Advanced Manufacturing of 1,4-Diazepane Derivatives for High-Purity Pharmaceutical Intermediates
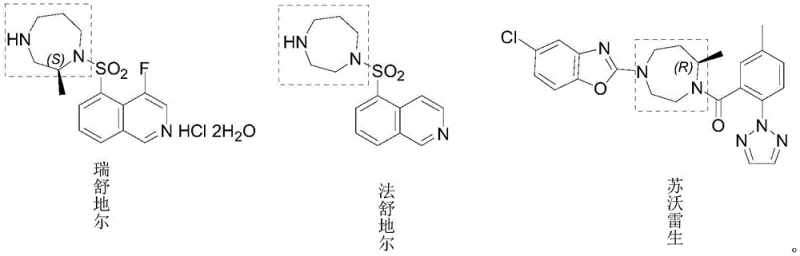
The pharmaceutical industry continuously seeks robust synthetic pathways for critical heterocyclic scaffolds, particularly the 1,4-diazepane ring system found in potent Rho kinase (ROCK) inhibitors such as Ripasudil, Fasudil, and Suvorexant. Patent CN116143713A, published on May 23, 2023, introduces a groundbreaking preparation method for 1,4-diazacycloheptane series derivatives that addresses significant limitations in prior art. This technology represents a paradigm shift from hazardous, low-yielding linear syntheses to a safer, convergent strategy that prioritizes impurity control and operational safety. By leveraging a unique nosyl-protection strategy and telescoped reaction sequences, this method ensures the production of high-purity intermediates essential for next-generation ophthalmic and cardiovascular therapeutics. The structural versatility of this approach allows for the efficient generation of various substituted 1,4-diazepanes, positioning it as a vital asset for reliable pharmaceutical intermediate supplier networks aiming to secure stable supply chains for complex API precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
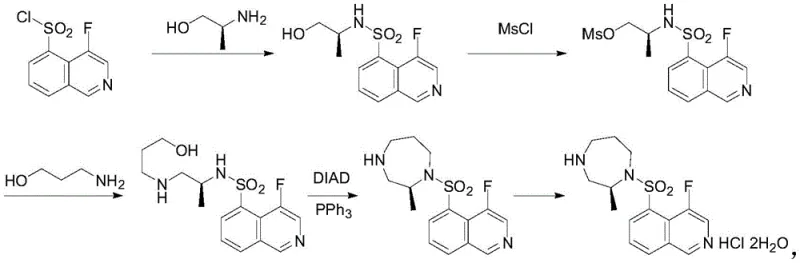
Historically, the synthesis of 1,4-diazepane derivatives has been plagued by economic and safety inefficiencies. One prevalent linear route initiates with 4-fluoroisoquinoline-5-sulfonyl chloride, an exceptionally expensive starting material. In this direct synthesis approach, the high-cost sulfonyl chloride is subjected to multiple reaction steps early in the sequence, meaning any yield loss in subsequent transformations results in disproportionate financial waste. Furthermore, linear processes often lack the modularity required for effective impurity purging, leading to carryover contaminants that complicate final crystallization. Alternatively, convergent routes attempting to build the seven-membered ring separately often rely on hazardous reagents such as methanesulfonyl chloride (MsCl) and azodicarboxylic acid diesters (e.g., DIAD). MsCl is a lachrymator and highly toxic, while DIAD poses severe explosion risks upon heating or impact. Additionally, these Mitsunobu-type conditions generate stoichiometric amounts of triphenylphosphine oxide (TPPO), a byproduct notoriously difficult to remove, thereby compromising the purity profile and increasing waste disposal costs significantly.
The Novel Approach
In stark contrast, the methodology disclosed in CN116143713A employs a strategic convergent synthesis that decouples the construction of the diazepane ring from the attachment of the expensive isoquinoline moiety until the final stages or utilizes alternative protecting groups that are easier to manage. The core innovation lies in the use of o-nitrobenzenesulfonyl chloride (Nosyl-Cl) as a temporary protecting and activating group. This reagent is not only more cost-effective than the isoquinoline counterpart but also possesses superior electronic properties that facilitate ring closure. The process avoids the use of explosive azo compounds and toxic mesyl chlorides entirely, replacing them with safer tosylating agents and ammonium iodide catalysts. By implementing a telescoped workflow where intermediates are not isolated between key alkylation and tosylation steps, the process minimizes solvent consumption and processing time. This results in a finished product with remarkably low impurity content and stable quality indices, effectively solving the purity and safety bottlenecks associated with traditional manufacturing.
