Advanced Catalytic Oxidation of DFF to FDCA: A Breakthrough in Sustainable Polymer Intermediate Manufacturing
The global shift towards sustainable materials has intensified the search for bio-based alternatives to petroleum-derived monomers, with 2,5-furandicarboxylic acid (FDCA) emerging as a critical platform chemical for next-generation polyesters like PEF. A pivotal advancement in this domain is documented in Chinese patent CN111393397B, which discloses a highly efficient method for preparing FDCA via the catalytic oxidation of 2,5-diformylfuran (DFF). Unlike traditional routes that struggle with selectivity issues or rely on expensive noble metals, this invention leverages a novel composite catalyst system containing a nitrogen-oxygen radical generator. For procurement specialists and R&D leaders seeking a reliable bio-based polymer intermediate supplier, this technology represents a significant leap forward in process economics and environmental compliance, offering a pathway to high-purity FDCA without the drawbacks of corrosive promoters.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of carboxylic acids similar to FDCA, such as terephthalic acid (PTA), has relied heavily on the Amoco process which utilizes Cobalt-Manganese-Bromine (Co/Mn/Br) catalyst systems. While effective, the presence of bromine introduces severe challenges, including high corrosiveness to reactor vessels which necessitates the use of expensive titanium-lined equipment and increases maintenance downtime. Furthermore, alternative approaches using noble metal catalysts like Gold (Au), Platinum (Pt), or Palladium (Pd) often require alkaline additives to proceed efficiently, complicating downstream purification and generating substantial salt waste. When attempting to oxidize 5-hydroxymethylfurfural (HMF) directly, the differing reactivity of the hydroxymethyl and aldehyde groups often leads to incomplete oxidation or the formation of stubborn intermediates like 5-hydroxymethyl-2-furancarboxylic acid (HMFCA), thereby capping the overall yield and purity of the desired FDCA product.
The Novel Approach
The methodology outlined in the patent data circumvents these historical bottlenecks by employing DFF as the substrate instead of HMF and replacing the corrosive bromine promoter with a nitrogen-oxygen radical generator such as N-hydroxyphthalimide (NHPI). This strategic shift allows the reaction to proceed under relatively mild conditions, typically between 60°C and 160°C, using molecular oxygen or air as the sole oxidant. The absence of bromine not only extends the lifespan of standard stainless steel reactors but also simplifies the waste stream, aligning perfectly with modern green chemistry principles. By optimizing the molar ratios of the Cobalt and Manganese co-catalysts alongside the NHPI promoter, the process achieves DFF conversion rates exceeding 99% with FDCA selectivity reaching as high as 98.7%, demonstrating a robust and scalable solution for cost reduction in green chemical manufacturing.
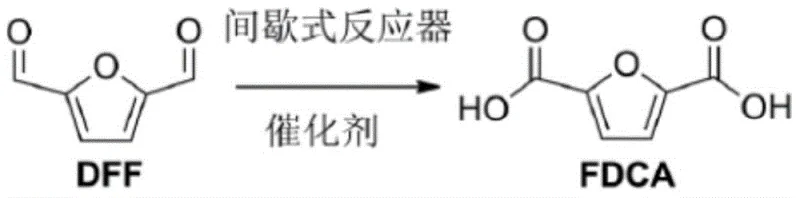
Mechanistic Insights into Co/Mn/NHPI Catalyzed Oxidation
The core of this technological breakthrough lies in the synergistic interaction between the transition metals and the organic radical generator. In this catalytic cycle, the N-hydroxyphthalimide (NHPI) acts as a precursor that, under the assistance of the Cobalt and Manganese species, generates the active phthalimide-N-oxyl (PINO) radical. This PINO radical is highly effective at abstracting hydrogen atoms from the aldehyde groups of the DFF substrate, initiating the oxidation sequence. The Cobalt and Manganese ions function as redox mediators, facilitating the regeneration of the PINO radical from NHPI by reacting with molecular oxygen, thus sustaining the catalytic cycle without being consumed. This mechanism ensures that the oxidation proceeds rapidly and selectively at the aldehyde positions, avoiding the random oxidative degradation of the furan ring that often plagues less controlled oxidation processes.
From an impurity control perspective, the choice of DFF as the starting material is mechanistically superior to HMF. Because DFF lacks the hydroxymethyl group, the pathway eliminates the formation of HMFCA, a common bottleneck intermediate that often requires harsh conditions to oxidize further. The kinetic data suggests that the aldehyde-to-acid oxidation is the rate-determining step, and the specific electronic environment created by the Co/Mn/NHPI complex lowers the activation energy for this specific transformation. Consequently, the formation of ring-opening byproducts like maleic anhydride (MA) is minimized, typically remaining below 3% even at high conversions. This high specificity is crucial for downstream polymerization applications where trace impurities can act as chain terminators, affecting the molecular weight and mechanical properties of the final bio-plastic.
How to Synthesize 2,5-Furandicarboxylic Acid Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the efficiency of the radical generation and oxygen transfer. The patent provides a clear framework for operationalizing this chemistry, emphasizing the importance of oxygen partial pressure and temperature modulation to balance reaction rate against selectivity. Operators must ensure that the molar ratio of the nitrogen-oxygen radical generator to the total metal catalyst is maintained within the optimal window of 1:1 to 10:1 to prevent radical quenching or insufficient initiation. The following guide outlines the standardized procedure derived from the experimental examples to ensure reproducible high-yield results suitable for pilot and commercial scale operations.
