Advanced Lipase Mutants Enable Room Temperature Asymmetric Hydrolysis for Pharmaceutical Intermediates
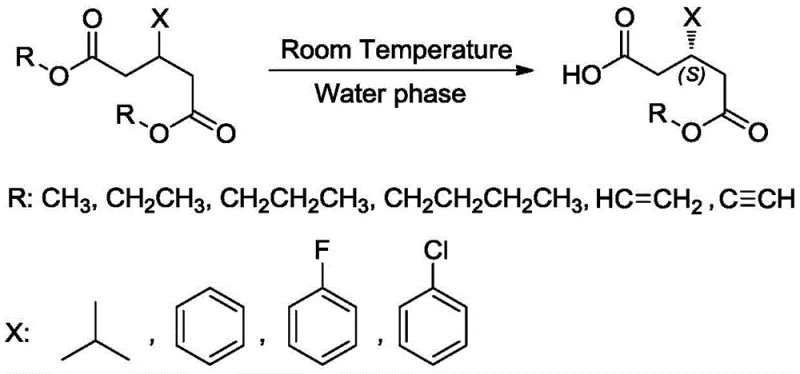
The pharmaceutical and fine chemical industries are constantly seeking robust biocatalytic solutions that can overcome the limitations of traditional chemical synthesis, particularly in the realm of chiral intermediate production. A significant breakthrough in this domain is documented in Chinese Patent CN113564144A, which details the development of novel lipase mutants derived from Candida antarctica (PAL) and Sporisorium reilianum (SRL). This intellectual property represents a paradigm shift in asymmetric hydrolysis, moving away from energy-intensive, solvent-heavy processes toward environmentally benign, high-efficiency biocatalysis. The core innovation lies in the semi-rational design of enzyme active sites, which allows for the precise asymmetric hydrolysis of prochiral diester substrates into optically pure S-monoester products. Unlike conventional methods that struggle with selectivity outside of narrow parameters, these engineered mutants demonstrate exceptional performance under mild conditions, specifically at room temperature ranging from 10-40°C and, crucially, within a pure water phase system. This technological advancement addresses critical pain points for R&D directors and process chemists who are tasked with improving the purity and sustainability of synthetic routes for key drug intermediates such as those used in the synthesis of Paroxetine and Duloxetine.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the enzymatic preparation of chiral intermediates via asymmetric hydrolysis has been plagued by significant operational constraints that hinder industrial viability. Traditional wild-type lipases often exhibit unstable stereoselectivity that is heavily dependent on rigorous reaction control, typically requiring low-temperature environments (often below 0°C) to maintain acceptable enantiomeric excess. Furthermore, these legacy processes frequently necessitate the addition of organic co-solvents such as dioxane, t-amyl alcohol, or acetonitrile to solubilize substrates or modulate enzyme activity. The reliance on these volatile organic compounds introduces substantial downstream processing challenges, including complex separation steps, increased waste treatment costs, and potential safety hazards associated with solvent handling. Additionally, when chiral resolution is employed instead of asymmetric hydrolysis, the theoretical yield is inherently capped at 50%, leading to significant material waste and the need for racemization recovery steps that further complicate the manufacturing workflow. Literature precedents, such as those cited in the patent background, indicate that even optimized wild-type systems often struggle to exceed 90% e.e. without these harsh conditions, limiting their utility for high-purity pharmaceutical applications.
The Novel Approach
The technology disclosed in CN113564144A fundamentally disrupts these limitations by introducing a suite of lipase mutants capable of operating with high fidelity under ambient conditions. By utilizing a semi-rational design strategy, researchers have successfully modified the enzyme's active pocket to enhance its intrinsic stereoselectivity, thereby eliminating the need for cryogenic cooling and organic co-solvents. This novel approach enables the direct asymmetric hydrolysis of various diester substrates in a pure water phase, achieving conversion rates greater than 99% and enantiomeric excess values exceeding 95%. The ability to conduct these reactions at room temperature (10-40°C) not only reduces energy consumption but also simplifies reactor requirements, making the process far more amenable to scale-up. This method effectively bypasses the 50% yield ceiling of resolution processes, offering a theoretical yield of 100% for the desired S-configuration product. For procurement and supply chain managers, this translates to a drastic reduction in raw material waste and a streamlined production cycle that avoids the complexities of solvent recovery and low-temperature maintenance.
