Advanced Chiral Synthesis of (R)-Selenomethylselenocysteine for Commercial Scale-Up
Introduction to Next-Generation Selenium Supplementation Technology
The escalating global demand for high-bioavailability selenium supplements has driven significant innovation in organic synthesis, particularly for optically active compounds like (R)-selenomethylselenocysteine. As detailed in patent CN110683976A, a groundbreaking methodology has emerged that fundamentally shifts the paradigm from hazardous, racemic syntheses to a streamlined, chiral-pool approach. This technology leverages L-serine as a foundational building block, ensuring that the stereochemical integrity of the final product is maintained throughout the entire synthetic sequence without the need for complex resolution steps. For R&D directors and procurement strategists, this represents a critical advancement, offering a pathway to produce high-purity nutritional ingredients with superior safety profiles and reduced environmental impact compared to legacy processes.
The significance of this patent lies not just in the chemical novelty, but in its direct address of the bottlenecks plaguing current industrial production. Traditional methods often struggle with low yields, toxic reagents, and the inability to consistently deliver the biologically active L-configuration (or R-configuration in specific nomenclature contexts) without expensive chiral chromatography. By establishing a robust route through a stable diketopiperazine intermediate, this process guarantees a reliable supply of this potent anticancer and antioxidant agent. As a reliable nutritional ingredients supplier, understanding these mechanistic underpinnings is essential for evaluating the long-term viability and cost-effectiveness of sourcing strategies for selenium-based therapeutics and dietary supplements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing selenomethylselenocysteine have been fraught with substantial chemical and operational hazards that render them increasingly obsolete for modern GMP manufacturing. The most prevalent legacy method, the methylselenoacetaldehyde process, relies heavily on the use of cyanide salts for cyclization, introducing severe safety risks related to toxicity and waste disposal that complicate regulatory compliance. Furthermore, these older pathways typically yield a racemic DL-mixture, necessitating a downstream resolution step to isolate the biologically active enantiomer, which inherently caps the maximum theoretical yield at 50% and drastically inflates production costs. Alternative routes involving alpha-amino acrylic acid derivatives suffer from the scarcity and high price of starting materials, alongside the instability of methylselenol salts which are difficult to purify and handle on a large scale.
Additionally, methods utilizing liquid bromine or sodium metal in liquid ammonia require cryogenic conditions and specialized equipment, creating significant barriers to entry for standard chemical manufacturers. The reliance on such harsh conditions not only increases energy consumption but also poses serious threats to equipment integrity and operator safety. Consequently, the industry has long sought a method that eliminates these toxic reagents, avoids the inefficiency of racemate resolution, and utilizes readily available chiral precursors. The inability of these conventional methods to scale efficiently has resulted in supply chain fragility and volatile pricing for high-purity selenium amino acids, limiting their broader application in preventative medicine and nutraceuticals.
The Novel Approach
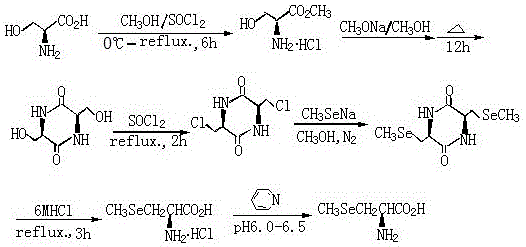
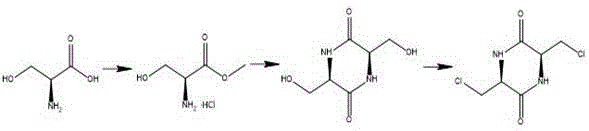
In stark contrast to these cumbersome legacy techniques, the novel approach disclosed in the patent utilizes a clever chiral pool strategy starting from L-serine, a ubiquitous and inexpensive amino acid. This method constructs a rigid (3S,6S)-3,6-dichloromethyl-2,5-diketopiperazine scaffold which serves as a perfect template for stereospecific nucleophilic substitution. By converting the hydroxyl groups of the serine-derived backbone into chloromethyl leaving groups, the process sets the stage for a clean displacement reaction with sodium methylselenolate. This strategic design ensures that the chirality of the starting material is faithfully transferred to the final product, completely bypassing the need for resolution and theoretically doubling the efficiency compared to racemic synthesis routes.
Moreover, the operational simplicity of this new route is a major advantage for cost reduction in nutritional ingredients manufacturing. The reactions proceed in common alcoholic solvents under reflux conditions, avoiding the need for cryogenic cooling or ultra-high pressure systems. The generation of the selenium nucleophile is performed in situ using sodium borohydride and dimethyl diselenide, which mitigates the handling risks associated with volatile methylselenol gas. This one-pot generation and immediate consumption of the reactive selenium species enhances overall process safety and yield. The final hydrolysis step is straightforward, utilizing standard hydrochloric acid to open the ring and reveal the free amino acid, followed by simple pH adjustment and recrystallization to achieve pharmaceutical-grade purity.
Mechanistic Insights into Diketopiperazine-Mediated Selenation
The core of this synthetic innovation lies in the formation and reactivity of the 2,5-diketopiperazine ring system, which acts as a protecting group and a conformational lock during the selenium incorporation step. Initially, L-serine methyl ester undergoes a base-catalyzed cyclization to form the piperazine ring, effectively masking the amine and carboxylic acid functionalities while positioning the side-chain hydroxymethyl groups for activation. Subsequent treatment with a chlorinating agent like thionyl chloride converts these primary alcohols into reactive chloromethyl groups. This transformation is critical because it creates excellent leaving groups that are susceptible to nucleophilic attack by soft nucleophiles like selenolates, facilitating the substitution reaction under relatively mild thermal conditions.
