Advanced LED-Driven Synthesis of N-Substituted Benzenesulfonamides for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways for synthesizing critical bioactive scaffolds. A significant breakthrough in this domain is documented in Chinese Patent CN114057616B, which discloses a novel method for synthesizing N-substituted benzenesulfonamide compounds. This technology represents a paradigm shift from traditional thermal and metal-catalyzed processes to a greener, photochemical approach. By leveraging visible light irradiation, specifically using LED sources, this method enables the coupling of commercially available nitro compounds and sodium arylsulfinates under remarkably mild conditions. For R&D directors and procurement strategists, this innovation offers a compelling value proposition: it simplifies the synthetic route, reduces reliance on scarce precious metals, and operates at room temperature, thereby addressing key pain points in both process development and supply chain management for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-substituted benzenesulfonamides has relied heavily on methodologies that present significant operational and economic challenges. Traditional routes often involve the condensation of benzenesulfonyl halides with amines, which generates stoichiometric amounts of corrosive waste and requires strict moisture control. More modern approaches have attempted to utilize transition metal catalysts, such as palladium or copper systems, to facilitate C-N bond formation. However, these metal-catalyzed protocols frequently necessitate high reaction temperatures, sometimes exceeding 100°C, and require inert atmospheres like argon to prevent catalyst deactivation. Furthermore, the use of explosive sulfonyl azides or unstable intermediates poses serious safety risks in a manufacturing environment. The removal of residual heavy metals from the final API intermediate adds complex purification steps, increasing both production time and overall cost, making these conventional methods less ideal for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
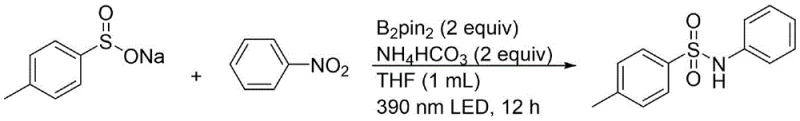
In stark contrast, the methodology outlined in patent CN114057616B introduces a streamlined, metal-free protocol that operates under ambient conditions. This novel approach utilizes bis(pinacolato)diboron (B2pin2) as a benign reducing agent in conjunction with a simple inorganic base, eliminating the need for expensive transition metal catalysts entirely. The reaction is driven by 390 nm LED light, allowing the transformation to proceed efficiently at room temperature without external heating. This not only drastically reduces energy consumption but also enhances safety profiles by avoiding high-pressure or high-temperature reactors. The scope of this reaction is broad, accommodating various substituted nitrobenzenes and arylsulfinates, which ensures versatility for diverse drug discovery programs. By simplifying the reaction setup to a basic irradiation vessel, this method significantly lowers the barrier to entry for producing high-purity N-substituted benzenesulfonamides.

Mechanistic Insights into LED-Driven Reductive Coupling
The core of this technological advancement lies in its unique mechanistic pathway, which bypasses the need for traditional oxidative addition and reductive elimination cycles associated with transition metals. Under LED illumination, the bis(pinacolato)diboron acts as a potent single-electron donor or radical precursor, facilitating the reduction of the nitro group on the aromatic ring. This photo-induced reduction converts the nitro compound into a reactive amine species in situ, which subsequently couples with the sulfur center of the sodium arylsulfinate. The absence of metal catalysts means there is no risk of metal leaching into the product stream, a critical factor for meeting stringent purity specifications required by regulatory bodies. The reaction mechanism is robust enough to tolerate a wide range of functional groups, including halogens and electron-withdrawing groups, without compromising yield or selectivity.
From an impurity control perspective, the mildness of the room-temperature conditions plays a pivotal role. High-temperature reactions often promote side reactions such as polymerization, over-reduction, or decomposition of sensitive functional groups. By maintaining the reaction at ambient temperature, the formation of these thermal degradation byproducts is minimized. Additionally, the use of stable starting materials like sodium arylsulfinates avoids the generation of hazardous gaseous byproducts common in sulfonyl chloride chemistry. The result is a cleaner reaction profile that simplifies downstream processing. For quality assurance teams, this translates to a more consistent impurity profile and easier validation of the cleaning procedures, ensuring that the final product meets the rigorous standards expected of a reliable pharmaceutical intermediates supplier.
