental compliance compared to traditional processes?
A: Traditional alkaline catalysis often generates substantial amounts of salt-containing wastewater that requires complex treatment. This novel approach utilizes precise catalyst dosing and acidic resin for neutralization, simplifying the workup. Furthermore, the mild temperature conditions (20-100°C) reduce energy consumption and prevent thermal degradation of the product, aligning better with green chemistry principles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pseudoionone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate balance of prenol control and mild reaction conditions is maintained even at full industrial scale. Our facilities are equipped with state-of-the-art rigorous QC labs capable of detecting trace impurities at the ppm level, guaranteeing that every batch of pseudoionone meets stringent purity specifications required for vitamin and fine chemical synthesis. We understand the critical nature of this intermediate in the global supply chain and are committed to delivering consistent quality that empowers our clients to produce superior end-products.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific application needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your total cost of ownership. Please contact our technical procurement team today to request specific COA data from our recent pilot runs and to discuss route feasibility assessments for your project. Let us help you secure a stable, high-quality supply of pseudoionone that drives your innovation forward.
