Scalable Synthesis of Artemisinin Intermediates Using Hydrogen Peroxide Oxidation Technology
Scalable Synthesis of Artemisinin Intermediates Using Hydrogen Peroxide Oxidation Technology
The global demand for effective antimalarial therapies continues to drive innovation in the synthesis of artemisinin and its derivatives. Patent CN102267973B introduces a transformative approach to constructing the critical peroxy bridge found in artemisinin, moving away from reliance on complex photo-oxidation techniques. This technology leverages hydrogen peroxide as a safe, inexpensive, and readily available oxygen source to functionalize sterically hindered epoxide precursors. By utilizing a specialized molybdenum-amino acid catalyst system, the process achieves high selectivity for the hydroperoxy group introduction at the C-12a position, a historically difficult transformation. This breakthrough not only simplifies the synthetic route but also aligns with modern green chemistry principles by eliminating the need for singlet oxygen generators. For pharmaceutical manufacturers, this represents a pivotal shift towards more robust and economically viable supply chains for essential antimalarial active pharmaceutical ingredients.
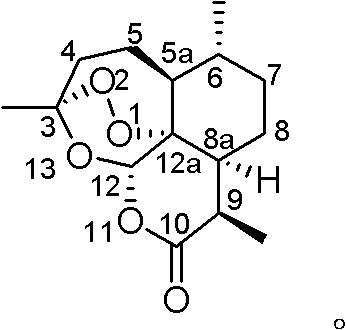
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total or semi-synthesis of artemisinin has been bottlenecked by the difficulty of introducing the endoperoxide bridge, which is essential for its biological activity. Traditional methods predominantly rely on photo-oxygenation using singlet oxygen, a process that requires specialized irradiation equipment and photosensitizers. This approach suffers from poor reproducibility, low throughput, and significant safety hazards associated with handling high-energy oxygen species on a large scale. Furthermore, the reaction often yields complex mixtures of by-products, necessitating rigorous and costly purification steps that erode overall process efficiency. The dependency on light penetration also limits reactor geometry, making scale-up from laboratory to industrial production notoriously difficult and capital-intensive. These factors collectively restrict the ability of suppliers to meet fluctuating global demand reliably and cost-effectively.
The Novel Approach
The methodology described in the patent circumvents these challenges by employing a chemical oxidation strategy using hydrogen peroxide. Instead of relying on light energy, the process utilizes a heterogeneous catalyst formed from sodium molybdate and amino acids to activate hydrogen peroxide for nucleophilic attack on the epoxide ring. This allows for the direct installation of the hydroperoxy moiety under mild thermal conditions, typically between -30°C and 30°C. The reaction demonstrates remarkable tolerance for multifunctional groups and steric bulk, successfully functionalizing the quaternary carbon center at C-12a which was previously inaccessible via standard hydroperoxylysis. This chemical route eliminates the need for photochemical reactors, allowing the use of standard stainless steel vessels and significantly lowering the barrier to entry for commercial manufacturing.
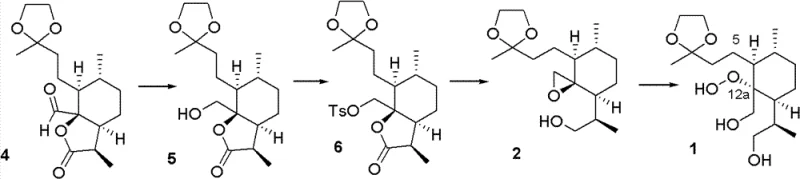
Mechanistic Insights into Molybdenum-Catalyzed Epoxide Hydroperoxylysis
The core of this technological advancement lies in the unique catalytic system that facilitates the ring-opening of the spiro-epoxide. The catalyst, generated in situ or pre-formed as a precipitate from Na2MoO4 and glycine, acts as a Lewis acid to coordinate with the epoxide oxygen, thereby increasing the electrophilicity of the adjacent carbons. Simultaneously, the molybdenum center activates the hydrogen peroxide molecule, enhancing its nucleophilicity. This dual activation is crucial for overcoming the significant steric hindrance presented by the substituents at the C-5a and C-8a positions of the decalin system. Unlike strong mineral acids which might lead to non-selective hydrolysis or rearrangement, this buffered catalytic environment directs the attack specifically to form the hydroperoxide at the desired tertiary carbon. The result is a clean transformation that preserves the integrity of other sensitive functional groups within the molecule.
Impurity control is inherently built into this mechanism due to the specificity of the catalyst-substrate interaction. In conventional acid-catalyzed openings, competing pathways such as pinacol rearrangements or simple hydration to diols are common, leading to difficult-to-separate impurities. However, the molybdenum-amino acid complex creates a specific coordination sphere that favors the formation of the O-O bond over C-O bond formation with water. Following the hydroperoxylysis, the resulting intermediate (Compound 1) can be smoothly cyclized under mild acid catalysis to form the 1,2,4-trioxane ring system (Compound 7). This sequential control ensures that the high-energy peroxy bond is formed early in a controlled manner and then locked into the stable cyclic structure, minimizing decomposition risks during downstream processing.
