Advanced Catalytic Synthesis of 2,5-Dichloro-p-phenylenediamine for Industrial Scale Manufacturing
The chemical industry is constantly evolving towards greener, more efficient synthetic pathways, particularly for high-volume intermediates like 2,5-dichloro-p-phenylenediamine. Patent CN1974540B introduces a transformative preparation process that addresses critical bottlenecks in traditional manufacturing, specifically targeting the environmental and yield limitations associated with acylation and reduction steps. This technology leverages a sophisticated four-step sequence—acylation, nitration, hydrolysis, and reduction—to convert 2,5-dichloroaniline into the target diamine with exceptional efficiency. By shifting from conventional acetic acid solvents to recyclable halogenated hydrocarbons and replacing polluting iron powder reduction with a catalytic hydrazine system, this method represents a significant leap forward in sustainable chemical engineering. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for securing a reliable supply chain of high-purity intermediates used in dyes, polymers, and pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,5-dichloro-p-phenylenediamine has been plagued by severe environmental and economic inefficiencies inherent to legacy processing techniques. The traditional acylation step typically employs glacial acetic acid as a solvent at elevated temperatures ranging from 105°C to 110°C, which not only consumes significant energy but also generates large volumes of acidic waste liquid that are difficult and costly to treat. Furthermore, the subsequent reduction step conventionally relies on iron powder in hydrochloric acid, a method notorious for producing massive quantities of iron sludge waste. This iron mud creates a substantial disposal burden, complicating wastewater treatment protocols and increasing the overall carbon footprint of the manufacturing process. Additionally, the cumulative yield of these older four-step methods often stagnates around 59%, indicating significant material loss at each stage and driving up the cost of goods sold for the final active ingredient.
The Novel Approach
The innovative process detailed in the patent data fundamentally re-engineers the synthetic route to mitigate these drawbacks through solvent optimization and catalytic advancement. In the initial acylation phase, the method substitutes glacial acetic acid with halogenated hydrocarbons such as 1,2-dichloroethane, allowing the reaction to proceed under much milder conditions at 70-80°C. This shift not only lowers energy consumption but also enables the solvent to be recovered via azeotropic distillation with a recovery rate exceeding 90%, creating a closed-loop system that drastically reduces raw material waste. 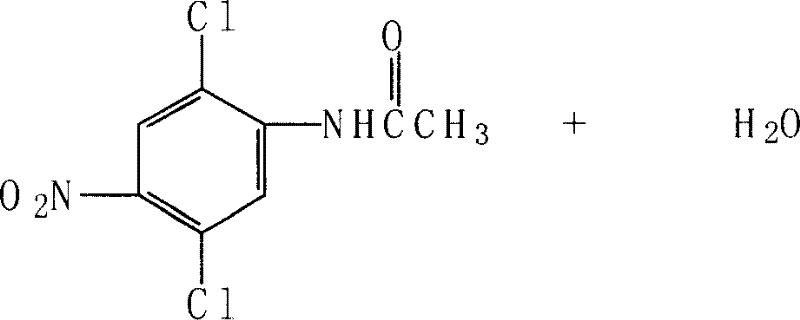 The most critical improvement, however, lies in the final reduction step, where the hazardous iron powder method is replaced by a catalytic reduction using hydrazine hydrate. This system utilizes ferric chloride and activated carbon as a catalyst in an ethanol solvent, completely eliminating the generation of iron sludge while boosting the reduction yield to over 90%. This holistic approach ensures a total process yield between 72.3% and 77.9%, offering a commercially superior alternative for the production of high-purity pharma intermediates.
The most critical improvement, however, lies in the final reduction step, where the hazardous iron powder method is replaced by a catalytic reduction using hydrazine hydrate. This system utilizes ferric chloride and activated carbon as a catalyst in an ethanol solvent, completely eliminating the generation of iron sludge while boosting the reduction yield to over 90%. This holistic approach ensures a total process yield between 72.3% and 77.9%, offering a commercially superior alternative for the production of high-purity pharma intermediates.
Mechanistic Insights into FeCl3-Catalyzed Hydrazine Reduction
The core technological breakthrough of this synthesis lies in the chemoselective reduction of the nitro group in the presence of chlorine substituents, a transformation that is notoriously difficult due to the risk of dehalogenation. The patent specifies a catalytic system comprising ferric chloride (FeCl3) and activated carbon, which facilitates the transfer of hydrogen from hydrazine hydrate to the nitro group with high specificity. Unlike heterogeneous metal catalysts like Raney Nickel or Pd/C, which can sometimes promote hydrodechlorination side reactions leading to impure products, this iron-based catalytic cycle maintains the integrity of the carbon-chlorine bonds. The activated carbon serves as a high-surface-area support that disperses the iron species, enhancing the contact efficiency between the catalyst, the hydrazine reducing agent, and the nitro-substrate dissolved in ethanol. This mechanism allows the reaction to proceed smoothly under reflux conditions for 1 to 3 hours, achieving conversion rates that push the yield of 2,5-dichloro-p-phenylenediamine to 90-93%.
