Advanced Pd-Catalyzed Synthesis of N-Alkyl Tetrahydrocarbazolone Derivatives for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical active pharmaceutical ingredient (API) intermediates, particularly those serving high-volume therapeutic areas such as oncology supportive care. Patent CN101914056B introduces a significant advancement in the preparation of N-alkyl substituted tetrahydrocarbazolone derivatives, which serve as pivotal precursors for the synthesis of Ondansetron, a widely prescribed 5-HT3 receptor antagonist. This patented methodology addresses long-standing challenges in heterocyclic chemistry by employing a direct oxidative coupling strategy that streamlines the construction of the carbazole core. Unlike traditional approaches that often rely on pre-functionalized hydrazines or harsh cyclization conditions, this innovation utilizes a palladium-catalyzed system with molecular oxygen, offering a cleaner and more atom-economical pathway. For R&D directors and process chemists, the ability to introduce diverse substituents on the aromatic ring while maintaining high regioselectivity represents a substantial leap forward in process flexibility. The technical breakthrough lies not just in the final yield, but in the operational simplicity that translates directly into manufacturing efficiency and reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetrahydrocarbazolone scaffolds has been dominated by the Fischer indole synthesis or variations of the Heck reaction, both of which present distinct disadvantages for modern GMP manufacturing. The Fischer synthesis typically requires strongly acidic conditions and elevated temperatures that can degrade sensitive functional groups, limiting the scope of accessible analogues and often leading to complex impurity profiles that are difficult to purge. Furthermore, conventional strategies frequently necessitate the prior synthesis of N-substituted hydrazines, which are potentially hazardous and unstable reagents, adding significant safety risks and cost burdens to the supply chain. Alternative methods involving intramolecular cyclization of halo-anilines often require stoichiometric amounts of base or expensive transition metal catalysts that are difficult to remove to ppm levels required for pharmaceutical products. These legacy processes often suffer from poor atom economy, generating substantial waste streams and requiring extensive downstream purification, which ultimately inflates the cost of goods and extends production lead times for critical anti-emetic medications.
The Novel Approach
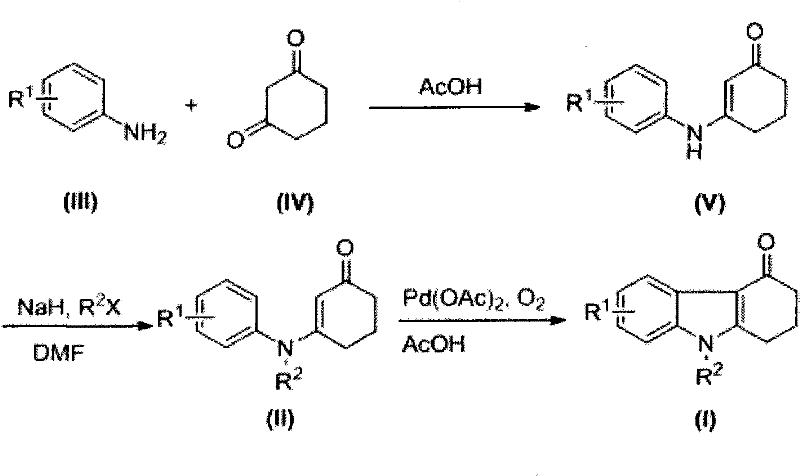
The methodology disclosed in CN101914056B circumvents these historical bottlenecks by utilizing a direct C-H activation strategy mediated by palladium catalysis. This novel approach begins with a mild condensation between readily available aniline derivatives and 1,3-cyclohexanedione, followed by a straightforward N-alkylation step to install the desired substituent. The cornerstone of this innovation is the final oxidative cyclization step, where the aromatic ring couples directly with the alpha-position of the enone system under an oxygen atmosphere. This eliminates the need for pre-halogenated starting materials, thereby reducing raw material costs and simplifying the synthetic sequence. By leveraging molecular oxygen as the terminal oxidant, the process generates water as the primary byproduct, aligning perfectly with green chemistry principles. This streamlined three-step sequence allows for the rapid generation of diverse libraries of N-alkyl substituted derivatives, enabling faster scale-up and more reliable supply continuity for downstream API manufacturers.
Mechanistic Insights into Pd-Catalyzed Oxidative Cyclization
The core chemical transformation in this patent relies on a sophisticated palladium-catalyzed C-H functionalization mechanism that achieves sp2-sp2 carbon bond formation with high precision. In the final cyclization step, the N-alkyl substituted aniline cyclohex-2-enone compound acts as the substrate, coordinating with the palladium acetate catalyst in glacial acetic acid. The mechanism likely proceeds through a concerted metalation-deprotonation (CMD) pathway, where the palladium center activates the ortho-C-H bond of the aniline moiety. Simultaneously, the enolate form of the cyclohexenone ring coordinates to the metal center, facilitating the formation of a palladacycle intermediate. The introduction of an oxygen stream serves a dual purpose: it re-oxidizes the reduced palladium species back to its active state, sustaining the catalytic cycle, and it drives the thermodynamic equilibrium towards the formation of the aromatic carbazole system. This oxidative dehydrogenation is crucial for aromatization, ensuring the final product possesses the rigid planar structure required for biological activity in serotonin receptor binding.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based cyclizations which often produce polymeric byproducts. The directed nature of the palladium coordination ensures that cyclization occurs specifically at the ortho-position relative to the nitrogen atom, minimizing the formation of regioisomers. The use of acetic acid as both solvent and proton source helps to stabilize the intermediate species and solubilize the polar transition states. Furthermore, the reaction conditions described, specifically temperatures between 100-120°C, are optimized to balance reaction kinetics with thermal stability, preventing the decomposition of the sensitive enone functionality. For quality assurance teams, understanding this mechanistic pathway is vital for defining critical process parameters (CPPs) such as oxygen flow rate and catalyst loading, which directly influence the residual metal content and overall purity of the final N-alkyl substituted tetrahydrocarbazolone derivative.