Mechanistic Insights into Nosyl-Mediated Cyclization and Alkylation
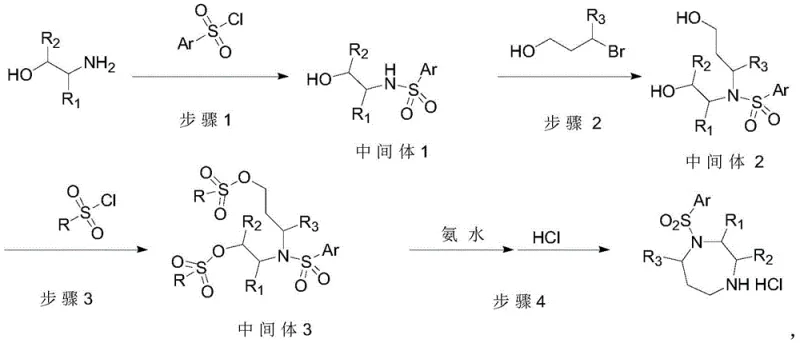
The chemical elegance of this synthesis is rooted in the specific reactivity of the o-nitrobenzenesulfonyl (Nosyl) group. In the initial step, the chiral amino alcohol undergoes nucleophilic attack on the sulfur atom of Nosyl-Cl, forming a sulfonamide. The strong electron-withdrawing nature of the ortho-nitro group activates the sulfonamide nitrogen, making it sufficiently nucleophilic for the subsequent alkylation with 3-bromopropanol in Step 2. This alkylation is catalyzed by tetrabutylammonium iodide (TBAI), which facilitates the displacement of the bromide via a Finkelstein-like mechanism, enhancing reaction rates in polar aprotic solvents like DMF. Following this, the terminal hydroxyl group is activated via tosylation using p-toluenesulfonyl chloride (TsCl) and DMAP. The resulting bis-electrophile precursor is primed for intramolecular cyclization. In the final step, treatment with ammonia induces a double nucleophilic substitution: ammonia attacks the tosylated carbon to displace the tosylate, and simultaneously, the Nosyl group acts as a leaving group (facilitated by the thiolate-like mechanism often seen with Nosyl deprotection under nucleophilic conditions), closing the seven-membered 1,4-diazepane ring. This mechanistic pathway ensures high regioselectivity and minimizes the formation of oligomeric byproducts.
Impurity control is inherently built into this mechanistic design. Traditional methods often struggle with isomeric impurities arising from non-selective alkylations or incomplete deprotections. In this novel route, the use of the Nosyl group provides a distinct chemical handle that can be selectively manipulated. The telescoping of Step 2 and Step 3 is particularly critical; by avoiding the isolation of the hydroxy-intermediate, the process prevents potential degradation or racemization that might occur during work-up. The reaction conditions are tightly controlled, with HPLC monitoring ensuring that starting materials like o-nitrobenzenesulfonyl chloride are consumed to levels below 1.0% before proceeding. The final cyclization in a pressure vessel at elevated temperatures (80-100°C) ensures complete conversion of the bis-tosylate intermediate, driving the equilibrium towards the desired cyclic amine. The resulting crude product exhibits high purity (up to 99.8% by HPLC in examples), demonstrating that the mechanistic choices effectively suppress side reactions and simplify the downstream purification landscape.
How to Synthesize 1,4-Diazepane Derivatives Efficiently
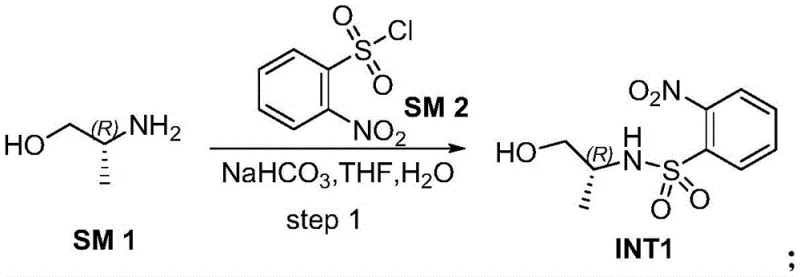
The synthesis of these valuable heterocyclic intermediates follows a logical four-step progression designed for maximum efficiency and minimal waste. The process begins with the protection of a chiral amino alcohol, followed by chain extension, activation, and finally cyclization. This sequence allows for the introduction of chirality at the very beginning using inexpensive chiral pool materials, ensuring the stereochemical integrity of the final API intermediate. The detailed standardized synthesis steps, including specific molar ratios, temperature controls, and work-up procedures, are outlined in the structured guide below, providing a clear roadmap for technical teams to replicate this high-performance protocol in pilot or production settings.
- Step 1: React chiral amino alcohol with o-nitrobenzenesulfonyl chloride using sodium bicarbonate in water/THF at 0-20°C to form Intermediate 1.