How to Synthesize N-Substituted Benzenesulfonamide Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting is straightforward and requires minimal specialized equipment beyond a standard LED light source. The process begins by charging a reaction vessel with the sodium arylsulfinate, the chosen nitro compound, the boron-based reducing agent, and a mild base such as ammonium bicarbonate in a solvent like tetrahydrofuran. The mixture is then subjected to LED irradiation for a defined period, typically between 6 to 15 hours, depending on the specific substrate electronics. This simplicity allows for rapid optimization and scaling. For detailed operational parameters and specific molar ratios tailored to your target molecule, please refer to the standardized synthesis guide below.
- Combine sodium arylsulfinate, nitro compound, bis(pinacolato)diboron (B2pin2), and ammonium bicarbonate in an organic solvent such as THF.
- Irradiate the reaction mixture with a 390 nm LED light source at room temperature for 6 to 15 hours under an air atmosphere.
- Perform post-treatment via column chromatography or preparative TLC to isolate the high-purity N-substituted benzenesulfonamide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photochemical method offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the substantial cost savings achieved through raw material selection and process simplification. By eliminating the need for precious metal catalysts like palladium or copper, companies can avoid the volatility associated with metal prices and the costly logistics of metal recovery and disposal. Furthermore, the use of commodity chemicals such as nitro compounds and diboron reagents ensures a stable and diversified supply base, reducing the risk of single-source dependency. The ability to run reactions at room temperature also implies lower utility costs, as there is no need for extensive heating or cooling infrastructure, contributing to a leaner manufacturing overhead.
- Cost Reduction in Manufacturing: The economic impact of removing transition metals from the synthesis cannot be overstated. Traditional methods often require expensive ligands and catalysts that account for a significant portion of the bill of materials. By switching to a metal-free system, the direct material costs are drastically simplified. Additionally, the downstream purification process is less burdensome; without heavy metals to remove, the need for specialized scavenging resins or complex extraction protocols is eliminated. This streamlining of the workflow reduces labor hours and consumable usage, leading to a more competitive cost structure for the final API intermediate without compromising on quality or yield.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available, non-hazardous starting materials. Sodium arylsulfinates and nitroarenes are commodity chemicals produced by numerous global suppliers, ensuring that procurement teams can secure multiple sources to mitigate disruption risks. The stability of these reagents also allows for longer shelf lives and easier storage compared to sensitive organometallic reagents or unstable sulfonyl azides. This reliability ensures continuous production schedules and reduces the lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demands and inventory fluctuations.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces thermal management challenges, but this LED-driven method inherently mitigates those risks. Since the reaction occurs at room temperature, heat dissipation is not a limiting factor, making the transition from gram-scale to kilogram-scale production much smoother. From an environmental standpoint, the process aligns with green chemistry principles by reducing energy consumption and avoiding toxic metal waste. This compliance with increasingly strict environmental regulations facilitates easier permitting and reduces the carbon footprint of the manufacturing site, enhancing the company's sustainability profile and long-term operational license.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this LED-mediated synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN114057616B operates without transition metal catalysts, utilizing inexpensive bis(pinacolato)diboron as a reducing agent instead, which significantly lowers raw material costs and eliminates heavy metal residue concerns.
Q: What are the reaction conditions regarding temperature and pressure?
A: The reaction proceeds efficiently at room temperature under standard atmospheric pressure (air atmosphere) using LED illumination, removing the need for energy-intensive heating or specialized high-pressure equipment.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent demonstrates successful scaling to gram-level reactions with high yields, and the mild conditions combined with commercially available reagents suggest strong potential for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Substituted Benzenesulfonamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green synthetic methodologies like the one described in CN114057616B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale discoveries are successfully translated into robust industrial processes. Our facilities are equipped with state-of-the-art photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications for complex pharmaceutical intermediates. We are committed to delivering high-quality products that adhere to the highest international standards, supporting your drug development pipeline with reliability and precision.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis to demonstrate how switching to this metal-free route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your target molecules, and let us help you accelerate your path to market with superior chemical solutions.
