Advanced Synthesis of 2,4-Diamino[4’-(4-Phenylethynylphthalimido)]Diphenyl Ether for High-Performance Resins
Introduction to Patent CN100586931C and Technical Breakthroughs
The chemical landscape for high-performance aerospace materials is constantly evolving, driven by the need for resins that can withstand extreme thermal and mechanical stress. Patent CN100586931C introduces a sophisticated synthetic methodology for producing 2,4-diamino[4’-(4-phenylethynylphthalimido)]diphenyl ether, a critical diamine monomer designed to enhance the properties of polyimide oligomers. This specific molecular architecture incorporates a phenylethynyl group directly into the side chain of the phthalimide structure, a strategic modification that fundamentally alters the curing behavior of the resulting polymer matrix. By shifting the reactive group from the chain end to the side chain, the invention addresses longstanding issues regarding steric hindrance and curing efficiency that have plagued previous generations of thermosetting imides. For R&D directors and procurement specialists in the advanced materials sector, understanding this patented route is essential for sourcing reliable polyimide monomer supplier partners who can deliver consistent quality for next-generation composite applications.
The significance of this technology extends beyond mere academic interest; it represents a tangible leap forward in the manufacturability of high-temperature resins used in adhesives, coatings, and composite matrix systems. Traditional methods often struggle to balance the trade-off between processability and final thermal performance, but this novel approach leverages the electron-withdrawing nature of the substituents to accelerate cure rates without compromising the integrity of the polymer backbone. The patent details a robust three-step synthesis that begins with readily available raw materials like p-aminophenol and 4-phenylethynylphthalic anhydride, ensuring that the supply chain remains resilient and cost-effective. As we delve deeper into the technical specifics, it becomes clear that this process offers a viable pathway for the commercial scale-up of complex polymer additives, meeting the rigorous demands of the global aerospace and electronics industries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenylethynyl-containing imide oligomers has relied heavily on end-capping strategies, where the reactive phenylethynyl groups are positioned at the termini of the polymer chains. While effective to a degree, this conventional architecture introduces significant kinetic barriers during the curing phase. The movement of these end-groups is severely restricted by the bulk of the polymer chain itself, creating a steric environment that hinders the cross-linking reaction. Consequently, manufacturers are often forced to employ excessively high curing temperatures or extend processing times significantly to achieve full conversion, which can lead to thermal degradation of other sensitive components in the formulation. Furthermore, the variability in cure rates associated with these end-capped monomers complicates quality control, making it difficult to predict the final mechanical properties of the cured resin with high precision. These inefficiencies translate directly into higher energy consumption and reduced throughput in industrial settings, posing a challenge for cost reduction in high-performance resin manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in CN100586931C reimagines the molecular design by anchoring the phenylethynyl functionality to the side chain of the phthalimide ring. This structural innovation ensures that the reactive groups are more accessible and less encumbered by the polymer backbone, thereby facilitating a much smoother curing process. The patent explicitly notes that the curing of the phenylethynyl group in this specific diamine follows second-order reaction kinetics with a rate constant k=0.45, a metric that indicates a significantly faster and more predictable reaction profile compared to its predecessors. Because the side-chain structure mimics the terminal group structure, the curing reaction between different oligomer chains is simplified, leading to a more uniform cross-linked network. This not only expands the processing window to higher temperatures, benefiting material flow during molding, but also results in a cured resin with superior thermal oxidation stability and dimensional stability. For supply chain heads, this translates to a more reliable product with consistent batch-to-batch performance.
Mechanistic Insights into Cyclodehydration and Nucleophilic Substitution
The core of this synthetic route lies in a meticulously orchestrated sequence of reactions that prioritize yield and purity at every stage. The first critical step involves the high-temperature cyclodehydration of p-aminophenol with 4-phenylethynylphthalic anhydride or its acid counterpart. This reaction is conducted at temperatures ranging from 120°C to 160°C in solvents such as glacial acetic acid, toluene, or phenolic derivatives, which serve to drive the equilibrium towards the formation of the N-(4-hydroxyphenyl)-4’-phenylethynylphthalimide intermediate. The choice of solvent is paramount here, as it must effectively solubilize the reactants while allowing for the removal of water generated during the imidization process. Following this, the intermediate undergoes a nucleophilic substitution reaction with 2,4-dinitrochlorobenzene. This step is catalyzed by bases such as triethylamine or anhydrous potassium carbonate in polar aprotic solvents like N-methyl-2-pyrrolidone (NMP) or dimethylformamide (DMF). The mechanism relies on the activation of the phenolic hydroxyl group, which attacks the electron-deficient aromatic ring of the dinitrochlorobenzene, displacing the chloride ion and forming the stable ether linkage.