The nucleophilic substitution mechanism involves the attack of the methylselenolate anion (generated from dimethyl diselenide and sodium borohydride) on the chloromethyl carbons of the diketopiperazine intermediate. This SN2-type displacement proceeds with inversion of configuration at the carbon center, but since the starting material is derived from L-serine and the reaction targets the side chain rather than the alpha-carbon, the overall stereochemical outcome preserves the desired optical activity. The use of a one-pot method for generating the sodium methylselenolate ensures that the concentration of the reactive selenium species is optimized for the substitution, minimizing side reactions such as oxidation or elimination. Following the substitution, acidic hydrolysis cleaves the amide bonds of the diketopiperazine ring, regenerating the free amine and carboxylic acid to yield the final (R)-selenomethylselenocysteine structure with high fidelity.
How to Synthesize (R)-Selenomethylselenocysteine Efficiently
The execution of this synthesis requires precise control over reaction parameters, particularly during the chlorination and hydrolysis stages, to maximize yield and minimize impurity formation. The process begins with the esterification of L-serine, followed by cyclization to form the piperazine core, and then chlorination to activate the side chains. The subsequent selenation step must be conducted under an inert nitrogen atmosphere to prevent oxidation of the selenium species. Detailed standard operating procedures regarding temperature gradients, stoichiometric ratios, and workup protocols are essential for reproducibility. For a comprehensive breakdown of the specific reaction conditions and purification techniques validated in the patent examples, please refer to the standardized synthesis guide below.
- Prepare (3S,6S)-3,6-dichloromethyl-2,5-diketopiperazine from L-serine via esterification, cyclization with sodium alkoxide, and chlorination with thionyl chloride.
- Generate sodium methylselenolate in situ by reacting dimethyl diselenide with sodium borohydride and sodium metal in an alcoholic solvent under nitrogen.
- Perform nucleophilic substitution between the dichloro-diketopiperazine and sodium methylselenolate, followed by acid hydrolysis and pH adjustment to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers profound advantages that directly address the pain points of cost volatility and supply insecurity in the fine chemicals sector. By shifting the raw material base to L-serine, a commodity chemical produced on a massive scale for the food and pharma industries, the process decouples production from the fluctuating markets of exotic or hazardous precursors. This fundamental change in the bill of materials leads to a significantly reduced cost of goods sold (COGS), allowing for more competitive pricing structures without compromising margin. Furthermore, the elimination of toxic cyanide and the avoidance of cryogenic operations simplify the environmental, health, and safety (EHS) compliance burden, reducing the overhead costs associated with waste treatment and specialized facility maintenance.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the high atom economy and the removal of the resolution step. In traditional racemic synthesis, half of the produced material is the unwanted enantiomer, representing a total loss of raw materials and processing energy; this new method theoretically doubles the yield per batch by starting with the correct chirality. Additionally, the use of inexpensive reagents like sodium borohydride and thionyl chloride, combined with the ability to recycle solvents like methanol and ethanol, contributes to substantial cost savings. The simplified purification process, relying on filtration and recrystallization rather than complex chromatography, further lowers the operational expenditure required to bring the product to market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of globally available starting materials that are not subject to the same geopolitical or logistical constraints as specialized organoselenium reagents. L-serine and dimethyl diselenide are established commodities with multiple qualified suppliers worldwide, mitigating the risk of single-source dependency. The robustness of the reaction conditions—operating at atmospheric pressure and moderate temperatures—means that production can be easily transferred between different manufacturing sites or scaled up in existing multipurpose reactors without requiring capital-intensive retrofitting. This flexibility ensures consistent delivery schedules and reduces the lead time for high-purity nutritional ingredients even during periods of market disruption.
- Scalability and Environmental Compliance: The green chemistry attributes of this process align perfectly with modern sustainability mandates, making it easier to secure regulatory approvals and maintain social license to operate. By eliminating cyanide and reducing the generation of heavy metal waste, the process minimizes the environmental footprint of production. The mild reaction conditions also translate to lower energy consumption for heating and cooling, supporting corporate carbon reduction goals. From a scalability standpoint, the linear nature of the synthesis and the stability of the intermediates allow for seamless transition from pilot plant batches to multi-ton commercial production, ensuring that supply can grow in tandem with market demand for selenium-based therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications for manufacturing partners. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement officers assessing the quality assurances of potential suppliers.
Q: Why is the diketopiperazine route superior to the methylselenoacetaldehyde method?
A: The traditional methylselenoacetaldehyde method relies on highly toxic cyanide and produces a racemic mixture requiring difficult resolution. The novel diketopiperazine route utilizes L-serine as a chiral pool starter, ensuring optical purity from the outset and eliminating the need for hazardous cyanide reagents, thereby significantly improving safety and environmental compliance.
Q: How does this process address the instability of methylselenol salts?
A: The patent describes a one-pot generation of sodium methylselenolate using sodium borohydride and dimethyl diselenide, which is immediately reacted with the dichloro-intermediate. This in situ generation avoids the isolation and storage of unstable methylselenol salts, reducing decomposition risks and simplifying the operational workflow for large-scale production.
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the method is explicitly designed for industrial application. It uses commercially available raw materials like L-serine and dimethyl diselenide, operates under mild reaction conditions (reflux in alcohol), and avoids extreme temperatures or pressures. The high yields and straightforward purification steps (filtration and recrystallization) make it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-Selenomethylselenocysteine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality oversight. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields reported in patent CN110683976A can be reliably replicated on an industrial scale. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify optical rotation, elemental composition, and impurity profiles for every batch of (R)-selenomethylselenocysteine we produce.
We invite forward-thinking partners to collaborate with us to leverage this advanced synthesis technology for your next-generation selenium products. Whether you require custom synthesis services or bulk supply of this critical nutritional intermediate, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can strengthen your supply chain and accelerate your product development timelines.
