Advanced Coumarin-Biphenyl Intermediates for Next-Generation Antihypertensive Drug Development
The pharmaceutical landscape for antihypertensive treatments is continuously evolving, driven by the need for agents with superior safety profiles and enhanced efficacy. Patent CN110156761B introduces a groundbreaking class of compounds featuring a unique coumarin-biphenyl skeleton, designed to act as potent Angiotensin II (Ang II) receptor antagonists. This innovation merges the naturally occurring bioactive coumarin framework with the pharmacophore of established Sartan drugs, creating a hybrid structure that demonstrates significant vasodilatory effects in preclinical models. For research and development teams seeking novel scaffolds, this technology offers a promising avenue for developing next-generation cardiovascular therapeutics with potentially reduced side effects compared to current market leaders.
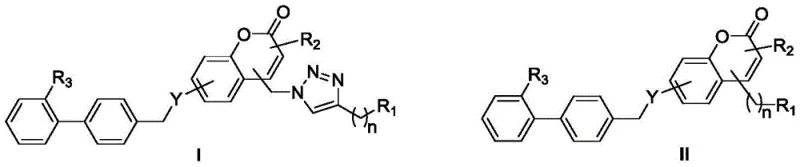
The structural versatility of these compounds allows for extensive modification at multiple positions, including the coumarin ring and the biphenyl linker, enabling fine-tuning of pharmacokinetic properties. By incorporating a triazole ring through efficient click chemistry methodologies, the molecules gain enhanced hydrogen bonding capabilities crucial for receptor binding. This strategic molecular design addresses the limitations of earlier antihypertensive agents while maintaining the core mechanism of blocking the renin-angiotensin system. As a reliable pharmaceutical intermediate supplier, understanding such novel architectures is critical for staying ahead in the competitive API market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of Ang II receptor antagonists often relies on complex multi-step sequences involving harsh reaction conditions and expensive catalysts that can hinder large-scale manufacturing. Conventional Sartans, such as Losartan or Valsartan, typically require the construction of imidazole or other heterocyclic cores which can present challenges in regioselectivity and purification. Furthermore, older synthetic routes may involve the use of toxic heavy metals or generate significant amounts of hazardous waste, complicating environmental compliance and increasing disposal costs. These factors collectively contribute to higher production costs and longer lead times for high-purity pharmaceutical intermediates, creating bottlenecks in the supply chain for generic and innovative drug manufacturers alike.
The Novel Approach
The methodology described in CN110156761B overcomes these hurdles by employing a modular synthesis strategy centered around the robust Copper-catalyzed Azide-Alkyne Cycloaddition (CuAAC), commonly known as Click Chemistry. This approach allows for the rapid assembly of the triazole-linked coumarin-biphenyl core under mild conditions, typically at room temperature or with gentle heating, significantly reducing energy consumption. The use of readily available starting materials like chloromethyl coumarins and halogenated biphenyls streamlines the procurement process, ensuring cost reduction in antihypertensive drug manufacturing. Additionally, the reactions proceed with high atom economy and generate fewer by-products, simplifying downstream processing and improving the overall sustainability of the production lifecycle.
Mechanistic Insights into Click Chemistry and Nucleophilic Substitution
The core of this synthetic innovation lies in the precise orchestration of nucleophilic substitution and cycloaddition reactions to build the complex molecular architecture. The process initiates with the conversion of chloromethyl coumarins into azido intermediates using sodium azide, a transformation that proceeds efficiently in polar aprotic solvents like acetonitrile or DMF. Subsequently, the azido group undergoes a 1,3-dipolar cycloaddition with terminal alkynes in the presence of a Cu(I) catalyst system, such as copper sulfate paired with sodium ascorbate. This step is pivotal as it forms the 1,2,3-triazole ring, which serves as a stable and bioactive linker between the coumarin and the rest of the molecule, ensuring high regioselectivity for the 1,4-disubstituted isomer.
