Revolutionizing Thiadiazole Production: A Solvent-Free Grinding Protocol for High-Purity Pharmaceutical Intermediates
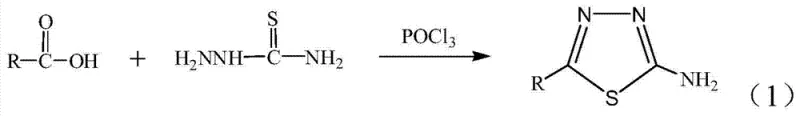
The landscape of heterocyclic chemistry is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic methodologies. Patent CN103880776A introduces a groundbreaking solid-phase grinding technique for the preparation of 2-amino-5-alkyl-1,3,4-thiadiazoles, a class of compounds renowned for their profound biological activities ranging from anticancer to antifungal properties. This innovation departs radically from conventional liquid-phase protocols by utilizing silica gel as a solid support and phosphorus oxychloride (POCl3) as a catalyst under mechanochemical conditions. For R&D directors and procurement specialists in the fine chemical sector, this represents a pivotal shift towards sustainable manufacturing that eliminates the need for volatile organic solvents during the reaction phase. The method achieves exceptional yields exceeding 91% while operating at ambient temperatures, thereby addressing critical pain points regarding energy consumption and operational safety in the production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for 1,3,4-thiadiazole derivatives typically rely on liquid-phase reactions that necessitate prolonged heating under reflux conditions, often involving hazardous solvents and complex workup procedures. These legacy methods are plagued by inherent inefficiencies, including extended reaction times that can span several hours to days, which severely bottlenecks throughput in a commercial setting. Furthermore, the reliance on bulk solvents creates substantial downstream burdens related to solvent recovery, waste disposal, and environmental compliance, all of which inflate the overall cost of goods sold. From a supply chain perspective, the requirement for specialized reflux equipment and rigorous temperature control systems increases capital expenditure and maintenance overheads. Additionally, liquid-phase cyclizations often suffer from side reactions and incomplete conversions, resulting in lower isolated yields and a more challenging purification process to meet the stringent purity specifications required for active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the methodology disclosed in the patent leverages the principles of mechanochemistry to drive the cyclization reaction through physical grinding rather than thermal energy. By employing silica gel as an inert yet highly effective carrier, the reactants—carboxylic acids and thiosemicarbazide—are brought into intimate contact on a molecular level within the pores of the solid support. This solid-state environment not only accelerates the reaction kinetics, reducing the active processing time to merely 5 to 10 minutes of grinding, but also simplifies the isolation procedure dramatically. The absence of bulk solvents during the reaction phase means that the workup involves a straightforward neutralization and filtration step to remove the silica carrier, bypassing the need for energy-intensive distillation columns. This streamlined approach not only enhances the economic viability of producing reliable pharmaceutical intermediate supplies but also aligns perfectly with modern green chemistry mandates by minimizing waste generation at the source.
Mechanistic Insights into POCl3-Catalyzed Solid-Phase Cyclization
The core of this synthetic breakthrough lies in the synergistic interaction between phosphorus oxychloride and the silica gel surface. Mechanistically, POCl3 acts as a potent dehydrating agent and electrophilic activator, converting the carboxylic acid moiety into a highly reactive acyl chloride or mixed anhydride intermediate in situ. In the absence of solvent, the silica gel serves a dual function: it acts as a physical matrix to disperse the reagents evenly, preventing localized hot spots that could lead to decomposition, and its surface silanol groups may assist in stabilizing transition states through weak hydrogen bonding interactions. The thiosemicarbazide then undergoes a nucleophilic attack on the activated carbonyl carbon, followed by an intramolecular cyclization and dehydration sequence to form the stable 1,3,4-thiadiazole ring. This pathway is exceptionally clean, as the solid support effectively traps byproducts and prevents polymerization, ensuring that the final crude product is of high quality prior to even the simplest purification steps.
Furthermore, the impurity profile generated by this solid-phase method is significantly superior to that of liquid-phase counterparts. In traditional solvent-based systems, competing hydrolysis of the acyl chloride intermediate or oligomerization of the thiosemicarbazide can occur, leading to difficult-to-remove impurities that compromise the biological efficacy of the final drug substance. The controlled microenvironment provided by the silica gel pores restricts the mobility of reactive species, effectively channeling the reaction towards the desired cyclization pathway. This selectivity is crucial for regulatory compliance, as it reduces the burden on analytical teams to identify and quantify trace genotoxic impurities. For manufacturers aiming for cost reduction in agrochemical intermediate manufacturing, this inherent purity translates directly into reduced chromatography loads and higher overall process mass intensity, making the technology scalable from gram-scale discovery to multi-ton commercial production without losing fidelity.