Mechanistic Insights into Semi-Rational Lipase Engineering
The success of this biocatalytic platform rests on a deep understanding of protein structure-function relationships, specifically targeting the acyl and alcohol binding pockets of the lipase enzyme. Through three-dimensional structural analysis using visualization software, the inventors identified specific amino acid residues that govern substrate binding conformation and catalytic triad accessibility. For the Candida antarctica lipase (PAL), key residues including T138, L140, V154, and I189 in the acyl pocket, along with L278, A281, and A282 in the alcohol group binding pocket, were pinpointed as critical determinants of enantioselectivity. By systematically mutating these sites—such as substituting Leucine at position 140 with Valine or Alanine at position 281 with Valine—the steric environment of the active site is altered to preferentially accommodate the pro-S transition state. This precise engineering prevents the binding or hydrolysis of the unwanted R-enantiomer, thereby driving the reaction towards high optical purity without external chemical modifiers. The same logic was applied to the Sporisorium reilianum lipase (SRL), mapping corresponding positions 145, 159, 287, and 288 to achieve similar enhancements in selectivity.
Furthermore, the mechanism involves a synergistic effect where combination mutants (e.g., L140V/A281V) outperform single-point mutations, suggesting a cooperative interaction between the acyl and alcohol pockets that stabilizes the substrate in the optimal orientation for hydrolysis. This structural rigidity in the mutant enzyme allows it to maintain high selectivity even in the absence of co-solvents, which traditionally act to rigidify the enzyme structure in wild-type variants. The result is a biocatalyst that is not only highly selective but also robust against variations in substrate structure, accommodating diverse R groups ranging from simple alkyl chains to aromatic rings with halogen substituents. This mechanistic robustness ensures consistent impurity profiles, a critical factor for R&D directors managing regulatory filings, as it minimizes the formation of hard-to-remove stereoisomeric impurities that could compromise the safety profile of the final active pharmaceutical ingredient.
How to Synthesize S-Monoesters Efficiently
The implementation of this technology into a manufacturing setting follows a straightforward biocatalytic workflow that leverages the high stability of the engineered mutants. The process begins with the cultivation of recombinant E. coli strains harboring the specific lipase mutant genes, followed by the harvesting of wet cell mass which serves as the biocatalyst. The reaction is initiated by suspending these cells in a standard buffer solution, such as citric acid-sodium citrate or phosphate buffer, adjusted to a pH between 4.0 and 9.0. The prochiral diester substrate is then introduced directly into this aqueous suspension, where the mutant lipase catalyzes the hydrolysis of one ester group with high precision. Detailed standardized synthesis steps for specific substrates like (S)-3-isobutyl-5-methoxy-5-oxopentanoic acid are outlined below.
- Prepare recombinant E. coli cells expressing specific lipase mutants (e.g., PAL L140V/A281V) and harvest wet cell mass via centrifugation.
- Suspend the wet cells in a buffer solution (pH 4.0-9.0) and add the prochiral diester substrate directly without organic co-solvents.