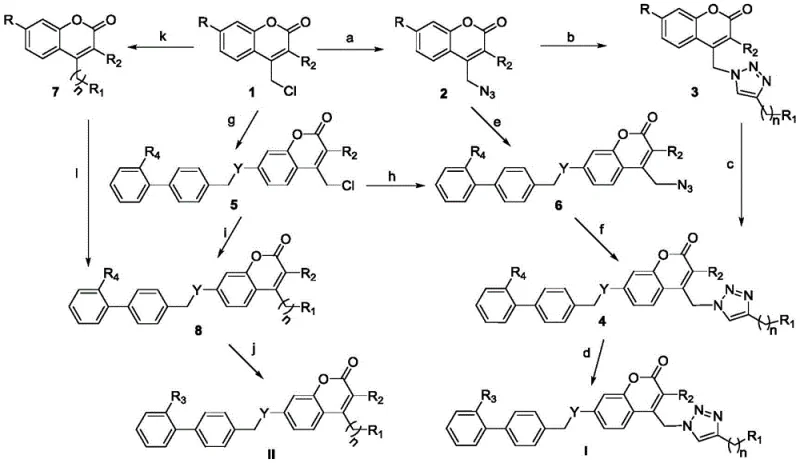
Following the formation of the triazole scaffold, the final assembly involves a nucleophilic substitution reaction where the phenolic hydroxyl or amino group on the coumarin moiety attacks a halogenated biphenyl derivative. This step connects the two major pharmacophores, establishing the essential biphenyl motif required for Ang II receptor antagonism. The reaction is facilitated by base catalysts like potassium carbonate in solvents such as acetone or THF, ensuring complete conversion without degrading sensitive functional groups. Finally, ester groups are hydrolyzed under basic conditions to reveal the free carboxylic acid, which is critical for the ionic interaction with the receptor. This mechanistic pathway ensures high purity and consistent quality, vital for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Coumarin-Biphenyl Intermediates Efficiently
The synthesis of these high-value intermediates requires strict adherence to the optimized reaction parameters outlined in the patent to ensure maximum yield and purity. The process involves distinct stages of azidation, click cyclization, and biphenyl coupling, each requiring specific control over temperature, stoichiometry, and workup procedures to minimize impurities. Detailed standard operating procedures for each transformation are essential for reproducibility, particularly when scaling from gram to kilogram quantities. For comprehensive technical guidance on reaction conditions and purification methods, please refer to the standardized synthesis protocol below.
- Preparation of azido-coumarin intermediates via nucleophilic substitution of chloromethyl coumarins with sodium azide.
- Execution of Copper-catalyzed Azide-Alkyne Cycloaddition (CuAAC) to introduce triazole moieties.
- Final coupling with halogenated biphenyl derivatives followed by hydrolysis to yield the target carboxylic acid compounds.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits regarding operational efficiency and risk mitigation. The reliance on commodity chemicals and widely available catalysts reduces dependency on specialized reagents that often suffer from supply volatility. This stability in raw material sourcing translates directly into enhanced supply chain reliability, ensuring consistent delivery schedules for downstream API production. Moreover, the simplicity of the reaction workups, which often involve standard extraction and crystallization techniques, minimizes the need for complex chromatographic separations on a large scale, thereby lowering operational expenditures.
- Cost Reduction in Manufacturing: The streamlined synthetic pathway eliminates the need for expensive transition metal catalysts often required in traditional cross-coupling reactions, leading to substantial cost savings in raw material procurement. By utilizing high-yielding steps that minimize waste generation, the overall process mass intensity is reduced, which lowers the cost of goods sold and improves profit margins for the final active pharmaceutical ingredient. The avoidance of cryogenic conditions or high-pressure equipment further reduces capital expenditure requirements for manufacturing facilities.
- Enhanced Supply Chain Reliability: The use of robust chemical transformations that are tolerant to minor variations in reaction conditions ensures consistent batch-to-batch quality, reducing the risk of production delays due to failed runs. Since the key starting materials are commercially accessible and not subject to strict regulatory controls like some precursors, the risk of supply disruption is significantly mitigated. This reliability allows for better inventory planning and reduces the need for excessive safety stock, optimizing working capital.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, having been demonstrated to work effectively from small laboratory scales to larger pilot batches without loss of efficiency. The reduced generation of hazardous by-products aligns with green chemistry principles, simplifying waste treatment processes and ensuring compliance with increasingly stringent environmental regulations. This environmental compatibility facilitates smoother regulatory approvals and enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new class of compounds is essential for making informed sourcing decisions. The following questions address common inquiries regarding the stability, activity, and production feasibility of these coumarin-biphenyl derivatives. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for your project needs.
Q: What is the primary advantage of the coumarin-biphenyl scaffold over traditional Sartans?
A: The coumarin-biphenyl scaffold combines the natural biological activity of coumarin with the proven Ang II receptor antagonism of biphenyl structures, offering potentially improved vasodilation and renal protection profiles compared to older generations.
Q: Is the synthesis route scalable for commercial production?
A: Yes, the patented method utilizes robust reactions like nucleophilic substitution and click chemistry which are well-suited for scale-up, avoiding extremely sensitive reagents and achieving total yields over 40%.
Q: What represents the key structural feature for activity in these compounds?
A: The presence of the triazole ring acts as a bioisostere with significant dipole moment and hydrogen bonding capability, facilitating better binding to AT1 receptors alongside the essential carboxyl or tetrazole acidic group.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Coumarin-Biphenyl Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the coumarin-biphenyl scaffold in the development of advanced antihypertensive therapies. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical candidate to market-ready drug is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch supports your critical research and development milestones with unwavering consistency and quality.
We invite you to collaborate with us to leverage this innovative technology for your pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your project timelines and optimize your budget for this promising class of pharmaceutical intermediates.