How to Synthesize Artemisinin Intermediate Efficiently
The synthesis pathway outlined in the patent provides a clear, step-by-step protocol for generating high-purity intermediates suitable for further conversion to artemisinin. The process begins with the reduction of a lactone precursor to a diol, followed by selective protection and activation steps to prepare the epoxide substrate. The critical oxidation step utilizes aqueous hydrogen peroxide extracted into an organic phase, ensuring efficient contact with the lipophilic substrate while maintaining safety. Subsequent acid-catalyzed cyclization closes the trioxane ring, yielding the advanced intermediate Compound 7. This modular approach allows for flexibility in production scheduling and quality control at each stage. Detailed standardized operating procedures for scaling this route from kilograms to tons are available upon request to qualified technical partners.
- Reduce the lactone substrate (Compound 4) using alkali metal borohydrides in alcoholic solvents at low temperatures to obtain the diol intermediate (Compound 5).
- Perform selective sulfonylation on Compound 5 using p-Toluenesulfonyl chloride and a base catalyst to activate the primary hydroxyl group, yielding Compound 6.
- Execute the critical epoxide hydroperoxylysis on the spiro-epoxide substrate using hydrogen peroxide and a molybdenum-amino acid catalyst to introduce the peroxy bond at C-12a.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this hydrogen peroxide-based synthesis offers profound strategic benefits beyond mere technical feasibility. The primary advantage is the drastic simplification of the manufacturing infrastructure required. By removing the dependency on photochemical reactors and singlet oxygen generators, facilities can utilize existing standard batch reactors, significantly reducing capital expenditure and maintenance costs. This compatibility with standard equipment also shortens the timeline for technology transfer and validation, allowing for faster ramp-up of production capacity to meet market surges. Furthermore, the use of commodity chemicals like hydrogen peroxide and common solvents ensures a stable and resilient supply chain, insulating production from the volatility associated with specialized reagents.
- Cost Reduction in Manufacturing: The elimination of expensive photo-sensitizers and specialized irradiation equipment leads to substantial operational cost savings. Additionally, the high selectivity of the molybdenum catalyst reduces the formation of by-products, which minimizes waste disposal costs and improves overall material throughput. The ability to run reactions at near-ambient temperatures also lowers energy consumption compared to processes requiring extreme cryogenic conditions or high-pressure oxygenation. These cumulative efficiencies translate into a more competitive cost structure for the final artemisinin intermediate, providing better margin protection for downstream API manufacturers.
- Enhanced Supply Chain Reliability: Sourcing hydrogen peroxide and molybdenum salts is far more reliable than securing specialized photochemical components or high-purity singlet oxygen precursors. This shift to commodity reagents mitigates the risk of supply disruptions caused by single-source vendor issues. Moreover, the robustness of the reaction conditions means that production is less susceptible to minor variations in environmental factors, ensuring consistent batch-to-batch quality. This reliability is critical for maintaining continuous supply to global health organizations and pharmaceutical partners who require strict adherence to delivery schedules for life-saving medications.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids the light-penetration limitations of photochemistry, allowing for larger reactor volumes without loss of efficiency. From an environmental perspective, the use of hydrogen peroxide generates water as the primary by-product, aligning with green chemistry initiatives and simplifying wastewater treatment protocols. The catalyst system can potentially be recovered and reused, further reducing the environmental footprint and hazardous waste generation. This compliance with stringent environmental regulations facilitates smoother permitting processes and enhances the sustainability profile of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this route into their existing pipelines. The answers are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What is the primary advantage of this synthesis method over traditional photo-oxidation?
A: This method replaces the need for expensive and hazardous singlet oxygen generation equipment with common hydrogen peroxide, significantly improving operational safety and reducing capital expenditure for reactor setup.
Q: How does the catalyst system handle steric hindrance at the C-12a position?
A: The patent utilizes a specific molybdenum-amino acid precipitate catalyst that effectively facilitates the ring-opening of sterically hindered spiro-epoxides, a transformation that typically fails with standard acidic conditions.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction conditions operate at mild temperatures (-30°C to 40°C) and utilize stable, commercially available reagents like 30% hydrogen peroxide, making it highly adaptable for metric-ton scale manufacturing without specialized photochemical infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Artemisinin Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced synthetic methodologies like the one described in CN102267973B to deliver high-quality pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped to analyze complex peroxy-containing compounds, guaranteeing that every batch meets the exacting standards required for antimalarial drug production. Our commitment to process safety and efficiency makes us an ideal partner for long-term supply agreements.
We invite global pharmaceutical companies and procurement officers to engage with our technical procurement team for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging this innovative hydrogen peroxide oxidation technology, we can help you optimize your supply chain and reduce overall manufacturing costs. Please contact us to request specific COA data, route feasibility assessments, and samples to evaluate the quality of our artemisinin intermediates. Let us collaborate to secure a sustainable and cost-effective supply of this critical life-saving medicine.
