Advanced Photoelectrochemical Manufacturing of Alpha-Alpha-Dichloroaryl Ketone Intermediates
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for greener, more sustainable synthetic methodologies. A pivotal advancement in this domain is detailed in patent CN115110104A, which discloses a novel photoelectrochemical synthesis method for producing α,α-dichloroaryl ketone compounds. This technology represents a paradigm shift from traditional stoichiometric oxidation towards a catalytic, electricity-driven process that leverages the power of light and electrochemistry to achieve high-efficiency transformations. The core innovation lies in the ability to utilize readily available aryl alkyne precursors, as illustrated by the general structural framework in the accompanying visual data, to generate valuable dichloro-ketone scaffolds without the reliance on toxic transition metals or harsh chemical oxidants.
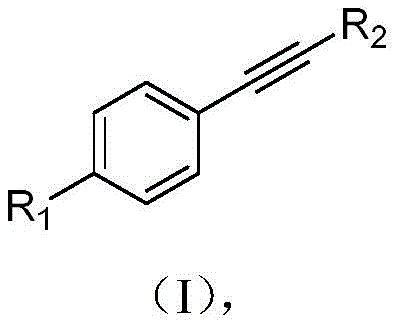
For R&D directors and process chemists, the implications of this patent are profound, offering a pathway to synthesize complex intermediates with superior purity profiles and reduced impurity burdens. The method employs a cerium-based photocatalytic system coupled with constant current electrolysis, creating a reaction environment that is both atom-economical and environmentally benign. By integrating light energy with electrochemical potential, the process activates chlorine sources efficiently, enabling the oxidative dihalogenation of alkynes under mild conditions. This approach not only addresses the growing regulatory pressure regarding heavy metal residues in pharmaceutical intermediates but also provides a robust platform for the scalable production of high-value chemical building blocks essential for drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of α,α-dihalogenated carbonyl derivatives has relied heavily on classical halogenation strategies that involve the use of stoichiometric amounts of hazardous reagents such as N-bromosuccinimide (NBS), trichloroisocyanuric acid (TCCA), or elemental halogens. These traditional protocols often necessitate the presence of strong acids like hydrochloric acid or sulfuric acid to drive the reaction forward, creating significant safety hazards and corrosion issues within industrial reactor systems. Furthermore, the reliance on chemical oxidants results in poor atom economy, generating substantial quantities of organic and inorganic waste that require costly disposal and treatment procedures. The use of separated electrolytic cells in earlier electrochemical attempts further complicated the setup, limiting the substrate scope particularly for internal alkynes, and failing to provide a universally applicable solution for diverse chemical libraries.
The Novel Approach
In stark contrast, the photoelectrochemical method described in the patent data introduces a streamlined, undivided cell configuration that operates under constant current conditions with visible light irradiation. This novel approach eliminates the need for exogenous oxidants by utilizing electricity as the clean reagent to drive the redox cycles of the cerium catalyst. The reaction system is remarkably simple, employing common inert electrodes that do not require specialized surface modifications, thereby reducing equipment costs and maintenance overheads. By avoiding strong acids and toxic oxidants, the process inherently mitigates safety risks associated with thermal runaways or hazardous gas evolution, while the mild reaction conditions preserve sensitive functional groups on the substrate, allowing for broader chemical diversity in the final products.
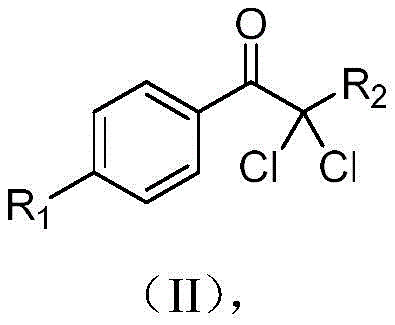
Mechanistic Insights into CeCl3-Catalyzed Photoelectrochemical Oxidation
The mechanistic elegance of this synthesis lies in the synergistic interplay between electrochemical oxidation and ligand-to-metal charge transfer (LMCT) processes initiated by light. The reaction initiates with the anodic oxidation of the cerium(III) chloride catalyst to generate a high-valent cerium(IV) species at the electrode surface. Upon irradiation with 395nm LEDs, this cerium(IV) complex undergoes a light-induced LMCT event, which facilitates the homolytic cleavage of the cerium-chlorine bond to release reactive chlorine radicals. These radicals are then captured by the aryl alkyne substrate to form a vinyl radical intermediate, which is subsequently oxidized at the anode to yield a carbocation species. This precise control over radical generation and propagation ensures high selectivity and minimizes side reactions that typically plague free-radical halogenation processes.
Following the formation of the carbocation, water molecules present in the solvent mixture act as nucleophiles, attacking the electrophilic center to form an enol intermediate after deprotonation. This enol species then undergoes a tandem sequence involving further deprotonation and electrochemical oxidation to generate an enol radical, which finally captures a second chlorine radical to afford the desired α,α-dichloroaryl ketone product. This intricate cascade effectively utilizes water as the oxygen source, demonstrating exceptional atom economy compared to methods requiring external oxygen donors. The continuous regeneration of the active cerium catalyst at the electrode ensures a sustained catalytic cycle, maintaining high reaction rates and consistent yields throughout the process duration without the accumulation of deactivated catalyst species.