How to Synthesize N-Alkyl Substituted Tetrahydrocarbazolone Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing reproducibility and ease of execution. The process initiates with the dehydration condensation of aniline derivatives and 1,3-cyclohexanedione in acetic acid, a reaction that proceeds smoothly at moderate temperatures of 30-70°C to yield the enaminone intermediate. Following isolation, the nitrogen atom is alkylated using sodium hydride and a haloalkane in DMF, a standard transformation that allows for the introduction of methyl, benzyl, or other alkyl groups essential for structure-activity relationship studies. The final and most critical step involves dissolving the N-alkylated precursor in dry glacial acetic acid and subjecting it to palladium acetate catalysis under an oxygen atmosphere. Detailed standardized synthetic steps for optimizing yield and purity are provided in the guide below.
- Condense aniline derivatives with 1,3-cyclohexanedione in acetic acid at 30-70°C to form the enaminone intermediate.
- Perform N-alkylation using sodium hydride and haloalkane in DMF to generate the N-substituted precursor.
- Execute oxidative cyclization using palladium acetate and oxygen flow in glacial acetic acid at 100-120°C to close the ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere chemical elegance. The primary value proposition lies in the significant simplification of the raw material portfolio; by eliminating the need for specialized hydrazines or pre-halogenated aromatics, purchasing teams can source commoditized anilines and cyclic diketones from multiple global suppliers, thereby mitigating single-source risk. This diversification of the supply base enhances negotiation leverage and ensures continuity of supply even during market fluctuations. Moreover, the operational simplicity of the three-step sequence reduces the requirement for specialized reactor configurations, allowing for production in standard glass-lined or stainless steel vessels commonly found in multipurpose API facilities. This flexibility lowers the barrier to entry for contract manufacturing organizations and accelerates the timeline from process validation to commercial launch.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive stoichiometric oxidants and the reduction of synthetic steps. Traditional routes often require costly protecting group strategies or harsh reagents that necessitate expensive waste disposal protocols. In contrast, this method utilizes molecular oxygen, which is virtually free and generates benign water as a byproduct, drastically reducing effluent treatment costs. The high yields reported across various substrates, often exceeding 80% in the initial condensation and alkylation steps, mean that less raw material is wasted per kilogram of finished product. Additionally, the efficient use of palladium catalyst, which can potentially be recovered or used at low loadings, minimizes the financial burden associated with precious metal consumption, leading to a lower overall cost of goods sold for the final intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions and the stability of the intermediates. The process tolerates a wide range of functional groups, including halogens, nitro groups, and ethers, meaning that a single platform technology can be used to manufacture a diverse array of derivatives without re-validating entirely new processes. This modularity allows manufacturers to respond rapidly to changes in demand for specific Ondansetron analogues or related therapeutic agents. The use of common solvents like acetic acid and DMF further simplifies logistics, as these materials are readily available in bulk quantities globally. By reducing the complexity of the synthesis, the risk of batch failures due to sensitive reaction parameters is minimized, ensuring a consistent and reliable flow of high-quality intermediates to downstream API producers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces heat transfer and mixing challenges that are absent in the laboratory, but this oxidative cyclization is inherently amenable to scale-up. The exothermic nature of the oxidation can be managed through controlled oxygen sparging, and the use of acetic acid as a solvent facilitates easy product isolation via crystallization or extraction. From an environmental standpoint, the process aligns with increasingly stringent regulatory requirements regarding volatile organic compounds (VOCs) and heavy metal waste. The absence of chlorinated solvents in the final step and the use of oxygen as a green oxidant significantly improve the E-factor of the process. This environmental profile not only reduces compliance costs but also enhances the corporate sustainability metrics of the manufacturing entity, a factor of growing importance to pharmaceutical customers evaluating their supplier base.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the competitive landscape of intermediate supply.
Q: What are the key advantages of this Pd-catalyzed route over traditional Fischer synthesis?
A: This method avoids harsh acidic conditions and complex multi-step sequences typical of Fischer indole synthesis, offering milder conditions and better tolerance for diverse functional groups like halogens and nitro groups.
Q: Is this process suitable for large-scale manufacturing of Ondansetron intermediates?
A: Yes, the use of molecular oxygen as a terminal oxidant and simple workup procedures makes this route highly scalable and environmentally compliant for industrial production.
Q: What purity levels can be achieved with this synthesis method?
A: The patent demonstrates high yields ranging from 60% to over 90% across various substrates, with final purification via column chromatography or recrystallization ensuring stringent pharmaceutical grade purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Alkyl Substituted Tetrahydrocarbazolone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and rigorous process engineering. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields observed in the laboratory are faithfully reproduced in our manufacturing plants. We understand that for critical oncology supportive care drugs like Ondansetron, consistency is paramount; therefore, our facilities are equipped with rigorous QC labs capable of detecting impurities at trace levels to meet stringent purity specifications. We do not just supply chemicals; we provide validated, scalable solutions that de-risk your supply chain and accelerate your time to market.
We invite you to collaborate with us to optimize your sourcing strategy for these high-value intermediates. Our technical procurement team is ready to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. By partnering with us, you gain access to specific COA data and route feasibility assessments that demonstrate exactly how we can enhance your production efficiency. Contact us today to discuss how our advanced Pd-catalyzed synthesis capabilities can support your long-term manufacturing goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
