Advanced One-Pot Synthesis of 2-Bromodifluoromethyl-1,3-Oxazoline Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access fluorinated heterocyclic scaffolds, which serve as critical building blocks for next-generation therapeutics and agrochemicals. Patent CN102180841B introduces a significant advancement in this domain by detailing a novel synthesis route for 2-bromodifluoromethyl-substituted 1,3-oxadiazoline compounds. These fluorine-containing structures are renowned for their ability to enhance metabolic stability and bioavailability in drug candidates. The core innovation lies in the efficient construction of the heterocyclic ring bearing a reactive bromodifluoromethyl group, which acts as a versatile handle for further functionalization. This technical breakthrough addresses the longstanding challenge of introducing gem-difluoro motifs into complex heterocycles without resorting to hazardous or multi-step procedures. The general structural framework of these high-value intermediates is characterized by a fused or saturated heterocyclic ring where the heteroatom can be oxygen, sulfur, or nitrogen, providing a broad scope for molecular diversity. 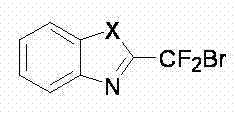 By leveraging this patented technology, manufacturers can access a library of compounds that are pivotal for the development of novel active pharmaceutical ingredients (APIs) and specialized functional materials.
By leveraging this patented technology, manufacturers can access a library of compounds that are pivotal for the development of novel active pharmaceutical ingredients (APIs) and specialized functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of fluorinated oxazolines and related heterocycles has been plagued by inefficiencies that hinder commercial viability. Conventional routes often require the pre-functionalization of starting materials, involving separate steps to introduce the fluorine atoms before cyclization can occur. These multi-step sequences inevitably lead to accumulated yield losses and generate substantial amounts of chemical waste, which poses both economic and environmental burdens. Furthermore, many classical methods rely on harsh reaction conditions, such as extremely low temperatures or the use of highly toxic fluorinating agents that require specialized containment and handling protocols. The isolation of reactive intermediates in these traditional pathways is often difficult due to their instability, leading to decomposition and the formation of complex impurity profiles that are costly to remove. Additionally, the reliance on stoichiometric amounts of expensive reagents in older methodologies drives up the cost of goods significantly, making the final intermediates less competitive in the global supply chain. These factors collectively create a bottleneck for R&D teams aiming to scale up fluorinated heterocycle production for clinical or commercial purposes.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN102180841B offers a streamlined, one-pot synthetic strategy that fundamentally reshapes the production landscape. This novel approach utilizes an Appel-type reaction mechanism where bromodifluoroacetic acid is activated in situ by a combination of triphenylphosphine and carbon tetrabromide. This activation allows for the direct condensation with various amine or phenol substrates, such as o-aminophenol or phenylalaninol, to form the target heterocyclic ring in a single operational step. The elimination of intermediate isolation steps not only saves time but also drastically reduces the consumption of solvents and purification materials. The reaction conditions are remarkably mild, often proceeding at temperatures ranging from room temperature to moderate heating, which enhances safety and energy efficiency. Moreover, the post-treatment process is simplified to filtration and solvent evaporation, followed by standard purification techniques, making it highly amenable to industrial application. This methodological shift represents a paradigm change in how fluorinated building blocks are manufactured, offering a clear path toward cost reduction and process intensification.
Mechanistic Insights into PPh3-CBr4 Mediated Cyclization
The success of this synthesis relies on a sophisticated yet elegant mechanistic pathway that ensures high conversion and selectivity. The reaction initiates with the activation of bromodifluoroacetic acid by triphenylphosphine and carbon tetrabromide, generating a highly reactive acyl bromide species. This intermediate subsequently undergoes nucleophilic attack by the amine or hydroxyl group of the substrate, leading to the formation of an imidoyl bromide intermediate. Kinetic studies suggest that the formation of this imidoyl bromide species is the rate-determining step of the entire transformation, dictating the overall reaction speed. Once formed, this key intermediate is primed for the subsequent intramolecular nucleophilic ring closure. The presence of an organic base, such as triethylamine or pyridine, facilitates the deprotonation necessary for the cyclization to proceed rapidly. This two-step cascade within a single pot ensures that the reactive species are consumed as soon as they are generated, minimizing side reactions. 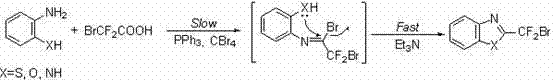 The precise control over these mechanistic steps allows for the accommodation of various substrates, including those with sensitive functional groups, without compromising the integrity of the final product.
The precise control over these mechanistic steps allows for the accommodation of various substrates, including those with sensitive functional groups, without compromising the integrity of the final product.
Controlling the impurity profile in fluorinated heterocycle synthesis is critical for meeting the stringent quality standards required by the pharmaceutical industry. The mechanism described inherently limits the formation of by-products because the reactive intermediates are not isolated, thereby reducing their exposure to potential degradation pathways such as hydrolysis or oxidation. The use of inert gas protection, typically nitrogen or argon, further safeguards the reaction mixture from moisture and oxygen, which are common sources of impurities in organophosphorus chemistry. Additionally, the choice of solvent plays a pivotal role in managing solubility and reaction kinetics; solvents like dichloromethane or acetonitrile provide an optimal medium that supports the dissolution of both organic substrates and inorganic salts formed during the reaction. The final purification via column chromatography effectively removes triphenylphosphine oxide and other phosphorus-containing by-products, ensuring a high-purity final product. This robust control over the chemical environment ensures that the resulting 2-bromodifluoromethyl-substituted compounds meet the rigorous specifications necessary for downstream applications in drug discovery.