How to Synthesize Alpha-Alpha-Dichloroaryl Ketones Efficiently
Implementing this photoelectrochemical protocol requires careful attention to the optimization of reaction parameters to maximize yield and purity. The standard procedure involves charging an undivided electrolytic cell with a specific mixture of cerium trichloride as the photocatalyst, magnesium chloride hexahydrate as the chlorine source, and lithium perchlorate as the supporting electrolyte in an acetonitrile-water solvent system. The reaction is conducted at room temperature under a nitrogen atmosphere to prevent unwanted side reactions with atmospheric oxygen, while a constant current of 4mA is applied to drive the electrochemical turnover. Detailed standardized synthesis steps for replicating this high-efficiency route are provided in the guide below.
- Prepare the electrolytic cell by adding cerium trichloride, lithium perchlorate, magnesium chloride hexahydrate, and a mixed solvent of acetonitrile and water.
- Introduce the aryl alkyne substrate and apply a constant current of 4mA under 395nm LED irradiation at room temperature for 6 hours.
- Upon completion, remove the solvent via rotary evaporation and purify the crude residue using flash silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers compelling advantages by fundamentally simplifying the raw material basket and reducing dependency on volatile commodity chemicals. The elimination of stoichiometric oxidants like NBS and TCCA removes a significant cost driver from the bill of materials, as these reagents are often subject to price fluctuations and strict transportation regulations due to their hazardous nature. Furthermore, the use of inexpensive and abundant inorganic salts such as magnesium chloride and cerium chloride ensures a stable and reliable supply chain, insulating production costs from the volatility associated with complex organic reagents. The simplicity of the workup procedure, which involves basic solvent removal and chromatography, further contributes to overall process efficiency and reduced operational expenditures.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the removal of expensive transition metal catalysts and stoichiometric oxidants, which traditionally account for a large portion of raw material costs in fine chemical synthesis. By substituting these costly inputs with electricity and inexpensive inorganic salts, manufacturers can achieve substantial savings in direct material expenses while simultaneously reducing waste disposal costs associated with heavy metal residues. The high atom efficiency of the reaction means that less raw material is wasted as byproducts, leading to improved overall process mass intensity and lower cost per kilogram of the final active pharmaceutical ingredient intermediate.
- Enhanced Supply Chain Reliability: Sourcing reliability is markedly improved as the key reagents, including cerium chloride and magnesium chloride, are commodity chemicals with robust global supply networks, unlike specialized oxidants that may have limited suppliers. The operational simplicity of the reactor setup, which utilizes standard inert electrodes without the need for custom fabrication or frequent replacement, ensures consistent production uptime and reduces the risk of supply disruptions caused by equipment failure. This stability allows for more accurate demand forecasting and inventory management, enabling procurement teams to secure long-term contracts with greater confidence in delivery schedules.
- Scalability and Environmental Compliance: The environmental profile of this photoelectrochemical method aligns perfectly with increasingly stringent global regulations regarding industrial emissions and waste management. The absence of toxic heavy metals and strong acids in the waste stream simplifies effluent treatment processes, reducing the burden on environmental compliance departments and lowering the risk of regulatory fines. Additionally, the mild reaction conditions and simple reactor design facilitate straightforward scale-up from laboratory to pilot and commercial scales, ensuring that the process remains efficient and safe even when producing metric ton quantities of material for global markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photoelectrochemical synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for evaluating the feasibility of adopting this method in your own manufacturing operations. Understanding these details is crucial for making informed decisions about process integration and supply chain strategy.
Q: What are the primary safety advantages of this photoelectrochemical method?
A: Unlike traditional methods requiring strong acids and stoichiometric oxidants like NBS or TCCA, this process utilizes mild electrolytes and avoids hazardous exogenous oxidants, significantly reducing safety risks and heavy metal residue concerns.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the method employs general inert platinum electrodes that require no complex modification and operates under ambient temperature and pressure, making the reaction system simple, safe, and highly adaptable for commercial scale-up.
Q: How does this method improve atom economy compared to conventional halogenation?
A: By using water as the oxygen source and electricity as the driving force for oxidation, the process eliminates the need for wasteful chemical oxidants, resulting in higher atomic efficiency and reduced environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Alpha-Dichloroaryl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced photoelectrochemical technologies in modernizing the production of critical pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN115110104A can be successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of alpha-alpha-dichloroaryl ketone delivered meets the highest standards required by global regulatory bodies.
We invite you to collaborate with our technical team to explore how this green synthesis route can optimize your specific supply chain requirements and reduce your overall manufacturing footprint. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and commercialization goals, ensuring a seamless transition from concept to commercial reality.
