Advanced Synthesis of Alpha-Pyrone Derivatives: Technical Upgrade and Commercial Scalability
The chemical landscape for heterocyclic compound synthesis is undergoing a significant transformation, driven by the urgent need for more sustainable and cost-effective manufacturing processes. Patent CN101085769B, published in 2010, introduces a groundbreaking methodology for the synthesis of α-pyrone derivatives, a class of compounds with profound biological activities ranging from anti-HIV to antifungal properties. This technical disclosure outlines a novel cyclization reaction that utilizes Baylis-Hillman adducts and 1,3-cyclohexanedione derivatives under base catalysis. For R&D directors and procurement managers in the pharmaceutical and agrochemical sectors, this patent represents a pivotal shift away from traditional, resource-intensive methods. The core innovation lies in its ability to achieve high yields under mild conditions, often without the need for expensive transition metal catalysts. This report provides a deep technical analysis of this synthesis route, evaluating its feasibility for commercial scale-up and its potential to optimize supply chain reliability for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the development of the technology described in CN101085769B, the synthesis of α-pyrone derivatives was fraught with significant technical and economic challenges that hindered efficient commercial production. Traditional methods often relied heavily on palladium-catalyzed coupling reactions, such as the coupling of β-iodoacrylic acid with allenyl tin compounds, or complex multi-step sequences involving strong bases and trifluoroacetic anhydride. These conventional routes are not only chemically cumbersome but also economically burdensome due to the high cost of noble metal catalysts and the stringent requirements for their removal from the final product. Furthermore, many existing processes suffer from low atom economy, generating substantial amounts of chemical waste that complicate environmental compliance and increase disposal costs. The reliance on harsh reaction conditions and difficult-to-source raw materials further exacerbates supply chain vulnerabilities, making it difficult for manufacturers to guarantee consistent delivery of high-purity intermediates to downstream pharmaceutical clients.
The Novel Approach
In stark contrast to the limitations of prior art, the novel approach detailed in this patent offers a streamlined, one-step cyclization process that dramatically simplifies the manufacturing workflow. By utilizing readily available Baylis-Hillman adducts and 1,3-cyclohexanedione derivatives, the reaction proceeds efficiently in the presence of inexpensive inorganic bases or organic amines. The versatility of this method is evident in its ability to operate effectively in various organic solvents such as acetone, ethanol, or toluene, and remarkably, it can even be conducted under solvent-free conditions. This flexibility allows manufacturers to tailor the process to specific facility capabilities while maximizing throughput. The reaction conditions are notably mild, typically ranging from 40°C to 80°C, which reduces energy consumption and enhances operational safety. This robust methodology not only improves the overall yield but also ensures a cleaner reaction profile, facilitating easier downstream purification and significantly reducing the burden on quality control laboratories.
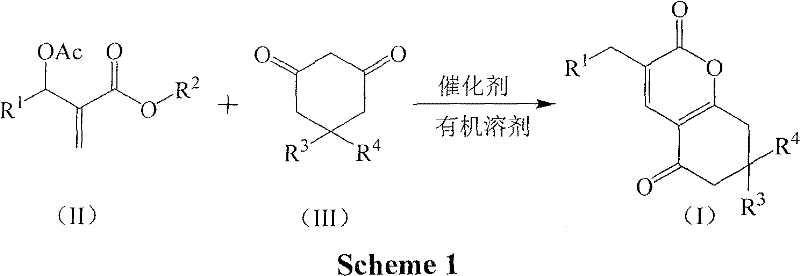
Mechanistic Insights into Base-Catalyzed Cyclization
The core of this technological advancement lies in the base-catalyzed cyclization mechanism, which orchestrates the formation of the α-pyrone ring with high precision and selectivity. The reaction initiates with the deprotonation of the active methylene group in the 1,3-cyclohexanedione derivative by the base catalyst, generating a nucleophilic enolate species. This enolate then attacks the electrophilic center of the Baylis-Hillman adduct, triggering a cascade of intramolecular rearrangements that ultimately lead to the closure of the pyrone ring. The elegance of this mechanism is its tolerance to a wide variety of functional groups on the aromatic rings, allowing for the synthesis of diverse derivatives with substituents such as nitro, fluoro, chloro, and methyl groups. This broad substrate scope is crucial for medicinal chemists who require structural diversity to optimize the biological activity of lead compounds. The use of mild bases like potassium carbonate or triethylamine ensures that sensitive functional groups remain intact, preserving the integrity of the molecular architecture throughout the synthesis.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. The absence of transition metals eliminates the risk of heavy metal contamination, a critical parameter for pharmaceutical intermediates intended for human consumption. The reaction pathway is highly selective, minimizing the formation of side products that often plague complex organic syntheses. Experimental data from the patent indicates that the crude products often possess high purity levels, which can be further enhanced to exceed 98% through simple recrystallization or column chromatography. This high level of purity reduces the need for extensive purification steps, thereby shortening the overall production cycle. For supply chain heads, this translates to a more predictable manufacturing timeline and a reduced risk of batch failures due to impurity profiles that fail to meet stringent regulatory specifications. The robustness of the mechanism ensures consistent quality across different batches, reinforcing supply chain continuity.