- Step 2: Alkylate Intermediate 1 with 3-bromopropanol using tetrabutylammonium iodide (TBAI) catalyst and potassium phosphate in DMF at 50-60°C to yield Intermediate 2.
- Step 3: Tosylate Intermediate 2 using p-toluenesulfonyl chloride (TsCl) and DMAP catalyst in DCM with triethylamine at 0-25°C to generate bis-tosylate Intermediate 3.
- Step 4: Cyclize Intermediate 3 with 25% ammonia water in acetonitrile at 90°C in a pressure vessel, followed by salt formation with HCl to obtain the final 1,4-diazepane hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic reduction of raw material risk and cost volatility. By eliminating the dependency on hazardous reagents like DIAD and MsCl, facilities can avoid the stringent storage, handling, and disposal costs associated with highly regulated toxic substances. This directly translates to cost reduction in pharmaceutical intermediate manufacturing, as the overhead for EHS compliance and waste treatment is significantly lowered. Furthermore, the avoidance of expensive starting materials in the early linear steps prevents capital tie-up in partially processed high-value inventory, improving cash flow and reducing financial exposure to batch failures.
- Cost Reduction in Manufacturing: The process utilizes commodity chemicals such as o-nitrobenzenesulfonyl chloride and 3-bromopropanol, which are readily available at competitive prices compared to specialized fluorinated isoquinoline sulfonyl chlorides. The telescoped nature of the synthesis reduces the number of isolation and drying steps, leading to significant savings in solvent usage, energy consumption, and labor hours. Additionally, the high purity of the crude product minimizes the need for extensive recrystallization or chromatographic purification, further driving down the cost of goods sold (COGS) and enhancing overall margin potential for the final API.
- Enhanced Supply Chain Reliability: Reliance on explosive or highly toxic reagents often creates single points of failure in the supply chain due to transportation restrictions or supplier shortages. By substituting these with stable, non-regulated alternatives, the manufacturing process becomes more resilient to external disruptions. The robustness of the reaction conditions, which tolerate minor variations without generating difficult-to-remove impurities, ensures consistent batch-to-batch quality. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, reducing lead time for high-purity pharmaceutical intermediates and preventing production stoppages caused by quality deviations.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, having been designed with industrial constraints in mind. The elimination of triphenylphosphine oxide waste, a major environmental burden in Mitsunobu reactions, simplifies effluent treatment and aligns with green chemistry principles. The ability to perform reactions in standard solvents like THF, DMF, and DCM without requiring cryogenic conditions or specialized high-pressure equipment (beyond standard autoclaves for ammonolysis) facilitates easy technology transfer from lab to plant. This scalability ensures that the reliable pharmaceutical intermediate supplier can meet surging demand for ROCK inhibitors without compromising on environmental standards or safety protocols.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 1,4-diazepane synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and chemical rationale behind the new method. Understanding these details is essential for R&D and procurement teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new process improve upon traditional Ripasudil intermediate synthesis?
A: Traditional linear routes consume expensive 4-fluoroisoquinoline-5-sulfonyl chloride early, leading to yield loss. Convergent routes often use hazardous reagents like azodicarboxylic acid diesters (DIAD) and methanesulfonyl chloride (MsCl). This new method avoids these toxic substances, utilizes cheaper starting materials, and employs a telescoped process that minimizes purification steps, resulting in higher overall purity (up to 99.8%) and safer operations.
Q: What are the key impurity control mechanisms in this synthesis?
A: The process utilizes o-nitrobenzenesulfonyl (Nosyl) protection which acts as a strong electron-withdrawing group, facilitating subsequent nucleophilic substitutions while suppressing side reactions. Furthermore, the telescoping of Steps 2 and 3 without intermediate isolation prevents the accumulation of impurities that typically arise during multiple work-up procedures, ensuring the final cyclization proceeds with high chemical fidelity.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for industrial scalability. It replaces explosive and highly toxic reagents with stable, commercially available alternatives like TsCl and TBAI. The reaction conditions (0-90°C) are manageable in standard stainless steel reactors, and the high tolerance for impurities in intermediate stages reduces the burden on QC and purification infrastructure, making it ideal for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Diazepane Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, safe, and scalable synthetic routes for complex heterocyclic intermediates like the 1,4-diazepane derivatives described in CN116143713A. Our team of expert chemists is well-versed in adapting such innovative patent technologies into robust commercial processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the exacting standards required for global pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your supply chain. By leveraging our expertise, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your drug development pipeline. Together, we can accelerate the delivery of life-saving medications to patients worldwide through superior chemical manufacturing.