How to Synthesize 2-Bromodifluoromethyl-1,3-Oxazoline Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and safety. The process begins by dissolving the amine substrate and bromodifluoroacetic acid in a suitable organic solvent under an inert atmosphere to prevent moisture ingress. Triphenylphosphine and carbon tetrabromide are then added in controlled molar ratios to ensure complete activation of the acid without excessive waste. The reaction mixture is maintained at a specific temperature range, typically between 0 to 140 degrees Celsius depending on the substrate reactivity, for a duration of 6 to 32 hours. Upon completion, the solid by-products are removed via filtration, and the solvent is evaporated under reduced pressure to isolate the crude material. The detailed standardized synthesis steps, including specific molar ratios and work-up procedures, are outlined in the guide below to ensure reproducibility and safety compliance.
- Dissolve bromodifluoroacetic acid and amine substrates in organic solvent under inert gas.
- Add triphenylphosphine, carbon tetrabromide, and organic base at controlled temperatures.
- Stir for 6-32 hours, filter, evaporate solvent, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method offers tangible strategic advantages that extend beyond mere technical feasibility. The primary benefit lies in the significant simplification of the manufacturing process, which directly translates to reduced operational expenditures. By consolidating multiple reaction steps into a single one-pot procedure, facilities can reduce the consumption of solvents, energy, and labor hours associated with intermediate handling and purification. This efficiency gain allows for a more competitive pricing structure for the final fluorinated intermediates, providing a cost advantage in the sourcing of critical raw materials for drug production. Furthermore, the reliance on readily available and stable reagents such as triphenylphosphine and carbon tetrabromide mitigates the risk of supply chain disruptions often associated with specialized or hazardous fluorinating agents. The robustness of the process also implies a higher success rate in batch production, ensuring consistent availability of materials for downstream synthesis.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the avoidance of multi-step isolation procedures result in substantial cost savings. Traditional methods often require expensive palladium or copper catalysts that necessitate rigorous removal steps to meet residual metal specifications, adding both time and cost to the process. In contrast, this organophosphorus-mediated reaction generates by-products that are easily removed through standard aqueous work-ups or filtration. The reduction in unit operations means less equipment occupancy time and lower utility costs per kilogram of product. Additionally, the high atom economy of the reaction minimizes the volume of chemical waste generated, reducing disposal costs and environmental compliance burdens. These factors collectively contribute to a leaner manufacturing model that enhances the overall profitability of producing high-value fluorinated intermediates.
- Enhanced Supply Chain Reliability: The use of commodity chemicals as reagents ensures a stable and resilient supply chain. Triphenylphosphine, carbon tetrabromide, and common organic bases are produced globally in large volumes, reducing the dependency on single-source suppliers for niche reagents. This availability allows for flexible procurement strategies and the ability to scale production rapidly in response to market demand without long lead times for raw material acquisition. The simplicity of the reaction setup also means that production can be easily transferred between different manufacturing sites or contract manufacturing organizations without the need for specialized equipment modifications. This flexibility is crucial for maintaining continuity of supply in the face of geopolitical or logistical challenges, ensuring that pharmaceutical production schedules remain uninterrupted.
- Scalability and Environmental Compliance: The process is inherently scalable from gram to multi-ton quantities due to its straightforward exothermic profile and lack of hazardous gas evolution. The reaction can be safely conducted in standard glass-lined or stainless steel reactors commonly found in fine chemical manufacturing plants. From an environmental perspective, the reduction in solvent usage and waste generation aligns with green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. The absence of heavy metals in the reaction mixture simplifies wastewater treatment and reduces the environmental footprint of the manufacturing site. This alignment with sustainability goals not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the supply chain, which is increasingly important for end-user pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these fluorinated heterocycles. The answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production workflows or R&D pipelines. The insights provided here aim to clarify the operational benefits and technical capabilities associated with this synthesis method.
Q: What is the key advantage of this synthesis method?
A: The method utilizes a one-pot reaction strategy that eliminates the need for isolating unstable intermediates, significantly simplifying post-treatment and improving overall operational efficiency.
Q: What substrates are compatible with this reaction?
A: The process is versatile, accommodating o-aminophenol, o-aminothiophenol, o-phenylenediamine, and phenylalaninol to produce oxazoles, thiazoles, imidazoles, and oxazolines respectively.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the use of readily available reagents and standard organic solvents like dichloromethane or toluene makes the protocol highly adaptable for commercial scale-up without specialized equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromodifluoromethyl-1,3-Oxazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in the development of innovative pharmaceuticals and advanced materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation. Our expertise in handling complex fluorination chemistries allows us to optimize the patented process described in CN102180841B for maximum yield and cost-efficiency, providing our partners with a competitive edge in their respective markets.
We invite procurement leaders and R&D directors to collaborate with us to explore how this advanced synthesis route can enhance your supply chain resilience and product performance. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact our technical procurement team to obtain specific COA data and route feasibility assessments tailored to your project needs. Let us be your partner in navigating the complexities of fine chemical manufacturing and delivering value through innovation.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