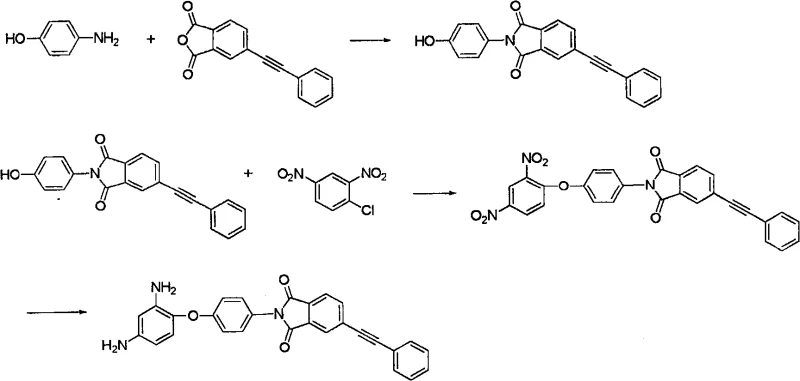
The final transformation involves the reduction of the dinitro intermediate to the target diamine, a step that requires careful control to avoid over-reduction or side reactions affecting the sensitive phenylethynyl triple bond. The patent describes the use of metal chlorides such as stannous chloride, ferrous chloride, or zinc chloride in concentrated hydrochloric acid media. This reduction is typically performed at moderate temperatures between 20°C and 100°C, ensuring that the delicate alkyne functionality remains intact while the nitro groups are efficiently converted to amines. Impurity control is rigorously managed through a series of washing steps involving dilute hydrochloric acid, ammonia water, and pure water, followed by recrystallization from dioxane. This multi-stage purification protocol is essential for achieving the high-purity OLED material or aerospace-grade monomer specifications required by downstream users. By understanding these mechanistic nuances, R&D teams can better appreciate the robustness of the process and its suitability for large-scale production environments where consistency is key.
How to Synthesize 2,4-Diamino[4’-(4-Phenylethynylphthalimido)]Diphenyl Ether Efficiently
Implementing this synthesis on an industrial scale requires a deep understanding of the reaction parameters and safety protocols associated with each step. The process is designed to be scalable, utilizing common chemical reagents and standard reactor configurations that are widely available in fine chemical manufacturing facilities. The initial imidization step sets the foundation for the entire sequence, demanding precise temperature control to maximize the formation of the phthalimide ring while minimizing the formation of byproducts. Subsequent steps build upon this intermediate, with the nucleophilic substitution requiring anhydrous conditions to prevent hydrolysis of the reactive chlorobenzene species. Finally, the reduction step must be managed with care to handle the exothermic nature of the reaction and the handling of acidic waste streams. For those seeking to replicate this high-purity polyimide monomer synthesis, adherence to the molar ratios and solvent volumes specified in the patent is crucial for maintaining optimal yields.
- Perform high-temperature cyclodehydration of p-aminophenol and 4-phenylethynylphthalic anhydride at 120-160°C to form the phthalimide intermediate.
- Conduct nucleophilic substitution between the hydroxy-phthalimide intermediate and 2,4-dinitrochlorobenzene using a base catalyst in polar aprotic solvents.
