Revolutionizing Chiral Beta-Amino Synthesis: A Metal-Free Organocatalytic Strategy for Commercial Scale Production
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral scaffolds, particularly beta-amino derivatives, which serve as critical building blocks for bioactive molecules. Patent CN109748883B introduces a groundbreaking approach to synthesizing these valuable structures through an asymmetric ring-opening reaction of azetidine quaternary ammonium salts. Unlike traditional methods that often rely on expensive transition metal catalysts or harsh reaction conditions, this invention leverages a chiral phosphoric acid organocatalytic system to achieve high stereoselectivity under mild conditions. The core innovation lies in utilizing the azetidine quaternary ammonium salt not merely as a catalyst but as a reactive substrate, enabling a divergent synthesis pathway that significantly broadens the scope of accessible chemical space. This technological advancement addresses long-standing challenges in stereochemical control and functional group compatibility, positioning it as a vital tool for modern medicinal chemistry and process development teams aiming for efficient route design.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral beta-amino derivatives has been fraught with significant technical hurdles that impede efficient large-scale manufacturing. Conventional strategies frequently depend on transition metal catalysis, which introduces the risk of heavy metal contamination in the final active pharmaceutical ingredient (API), necessitating costly and time-consuming purification steps to meet regulatory limits. Furthermore, many existing protocols require pre-functionalized chiral substrates or extreme reaction conditions such as high temperatures and strong bases, which severely limit the tolerance for sensitive functional groups often present in complex drug candidates. These harsh environments can lead to decomposition, racemization, or unwanted side reactions, resulting in lower overall yields and increased waste generation. The reliance on stoichiometric chiral auxiliaries in some older methods further exacerbates the atom economy issues, making the process less sustainable and economically viable for commercial production where cost efficiency is paramount.
The Novel Approach
The methodology disclosed in CN109748883B represents a paradigm shift by employing a metal-free organocatalytic system that operates effectively at room temperature or slightly cooled conditions (5°C). By utilizing a chiral spiro-cycle phosphoric acid catalyst in conjunction with an inorganic base like disodium hydrogen phosphate, the reaction facilitates a highly selective ion-exchange mechanism. This unique interaction promotes the asymmetric ring-opening of the azetidine ring with excellent stereocontrol, bypassing the need for toxic metals. The process demonstrates remarkable versatility, accommodating a wide range of nucleophiles including mercaptobenzothiazoles, and tolerates diverse substituents on the azetidine scaffold. This mild yet powerful approach not only simplifies the operational workflow but also enhances the safety profile of the synthesis, making it an attractive alternative for the production of high-purity chiral intermediates required in the pharmaceutical sector.
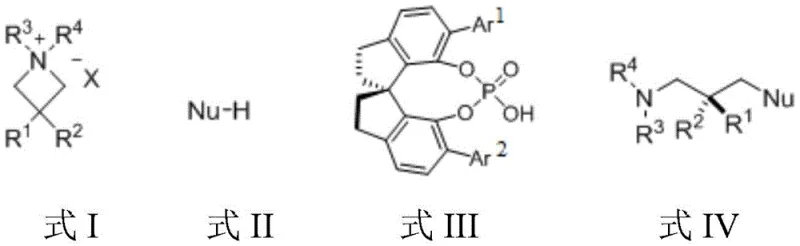
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Ring-Opening
The success of this transformation hinges on the intricate interplay between the chiral phosphoric acid catalyst and the azetidine quaternary ammonium salt substrate. Mechanistically, the chiral phosphoric acid acts as a Brønsted acid that engages in hydrogen bonding or ion-pairing interactions with the nucleophile and the substrate. Crucially, the inorganic base facilitates the formation of a chiral phosphate salt through anion exchange with the quaternary ammonium counterion. This generates a tight chiral ion pair in situ, which creates a well-defined chiral environment around the electrophilic center of the azetidine ring. As the nucleophile attacks, the chiral phosphate anion directs the trajectory of the approach, ensuring that the ring-opening occurs with high facial selectivity. This ion-pairing strategy effectively overcomes the inherent difficulty of controlling stereochemistry in intermolecular reactions involving flexible acyclic intermediates formed after ring opening. The rigid spiro-cycle backbone of the catalyst further restricts conformational freedom, locking the transition state into a favorable geometry that maximizes enantioselectivity while minimizing background non-catalyzed reactions.
Impurity control is inherently built into this mechanistic design due to the high specificity of the chiral catalyst. The mild reaction conditions prevent thermal degradation of sensitive functional groups, which is a common source of impurities in harsher metal-catalyzed processes. Moreover, the absence of transition metals eliminates the formation of metal-complexed byproducts that are notoriously difficult to remove. The use of a specific inorganic base ensures that the pH of the reaction medium remains optimal for the catalytic cycle without promoting hydrolysis or other decomposition pathways. By carefully tuning the catalyst loading and base equivalents, the process suppresses racemic background reactions, ensuring that the final product profile is dominated by the desired enantiomer. This high level of chemical and stereochemical purity reduces the burden on downstream purification, allowing for simpler workup procedures such as direct column chromatography to isolate the target compound in high yield and optical purity.