- Maintain the reaction mixture at room temperature (10-40°C) with shaking until conversion exceeds 99%, then extract the product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and operational continuity, the transition to this mutant lipase technology offers profound economic and logistical benefits. The elimination of organic co-solvents and low-temperature requirements fundamentally alters the cost structure of chiral intermediate manufacturing, removing significant overheads associated with solvent purchase, recovery, and disposal. Moreover, the high conversion efficiency ensures that raw materials are utilized to their maximum theoretical potential, reducing the cost per kilogram of the final product. From a supply chain perspective, the robustness of the water-phase system enhances reliability by reducing the risk of batch failures due to temperature fluctuations or solvent quality issues. This process stability supports consistent delivery schedules and simplifies the qualification of suppliers for regulated markets.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the complete removal of organic co-solvents like t-amyl alcohol or dioxane from the reaction matrix. In traditional processes, these solvents represent a significant variable cost, not only in terms of purchase price but also in the energy and infrastructure required for their distillation and recycling. By operating in a pure water phase, the downstream processing is drastically simplified, often requiring only a simple extraction or filtration step to isolate the product. Additionally, the ability to run reactions at room temperature eliminates the need for expensive cryogenic cooling systems and the associated energy consumption, leading to substantial operational expenditure savings. The high conversion rate (>99%) further contributes to cost efficiency by minimizing the loss of valuable starting materials, ensuring that nearly every gram of substrate is converted into saleable product.
- Enhanced Supply Chain Reliability: The robustness of the mutant lipases under ambient conditions significantly de-risks the manufacturing process. Conventional enzymatic processes that rely on strict low-temperature control are vulnerable to equipment failures or power fluctuations that can compromise an entire batch. In contrast, this new technology tolerates a broader temperature range (10-40°C), providing a safety margin that ensures consistent product quality even in varying plant conditions. Furthermore, the use of water as the primary reaction medium removes the safety hazards and regulatory burdens associated with storing and handling large volumes of flammable organic solvents. This simplifies logistics and warehousing requirements, allowing for more flexible and resilient supply chain operations that can adapt quickly to changes in demand without compromising safety or compliance standards.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is often hindered by heat transfer limitations and solvent safety concerns, both of which are mitigated by this water-phase technology. The absence of volatile organic compounds (VOCs) simplifies environmental compliance, reducing the burden on waste treatment facilities and lowering the facility's overall carbon footprint. The high substrate concentration tolerance (up to 300 g/L) demonstrated in the patent examples indicates that the process is highly efficient in terms of reactor volume utilization, allowing for greater throughput in existing infrastructure. This scalability is crucial for meeting the growing global demand for chiral pharmaceutical intermediates, enabling manufacturers to ramp up production from pilot scale to multi-ton commercial quantities without the need for specialized, solvent-rated reactors or extensive process re-validation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these lipase mutants in industrial settings. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the technology's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this biocatalytic route for their specific product portfolios.
Q: What are the primary advantages of these lipase mutants over wild-type enzymes?
A: Unlike wild-type lipases that often require low temperatures and organic co-solvents to achieve moderate selectivity, these mutants operate efficiently at room temperature (10-40°C) in a pure water phase, delivering superior enantiomeric excess (>95% e.e.) and conversion rates (>99%).
Q: Which specific amino acid sites were targeted for mutation to improve stereoselectivity?
A: The patent identifies key mutation sites in the acyl and alcohol binding pockets, specifically positions 140, 154, 281, and 282 for Candida antarctica lipase (PAL), and corresponding positions 145, 159, 287, and 288 for Sporisorium reilianum lipase (SRL).
Q: Can this process be scaled for industrial production of chiral intermediates?
A: Yes, the elimination of hazardous organic co-solvents and the ability to run reactions at ambient temperatures significantly simplifies downstream processing and safety protocols, making the process highly suitable for large-scale commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-Monoesters Supplier
The technological potential of these lipase mutants represents a significant opportunity for the pharmaceutical industry to produce high-value chiral intermediates more sustainably and cost-effectively. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative biocatalytic routes into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped to handle the precise analytical requirements of chiral molecules, guaranteeing that every batch meets the exacting standards required by global regulatory bodies.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be tailored to your specific manufacturing needs. By leveraging our capabilities, you can access a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this water-phase enzymatic process for your target molecules. We encourage you to contact us to request specific COA data for similar chiral intermediates and to discuss route feasibility assessments that can accelerate your project timelines and reduce your overall development costs.