Furthermore, the mechanistic pathway ensures exceptional impurity control, which is paramount for applications in sensitive fields like hair dyes and photographic chemicals. The use of hydrazine hydrate as the reducing agent results in nitrogen gas and water as the primary byproducts, alongside ammonium nitrate, which are far easier to separate from the organic product than the complex sludge mixtures formed by iron reduction. The ethanol solvent system further aids in purification, as the product can be crystallized directly from the reaction mixture upon cooling and dilution with water. This streamlined work-up procedure minimizes the number of unit operations required, thereby reducing the potential for product degradation or contamination during isolation. The result is a final product with HPLC purity consistently greater than 99%, meeting the stringent quality standards required by global regulatory bodies for fine chemical intermediates.
How to Synthesize 2,5-Dichloro-p-phenylenediamine Efficiently
Implementing this advanced synthetic route requires precise control over reaction parameters, particularly temperature and stoichiometry, to maximize the benefits of the catalytic system. The process begins with the acylation of 2,5-dichloroaniline using acetic anhydride in a halogenated solvent, followed by nitration with mixed acid at low temperatures to ensure regioselectivity. Subsequent alkaline hydrolysis removes the protecting group, yielding the nitro-intermediate ready for the critical reduction step. The detailed standardized synthesis steps, including specific molar ratios and safety protocols for handling hydrazine hydrate, are outlined below to guide process engineers in scaling this technology.
- Perform acylation of 2,5-dichloroaniline using acetic anhydride in a halogenated hydrocarbon solvent at 70-80°C.
- Conduct nitration of the acylated product using mixed acid (sulfuric and nitric acid) at low temperatures (5-10°C).
- Execute alkaline hydrolysis using sodium hydroxide solution followed by catalytic reduction with hydrazine hydrate and FeCl3/activated carbon.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates into tangible strategic advantages regarding cost stability and operational reliability. The elimination of iron sludge waste removes a significant variable cost associated with hazardous waste disposal and environmental compliance, which can fluctuate wildly based on regional regulations. Moreover, the ability to recover and reuse both the halogenated acylation solvent and the ethanol reduction solvent creates a more resilient supply chain that is less vulnerable to volatile raw material pricing. By reducing the dependency on bulk consumables like acetic acid and iron powder, manufacturers can achieve a leaner inventory profile and lower working capital requirements, ensuring consistent availability of the final intermediate even during market shortages.
- Cost Reduction in Manufacturing: The transition to a catalytic reduction system fundamentally alters the cost structure of production by removing the need for expensive waste treatment associated with iron mud. While specific percentage savings depend on local utility and disposal costs, the qualitative impact is profound; the process eliminates the filtration and washing steps required to separate product from iron sludge, thereby reducing labor hours and water consumption. Additionally, the high recovery rate of the halogenated solvent means that the effective consumption of this reagent per kilogram of product is drastically minimized, leading to substantial long-term operational expenditure reductions without compromising on reaction efficiency or product quality.
- Enhanced Supply Chain Reliability: The mild reaction conditions, particularly the lower temperature range of 70-80°C for acylation compared to the traditional 110°C, reduce the thermal stress on equipment and lower the risk of runaway reactions or safety incidents. This operational safety margin enhances the reliability of production schedules, minimizing unplanned downtime that can disrupt supply to downstream customers. Furthermore, the use of widely available reagents like hydrazine hydrate and ethanol, combined with a robust catalyst system, ensures that the manufacturing process is not bottlenecked by the sourcing of exotic or highly regulated specialty chemicals, thereby securing a continuous flow of materials for commercial scale-up.
- Scalability and Environmental Compliance: From a scalability perspective, the absence of solid waste generation in the reduction step simplifies the engineering requirements for large-scale reactors, as there is no need for complex sludge handling infrastructure. This makes the technology highly adaptable for expansion from pilot plant scales to multi-ton annual production capacities with minimal capital investment in waste management systems. The cleaner process profile also aligns perfectly with increasingly strict global environmental standards, future-proofing the manufacturing site against tighter emissions regulations and ensuring that the supply of this critical intermediate remains uninterrupted by environmental enforcement actions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the patent specifications and experimental data. These insights are designed to clarify the operational benefits and technical feasibility for potential partners looking to integrate this high-efficiency route into their supply chains. Understanding these details is crucial for evaluating the compatibility of this process with existing manufacturing infrastructure and quality assurance protocols.
Q: How does the new catalytic reduction method improve upon traditional iron powder reduction?
A: The novel method replaces iron powder reduction with hydrazine hydrate catalyzed by ferric chloride and activated carbon. This eliminates the generation of massive amounts of iron sludge waste, significantly simplifying wastewater treatment and improving overall yield from roughly 59% to over 77%.
Q: What are the purity specifications achievable with this process?
A: Through optimized acylation in halogenated solvents and precise catalytic reduction, the final product achieves HPLC purity greater than 99%, with intermediate purities consistently exceeding 97% throughout the synthetic route.
Q: Is the solvent system in the acylation step recyclable?
A: Yes, the process utilizes halogenated hydrocarbons like 1,2-dichloroethane which can be recovered via azeotropic distillation with water. The recovery rate is approximately 90-92%, allowing for mechanical reuse and substantial reduction in solvent procurement costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Dichloro-p-phenylenediamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global fine chemical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1974540B are fully realized in practical, industrial settings. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2,5-dichloro-p-phenylenediamine meets the highest standards of quality, free from the impurities often associated with older reduction technologies.
We invite you to collaborate with us to leverage these technological advancements for your specific application needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and product performance.