How to Synthesize 2-Amino-5-Alkyl-1,3,4-Thiadiazole Efficiently
The operational simplicity of this protocol makes it accessible for both laboratory-scale optimization and industrial implementation. The process begins with the precise stoichiometric mixing of thiosemicarbazide, the chosen carboxylic acid substrate, and phosphorus oxychloride onto a bed of dry silica gel. The reaction progress is conveniently monitored via thin-layer chromatography (TLC), where the disappearance of the starting thiosemicarbazide spot signals complete conversion, typically achieved within a short 5 to 10-minute grinding window. Following a brief standing period to allow the cyclization to finalize, the crude solid is treated with a mild alkaline solution to neutralize acidic byproducts, followed by filtration to separate the valuable organic product from the inorganic silica support. The detailed standardized synthesis steps, including specific molar ratios and workup parameters for various substrates, are outlined below to ensure reproducibility and safety.
- Mix thiosemicarbazide, carboxylic acid, phosphorus oxychloride, and silica gel in a dry vessel and grind at room temperature until TLC indicates completion.
- Allow the mixture to stand for 30 to 60 minutes to ensure full conversion of the intermediate species.
- Neutralize the crude product with sodium carbonate solution, filter to remove silica, extract with DMF, and concentrate to isolate the pure thiadiazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solid-phase technology offers transformative benefits that extend far beyond mere chemical yield. The elimination of bulk solvents during the reaction phase drastically reduces the volume of hazardous waste requiring treatment, leading to substantial cost savings in environmental compliance and waste disposal fees. Moreover, the ability to operate at room temperature removes the dependency on steam or electrical heating infrastructure, significantly lowering utility costs and enhancing the safety profile of the manufacturing facility by mitigating thermal runaway risks. This robustness ensures enhanced supply chain reliability, as the process is less susceptible to fluctuations in energy supply or cooling water availability, guaranteeing consistent delivery schedules for critical intermediates.
- Cost Reduction in Manufacturing: The economic impact of switching to this solvent-free grinding method is profound, primarily driven by the removal of expensive solvent purchase and recovery cycles. By utilizing silica gel, a low-cost and reusable material, as the reaction medium, the process eliminates the need for large-scale distillation units, thereby reducing both capital investment and operational energy consumption. The high conversion rates observed, often exceeding 90%, minimize raw material wastage, ensuring that every kilogram of input generates maximum output value. Additionally, the simplified workup procedure reduces labor hours and solvent usage in the purification stage, creating a leaner, more cost-effective production model that improves margin potential for high-volume contracts.
- Enhanced Supply Chain Reliability: From a logistics perspective, the raw materials required for this synthesis—carboxylic acids, thiosemicarbazide, and POCl3—are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages. The simplicity of the equipment requirements, essentially needing only grinding machinery and filtration units, allows for flexible manufacturing across multiple sites, thereby diversifying supply risk. The short reaction cycle time of under one hour per batch significantly increases plant throughput capacity, enabling manufacturers to respond rapidly to sudden spikes in demand from downstream pharmaceutical clients without the need for extensive inventory buffering.
- Scalability and Environmental Compliance: Scaling mechanochemical processes is increasingly feasible with modern industrial milling technologies, allowing for a seamless transition from pilot plant to full commercial production. The solid-state nature of the reaction inherently contains emissions, as there are no solvent vapors to capture, simplifying the design of ventilation and scrubbing systems. This aligns with increasingly stringent global environmental regulations, positioning manufacturers who adopt this technology as leaders in sustainable chemistry. The reduced ecological footprint not only satisfies regulatory bodies but also appeals to end-clients who are prioritizing green suppliers in their vendor qualification audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solid-phase synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on substrate scope, safety, and scalability. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this method into existing production lines or for sourcing teams negotiating supply agreements for thiadiazole derivatives.
Q: What are the primary advantages of using silica gel as a carrier in this synthesis?
A: Silica gel provides a large specific surface area and thermal stability, which facilitates heat dissipation during the exothermic grinding process and prevents coking, leading to higher purity and yields exceeding 91%.
Q: Can this solid-phase method be applied to aromatic carboxylic acids?
A: Yes, the protocol is highly versatile and successfully accommodates various substituted phenoxyacetic acids, including chloro-, fluoro-, bromo-, and nitro- derivatives, maintaining high efficiency.
Q: How does this method compare to traditional liquid-phase synthesis in terms of equipment requirements?
A: Unlike traditional methods requiring reflux setups and extensive solvent recovery systems, this mechanochemical approach operates at room temperature with minimal equipment, significantly lowering capital expenditure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-5-Alkyl-1,3,4-Thiadiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting advanced synthetic methodologies like the solid-phase grinding protocol to deliver superior value to our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity demonstrated in the lab are faithfully reproduced at an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and quality of every batch of 2-amino-5-alkyl-1,3,4-thiadiazole we produce. Our commitment to excellence ensures that the intermediates supplied meet the exacting standards required for the synthesis of complex APIs and agrochemical actives.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage potential partners to contact us directly to obtain specific COA data for our current inventory and to discuss route feasibility assessments for custom derivatives. Let us help you optimize your supply chain with high-quality, cost-effective thiadiazole intermediates produced through sustainable and efficient manufacturing practices.
