Advanced Titanium-Mediated Synthesis of 1,1,4,4-Tetraalkoxy-2-Butene for Industrial Scale-Up
The global demand for high-purity carotenoids, widely utilized in pharmaceuticals, nutraceuticals, and food additives, continues to drive innovation in intermediate synthesis. Patent CN114315532A introduces a groundbreaking method for preparing 1,1,4,4-tetraalkoxy-2-butene, a critical precursor in the synthesis of dodecenal and subsequent carotenoid molecules. This technology addresses long-standing inefficiencies in the supply chain by shifting away from expensive furan-based starting materials toward more economical acetaldehyde derivatives. By leveraging low-valent titanium chemistry, this process achieves exceptional yields and operational simplicity, positioning it as a transformative solution for reliable pharmaceutical intermediates supplier networks seeking to optimize their production capabilities.
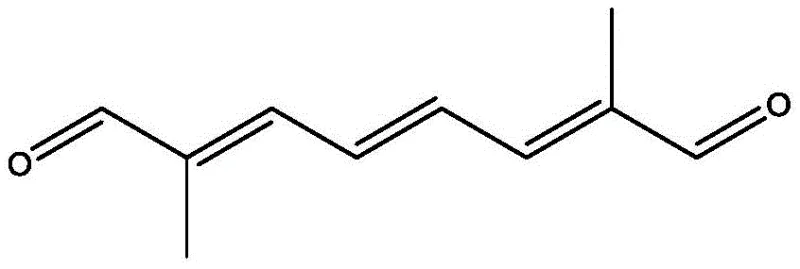
Carotenoids such as beta-carotene and astaxanthin rely on precise C40 symmetric structures, where the integrity of the carbon backbone is paramount. The intermediate dodecenal, depicted in the structural analysis above, serves as a pivotal building block in these complex syntheses. Traditionally, the production of the necessary butene precursors has been bottlenecked by low selectivity and harsh reaction conditions. The introduction of this novel titanium-mediated pathway offers a robust alternative that aligns with the rigorous purity standards required by R&D directors in the fine chemical sector, ensuring that downstream synthesis steps proceed with minimal impurity burden.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 1,1,4,4-tetraalkoxy-2-butene has relied heavily on the acid-catalyzed reaction of 2,5-dimethoxy-2,5-dihydrofuran with alcohols. While established, this legacy approach suffers from significant drawbacks that hinder cost reduction in pharmaceutical intermediates manufacturing. The reliance on strong inorganic or organic acids creates severe corrosion challenges for reactor equipment, necessitating expensive specialized materials and increasing maintenance downtime. Furthermore, the market price of the key starting material, 2,5-dimethoxy-2,5-dihydrofuran, is prohibitively high, often ranging between 140,000 to 160,000 yuan per ton. Combined with moderate yields typically hovering around 78% to 87%, the overall economic efficiency of these conventional routes is suboptimal for large-scale operations.
The Novel Approach
In stark contrast, the methodology disclosed in CN114315532A utilizes 2,2-dialkoxyacetaldehyde as the primary substrate, a raw material that is significantly more accessible and cost-effective. By employing a reductive coupling mechanism mediated by high-valence titanium compounds and common reducing agents, this new route bypasses the need for corrosive liquid acids entirely. The process operates under milder conditions initially, with temperatures controlled between -40°C and 10°C during the activation phase, before proceeding to reflux. This shift not only enhances operator safety and equipment longevity but also dramatically improves reaction selectivity. The result is a streamlined workflow that delivers superior yields, often exceeding 90%, thereby providing a compelling value proposition for procurement managers focused on margin improvement.
Mechanistic Insights into Titanium-Mediated Reductive Coupling
The core of this technological advancement lies in the generation of low-valent titanium species in situ, which facilitate the homocoupling of the aldehyde functionality. As illustrated in the reaction scheme below, the process begins with the reduction of a Ti(III) or Ti(IV) source, such as TiCl3 or TiCl4, by a metal reductant like Zinc or Magnesium. This generates a highly reactive titanium surface or cluster capable of coordinating with the carbonyl oxygen of the 2,2-dialkoxyacetaldehyde. The subsequent electron transfer promotes the formation of a carbon-carbon double bond between two aldehyde units, effectively dimerizing the substrate into the desired 1,1,4,4-tetraalkoxy-2-butene skeleton. This mechanism is akin to McMurry coupling but optimized for acetal-protected substrates, ensuring that the sensitive alkoxy groups remain intact throughout the transformation.
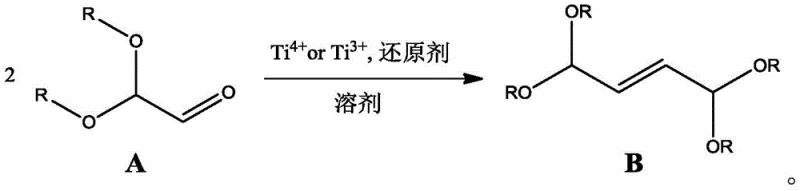
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed dehydration routes. Traditional acid methods often lead to polymerization side reactions and the formation of complex ether byproducts due to the instability of the oxocarbenium intermediates. In the titanium-mediated system, the reaction environment is fundamentally different; the absence of strong protons minimizes acid-catalyzed degradation pathways. Furthermore, the use of specific solvents like tetrahydrofuran or diethyl ether helps stabilize the organometallic intermediates, ensuring a cleaner reaction profile. For quality control teams, this translates to a simpler purification process, often requiring only a basic aqueous quench and phase separation to achieve high-purity product, thus reducing the load on downstream distillation columns.