- Charge a pressure reactor with 2,5-diformylfuran (DFF), a composite catalyst comprising Cobalt acetate, Manganese acetate, and N-hydroxyphthalimide (NHPI), and acetic acid solvent.
- Seal the reactor and pressurize with oxygen or air to a partial pressure between 0.2 MPa and 1.5 MPa to ensure sufficient oxidant availability.
- Heat the reaction mixture to a temperature range of 80°C to 140°C with stirring for 1 to 3 hours, then cool and analyze for FDCA yield and DFF conversion.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain directors and procurement managers, the adoption of this DFF oxidation technology offers tangible strategic benefits beyond mere chemical yield. The primary advantage stems from the drastic simplification of the catalyst system, which eliminates the need for precious metals and corrosive halides. This shift directly translates to a more resilient supply chain, as Cobalt and Manganese salts are commodity chemicals with stable pricing and widespread availability, unlike the volatile market for Gold or Platinum group metals. Furthermore, the operational safety profile is enhanced by the ability to use air or low-pressure oxygen rather than aggressive stoichiometric oxidants like nitric acid, reducing the regulatory burden and insurance costs associated with hazardous material handling and storage.
- Cost Reduction in Manufacturing: The economic impact of replacing noble metal catalysts with base metals cannot be overstated in terms of long-term operational expenditure. By utilizing inexpensive Cobalt and Manganese acetates alongside organic promoters like NHPI, the direct material cost of the catalyst package is significantly reduced compared to Au or Pt-based systems. Additionally, the elimination of bromine promoters removes the necessity for specialized corrosion-resistant alloys in reactor construction, allowing for the use of standard high-grade stainless steel equipment which represents a substantial capital expenditure saving. The high selectivity of the reaction also minimizes the loss of valuable DFF feedstock to byproducts, ensuring that a greater proportion of the raw material input is converted into saleable high-purity FDCA product.
- Enhanced Supply Chain Reliability: Relying on biomass-derived DFF as a feedstock diversifies the raw material base away from fluctuating petrochemical markets. Since DFF can be produced from abundant sugars like glucose and fructose, the upstream supply chain is anchored in renewable agriculture rather than fossil fuel extraction, providing a hedge against oil price volatility. The mild reaction conditions (60°C to 160°C) and the use of acetic acid as a common solvent mean that the process can be easily integrated into existing fine chemical infrastructure without requiring bespoke high-temperature or high-pressure units. This compatibility accelerates the timeline for technology transfer and scale-up, ensuring that production capacity can be ramped up quickly to meet market demand without lengthy lead times for custom equipment fabrication.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly lower than conventional methods, primarily due to the use of molecular oxygen which produces water as the only byproduct of the oxidant reduction. The absence of heavy metal waste streams associated with noble metal catalysts simplifies effluent treatment and reduces the cost of waste disposal. Moreover, the high conversion rates achieved (>99%) mean that unreacted starting material recycling loops can be minimized or eliminated, streamlining the process flow. This alignment with green chemistry metrics makes the resulting FDCA highly attractive for end-users in the packaging and textile industries who are under increasing pressure to certify the sustainability credentials of their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic oxidation technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on process robustness and product quality. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is DFF preferred over HMF as a starting material for FDCA production in this patent?
A: DFF (2,5-diformylfuran) possesses only aldehyde functional groups, whereas HMF contains both hydroxymethyl and aldehyde groups with different reactivities. This structural simplicity in DFF minimizes side reactions such as self-polymerization and the formation of intermediates like HMFCA, leading to significantly higher selectivity and purity of the final FDCA product compared to direct HMF oxidation.
Q: Does this catalytic system require corrosive bromine promoters?
A: No, a key innovation of patent CN111393397B is the elimination of bromine promoters. By utilizing a nitrogen-oxygen radical generator like NHPI in synergy with Cobalt and Manganese, the process achieves high oxidation efficiency without the severe equipment corrosion associated with traditional bromine-containing Amoco-type processes.
Q: How does this method compare to noble metal catalysts in terms of cost?
A: This method utilizes abundant and inexpensive base metals (Cobalt and Manganese) instead of costly noble metals like Gold, Platinum, or Palladium. This substitution drastically reduces the raw material cost of the catalyst system, making the process economically superior for large-scale industrial applications while maintaining high conversion rates above 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Furandicarboxylic Acid Supplier
As the demand for bio-based polymers continues to surge, securing a stable supply of high-quality monomers like FDCA is paramount for maintaining competitive advantage in the materials sector. NINGBO INNO PHARMCHEM stands ready to support your development needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for polymerization-grade intermediates. We understand the critical nature of supply continuity and have optimized our logistics to ensure timely delivery of specialty chemicals to global partners.
We invite you to engage with our technical procurement team to discuss how this advanced oxidation technology can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits specific to your operation. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your transition to sustainable bio-based materials is both technically sound and commercially viable.