How to Synthesize Chiral Beta-Amino Derivatives Efficiently
To implement this synthesis effectively, one must adhere to the optimized parameters outlined in the patent to ensure reproducibility and high performance. The process begins with the precise weighing of the 3-substituted azetidine quaternary ammonium salt and the chosen nucleophile, which are then dissolved in a suitable organic solvent such as trifluorotoluene. The addition of the chiral phosphoric acid catalyst and the inorganic base must be done under controlled conditions to initiate the ion-exchange process effectively. Maintaining the reaction temperature within the specified range is critical for balancing reaction rate and stereoselectivity. Following the reaction period, standard aqueous workup and purification techniques are employed to isolate the product. For detailed operational specifics regarding reagent grades, stirring rates, and exact quenching procedures, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by dissolving the 3-substituted azetidine quaternary ammonium salt and the nucleophile (e.g., 2-mercaptoarylthiazole) in an organic solvent such as trifluorotoluene.
- Add an inorganic base like disodium hydrogen phosphate (Na2HPO4) and a chiral phosphoric acid catalyst (CPA) to the solution to facilitate ion exchange and asymmetric induction.
- Stir the reaction at room temperature or 5°C for a specified period (e.g., 96 hours) to achieve high enantioselectivity, followed by purification via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this organocatalytic methodology offers substantial strategic benefits that align with modern manufacturing goals of sustainability and cost efficiency. The elimination of precious transition metals from the synthetic route directly translates to reduced raw material costs and removes the need for specialized scavenging resins or complex filtration systems typically required to meet residual metal specifications. This simplification of the purification train significantly shortens the production cycle time and lowers the overall cost of goods sold (COGS). Furthermore, the use of stable, commercially available inorganic bases and organocatalysts enhances supply chain reliability by reducing dependence on volatile metal markets or single-source catalyst suppliers. The robustness of the reaction conditions also implies a lower risk of batch failure due to sensitivity to moisture or oxygen, thereby improving manufacturing throughput and consistency.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the associated purification infrastructure leads to significant cost savings in the production of pharmaceutical intermediates. By avoiding the need for rigorous metal removal steps, manufacturers can reduce solvent consumption and waste disposal costs, contributing to a leaner and more economical process. The high yields reported in the patent examples further enhance material efficiency, ensuring that a greater proportion of input materials are converted into valuable product rather than waste.
- Enhanced Supply Chain Reliability: Utilizing widely available organocatalysts and inorganic salts mitigates supply risks associated with scarce metal resources. This stability allows for more predictable procurement planning and inventory management, ensuring continuous production schedules without interruptions caused by raw material shortages. The mild reaction conditions also reduce energy consumption for heating or cooling, adding another layer of operational stability and cost predictability to the manufacturing process.
- Scalability and Environmental Compliance: The metal-free nature of this synthesis simplifies regulatory compliance regarding heavy metal limits in drug substances, facilitating faster approval timelines. The process generates less hazardous waste compared to traditional metal-catalyzed routes, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing facility. This eco-friendly profile is increasingly valued by global partners and regulators, enhancing the marketability of the final products derived from this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide accurate guidance for R&D and process teams. Understanding these nuances is essential for successfully adapting this methodology to specific project requirements and ensuring optimal outcomes in both laboratory and pilot plant settings.
Q: What are the key advantages of using chiral phosphoric acid catalysts over transition metals for this synthesis?
A: The primary advantage is the elimination of toxic heavy metal residues, which simplifies downstream purification and ensures compliance with stringent pharmaceutical impurity guidelines. Additionally, the organocatalytic system operates under milder conditions, enhancing functional group tolerance.
Q: Can this methodology be scaled for commercial production of pharmaceutical intermediates?
A: Yes, the process utilizes readily available inorganic bases and stable organocatalysts without requiring cryogenic temperatures or inert atmosphere techniques for the initial setup, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What is the typical enantiomeric excess (ee) achieved with this protocol?
A: The patent data demonstrates that optimized conditions using specific chiral spiro-cycle phosphoric acid catalysts can consistently achieve enantiomeric excess values ranging from 85% to 96%, ensuring high optical purity for chiral drug synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Beta-Amino Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced synthetic technologies to deliver high-quality chemical solutions to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN109748883B can be seamlessly translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral beta-amino derivatives meets the highest standards required for pharmaceutical applications. Our commitment to quality and technical excellence makes us a trusted partner for companies seeking reliable sources of complex chiral intermediates.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your development timeline and optimize your supply chain efficiency. Let us help you transform this promising academic innovation into a commercial reality.