- Reduce the dinitro compound using metal chlorides (SnCl2, FeCl2, or ZnCl2) in acidic media to yield the final diamino monomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers compelling advantages that resonate deeply with procurement managers and supply chain leaders focused on efficiency and reliability. The primary benefit lies in the simplification of the curing process for the end-user, which indirectly reduces the manufacturing costs for composite part producers by lowering energy requirements and cycle times. For the chemical supplier, the route is advantageous because it relies on commodity chemicals like p-aminophenol and dinitrochlorobenzene, which are readily available in the global market, thereby mitigating the risk of raw material shortages. This accessibility ensures a stable supply chain, reducing lead time for high-purity polyimide monomers and allowing for more accurate demand forecasting. Furthermore, the elimination of exotic or highly specialized catalysts in favor of common bases and metal salts streamlines the procurement process and reduces the overall cost of goods sold.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing inexpensive and abundant starting materials, avoiding the need for precious metal catalysts that often drive up production expenses in fine chemical synthesis. The high yields reported in the patent examples, often exceeding 80% across multiple steps, indicate a highly efficient atom economy that minimizes waste and maximizes output per batch. Additionally, the use of standard solvents that can potentially be recovered and recycled further contributes to substantial cost savings in the long run. By removing the dependency on complex, multi-step protection-deprotection strategies common in other monomer syntheses, the overall operational expenditure is drastically simplified, making the final product more competitive in price-sensitive markets.
- Enhanced Supply Chain Reliability: The reliance on bulk chemicals with established global supply networks ensures that production schedules are not disrupted by the scarcity of niche reagents. This robustness is critical for maintaining continuous operations in high-volume manufacturing environments where downtime is costly. The synthetic pathway is also flexible regarding solvent choices, offering alternatives like toluene, acetic acid, or DMF depending on regional availability and regulatory constraints. This adaptability allows manufacturers to pivot quickly in response to supply chain fluctuations, ensuring that delivery commitments to aerospace and electronics clients are consistently met without compromise.
- Scalability and Environmental Compliance: The reaction conditions, primarily operating at atmospheric pressure and moderate temperatures, are inherently safer and easier to scale from pilot plants to multi-ton production facilities. The workup procedures involve straightforward filtration and washing steps, which are easily automated and integrated into continuous processing lines. Moreover, the ability to recycle solvents and the absence of heavy metal contaminants in the final product simplify waste treatment and disposal, aligning with increasingly stringent environmental regulations. This ease of scale-up supports the commercial expansion of complex polymer additives, enabling suppliers to meet growing market demand for advanced materials without significant capital investment in specialized equipment.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this technology, we have compiled a set of frequently asked questions based on the detailed specifications within the patent documentation. These inquiries address common concerns regarding the curing behavior, solvent selection, and application suitability of the synthesized monomer. Understanding these aspects is vital for technical teams evaluating the feasibility of incorporating this material into their existing formulations. The answers provided are derived directly from the experimental data and theoretical framework presented in the patent, ensuring accuracy and relevance for decision-makers.
Q: How does the side-chain phenylethynyl group affect curing kinetics?
A: Unlike end-capped groups which suffer from steric hindrance, the side-chain phenylethynyl group in this monomer follows second-order reaction kinetics with a rate constant k=0.45, allowing for simplified curing at lower temperatures.
Q: What are the primary solvents used in the imidization step?
A: The process utilizes solvents such as glacial acetic acid, toluene, xylene, or phenolic derivatives, typically at 4-10 times the weight of the reactants to ensure efficient cyclodehydration.
Q: Why is this monomer preferred for aerospace applications?
A: The resulting polyimide oligomers exhibit high thermal oxidation stability, high glass transition temperatures, and superior hydrolysis resistance, making them ideal for harsh aerospace environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Diamino[4’-(4-Phenylethynylphthalimido)]Diphenyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance monomers play in the development of next-generation aerospace and electronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with unwavering consistency. We are committed to delivering products that meet stringent purity specifications, backed by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Whether you are developing new polyimide resins or optimizing existing composite formulations, our capability to manufacture complex intermediates like 2,4-diamino[4’-(4-phenylethynylphthalimido)]diphenyl ether positions us as a strategic partner in your supply chain.
We invite you to engage with our technical procurement team to discuss how this patented synthesis can benefit your specific application needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how switching to our optimized manufacturing route can improve your bottom line. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project timelines. Let us collaborate to bring high-quality, cost-effective advanced materials to the market, driving innovation in the fields of polymers and specialty chemicals together.