How to Synthesize Alpha-Pyrone Derivatives Efficiently
Implementing this synthesis route in a commercial setting requires a clear understanding of the operational parameters that drive efficiency and yield. The process begins with the precise weighing and mixing of the Baylis-Hillman adduct and the 1,3-cyclohexanedione derivative in a molar ratio that typically favors a slight excess of the dione to drive the reaction to completion. The choice of base catalyst is critical, with potassium carbonate and triethylamine emerging as preferred options due to their balance of reactivity and cost. The reaction mixture is then heated to a temperature between 40°C and 80°C, a range that provides sufficient thermal energy to overcome the activation barrier without risking thermal degradation of the product. Monitoring the reaction progress via TLC ensures that the conversion is complete before proceeding to workup. The detailed standardized synthesis steps for this specific pathway are outlined in the guide below, providing a clear roadmap for technical teams to replicate these results in a pilot or production plant environment.
- Prepare the reaction mixture by combining Baylis-Hillman adducts and 1,3-cyclohexanedione derivatives with a base catalyst such as potassium carbonate or triethylamine.
- Conduct the cyclization reaction in an organic solvent like acetone or ethanol, or under solvent-free conditions, maintaining a temperature between 40°C and 80°C.
- Upon completion, perform standard workup including brine wash, organic extraction, drying, and purification via recrystallization or column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis technology offers compelling economic and operational benefits that extend beyond simple chemical yield. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for expensive palladium catalysts and complex organometallic reagents, the direct material cost of the synthesis is significantly reduced. This cost reduction in fine chemical manufacturing is achieved not through marginal tweaks but through a fundamental redesign of the chemical pathway. Furthermore, the ability to run the reaction under solvent-free conditions or with common solvents like ethanol and acetone reduces the dependency on specialized, high-cost solvents. This flexibility allows procurement teams to leverage existing supply contracts for bulk commodities, insulating the production process from volatility in the specialty chemical market. The simplified post-treatment process also reduces the consumption of auxiliary materials such as drying agents and chromatography media, contributing to overall cost efficiency.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive metal scavenging steps, which are often required to meet regulatory limits for heavy metals in pharmaceutical ingredients. This simplification directly lowers the cost of goods sold by reducing both reagent expenses and processing time. Additionally, the high atom economy of the reaction means that a larger proportion of the starting materials are incorporated into the final product, minimizing waste generation. This efficiency translates into substantial cost savings over the lifecycle of the product, making it a highly attractive option for large-scale commercial production where margin optimization is critical.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, such as 1,3-cyclohexanedione and various Baylis-Hillman adducts, are commercially available from multiple global suppliers. This abundance reduces the risk of supply disruptions that can occur with proprietary or single-source reagents. The mild reaction conditions also mean that the process can be executed in a wider range of manufacturing facilities without requiring specialized high-pressure or cryogenic equipment. This flexibility enhances the resilience of the supply chain, allowing for faster qualification of alternative manufacturing sites if needed. Reducing lead time for high-purity pharmaceutical intermediates becomes feasible when the synthesis is robust and less dependent on fragile catalytic systems.
- Scalability and Environmental Compliance: The potential for solvent-free operation is a significant advantage for environmental compliance and scalability. Reducing solvent usage lowers the volume of hazardous waste that must be treated and disposed of, aligning with increasingly strict environmental regulations. The mild temperatures and atmospheric pressure conditions simplify the engineering requirements for scale-up, allowing for the use of standard glass-lined or stainless steel reactors. This ease of scale-up ensures that the transition from laboratory grams to commercial tons can be achieved with minimal technical risk. The commercial scale-up of complex pharmaceutical intermediates is often bottlenecked by safety and environmental concerns, but this methodology effectively mitigates those risks, ensuring a smoother path to market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The insights provided here aim to clarify the operational parameters and quality expectations associated with this novel method.
Q: What are the primary advantages of this synthesis method over traditional palladium-catalyzed routes?
A: This method eliminates the need for expensive transition metal catalysts like palladium, significantly reducing raw material costs and simplifying the removal of heavy metal impurities, which is critical for pharmaceutical compliance.
Q: Can this process be adapted for solvent-free manufacturing to enhance environmental compliance?
A: Yes, the patent data explicitly supports solvent-free conditions at elevated temperatures, which drastically reduces volatile organic compound (VOC) emissions and lowers waste treatment costs for large-scale production.
Q: What is the typical purity profile achievable with this cyclization technique?
A: Experimental data indicates that HPLC purity can consistently exceed 98% after standard recrystallization, meeting the stringent specifications required for high-value pharmaceutical intermediates and fine chemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Pyrone Derivatives Supplier
The technical potential of this base-catalyzed cyclization route is immense, offering a pathway to high-quality intermediates that meet the rigorous demands of the global pharmaceutical industry. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to fruition. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the high HPLC purity levels demonstrated in the patent data. We understand the critical nature of supply chain continuity and are committed to delivering consistent quality that supports your R&D and commercial manufacturing needs. Our technical team is ready to assess the specific requirements of your project and adapt this synthesis route to ensure optimal performance in a commercial setting.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your production volume. We encourage potential partners to contact us for specific COA data and route feasibility assessments to validate the performance of this technology against your current standards. Our goal is to provide a transparent and data-driven partnership that drives value for your organization. Let us help you navigate the complexities of fine chemical manufacturing with a solution that balances technical excellence with commercial viability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