How to Synthesize 1,1,4,4-Tetraalkoxy-2-Butene Efficiently
Implementing this synthesis requires careful attention to the order of addition and temperature control to maximize the activity of the low-valent titanium species. The protocol dictates that the reducing agent must be fully dispersed in the solvent before the introduction of the titanium source to ensure uniform activation. Once the titanium complex is formed, the addition of the 2,2-dialkoxyacetaldehyde should be managed to prevent localized exotherms that could degrade the sensitive intermediates. The detailed standardized synthesis steps, including specific molar ratios and quenching procedures validated by the patent examples, are outlined in the guide below for technical reference.
- Under nitrogen protection, dissolve a reducing agent (such as Zn, Mg, or LiAlH4) in an organic solvent like THF or diethyl ether and cool the mixture to between -40°C and 10°C.
- Add a high-valence titanium auxiliary agent (Ti3+ or Ti4+ compound like TiCl3 or TiBr4) to the reaction vessel under stirring conditions.
- Introduce the 2,2-dialkoxyacetaldehyde substrate, raise the temperature to reflux for 10-15 hours, and finally quench with an alkaline aqueous solution to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders, the transition to this novel synthetic route represents a strategic opportunity to de-risk the procurement of critical carotenoid intermediates. By decoupling production from the volatile pricing of furan-based starting materials, manufacturers can achieve greater stability in their cost structures. The elimination of corrosive acids also reduces the total cost of ownership for production facilities, as standard stainless steel reactors can often be utilized instead of glass-lined or Hastelloy vessels. This flexibility allows for faster turnaround times between batches and reduces the dependency on specialized maintenance crews, directly contributing to enhanced supply chain reliability and continuity.
- Cost Reduction in Manufacturing: The most immediate impact is seen in raw material expenditures. Since 2,2-dialkoxyacetaldehyde is significantly cheaper than 2,5-dimethoxy-2,5-dihydrofuran, the direct material cost per kilogram of product is drastically lowered. Additionally, the high single-pass isolation yields reported, reaching up to 97% in optimized examples, mean that less raw material is wasted in purification loops. The removal of expensive dehydrating agents like trimethyl orthoformate further strips away unnecessary cost layers, resulting in substantial overall savings without compromising product quality.
- Enhanced Supply Chain Reliability: The versatility of the reagent list enhances supply security. The process tolerates a wide range of reducing agents, including Zinc, Magnesium, and various hydrides, allowing procurement teams to source based on availability and spot pricing without halting production. Similarly, the titanium sources can be varied between chlorides, bromides, or iodides. This flexibility ensures that a shortage of one specific reagent does not become a bottleneck, thereby reducing lead time for high-purity intermediates and maintaining consistent output even during market fluctuations.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process is inherently greener. The absence of strong mineral acids eliminates the generation of large volumes of acidic wastewater, simplifying effluent treatment and reducing compliance costs. The solvents used, such as toluene and THF, are standard industrial chemicals with well-established recovery and recycling protocols. This makes the commercial scale-up of complex intermediates much more straightforward, as the process aligns easily with existing waste management infrastructure and sustainability goals mandated by global regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this titanium-mediated synthesis. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the technology's fit within your current manufacturing portfolio. Understanding these nuances is essential for making informed decisions about process adoption and vendor qualification.
Q: What are the primary advantages of this titanium-mediated method over traditional acid-catalyzed routes?
A: The novel titanium-mediated reductive coupling eliminates the need for corrosive strong acids and expensive starting materials like 2,5-dimethoxy-2,5-dihydrofuran. It utilizes cheaper 2,2-dialkoxyacetaldehyde and achieves significantly higher yields, often exceeding 90%, while simplifying post-reaction processing.
Q: Which reducing agents and titanium sources are compatible with this synthesis?
A: The process demonstrates broad compatibility with various reducing agents including Zinc (Zn), Magnesium (Mg), Lithium Aluminum Hydride (LiAlH4), and Sodium Borohydride (NaBH4). Compatible titanium sources include TiCl3, TiCl4, TiBr3, TiBr4, and TiI4, allowing for flexibility in sourcing and cost optimization.
Q: Is this process suitable for large-scale industrial production of carotenoid intermediates?
A: Yes, the method is designed for scalability. It avoids the equipment corrosion issues associated with liquid strong acids and uses standard organic solvents like THF and toluene. The high single-pass isolation yields reported in the patent data indicate strong potential for cost-effective commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1,4,4-Tetraalkoxy-2-Butene Supplier
As the industry shifts towards more efficient and sustainable synthetic routes, NINGBO INNO PHARMCHEM stands ready to support your transition with our advanced manufacturing capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel titanium-mediated chemistry are realized at the volume you require. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of verifying the low impurity profiles characteristic of this new method, guaranteeing that every batch meets the exacting standards of the global pharmaceutical and nutraceutical markets.
We invite you to engage with our technical team to explore how this process can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits specific to your operation. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and commercial viability.
