Advanced Benzylic C-H Oxidation for Scalable Pharmaceutical Intermediate Manufacturing
Advanced Benzylic C-H Oxidation for Scalable Pharmaceutical Intermediate Manufacturing
The landscape of fine chemical synthesis is undergoing a significant transformation driven by the urgent need for greener, more sustainable manufacturing processes. A pivotal advancement in this domain is detailed in patent CN107602393B, which discloses a novel method for the direct oxidation of benzylic C-H bonds into valuable ketones or carboxylic acids. This technology represents a paradigm shift away from traditional, hazardous oxidation protocols, utilizing trichloroisocyanuric acid (TCCA) as a robust oxidant in conjunction with a catalytic system comprising sodium bromide and tetramethylpiperidine oxynitride (TEMPO). For R&D directors and procurement specialists alike, this innovation offers a compelling solution to the dual challenges of environmental compliance and cost efficiency. By operating under mild reaction conditions ranging from 0°C to 40°C, this method circumvents the safety risks and energy demands associated with high-temperature reflux or high-pressure systems, positioning it as an ideal candidate for the reliable production of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the selective oxidation of benzylic C-H bonds has relied heavily on stoichiometric amounts of aggressive oxidants such as chromium trioxide or expensive iodine reagents. These conventional pathways are fraught with significant drawbacks that hinder their viability in modern industrial settings. The use of chromium-based oxidants generates substantial quantities of toxic heavy metal waste, creating a severe environmental burden and necessitating costly disposal procedures to meet stringent regulatory standards. Furthermore, many traditional metal-catalyzed processes require harsh reaction conditions, including high pressures and elevated temperatures, which demand specialized, capital-intensive equipment and pose inherent safety hazards. The reliance on liquid oxidants like sodium hypochlorite also introduces logistical challenges due to their instability and tendency to decompose upon exposure to light, leading to inconsistent batch quality and supply chain vulnerabilities.
The Novel Approach
In stark contrast, the methodology outlined in CN107602393B leverages the stability and high oxidizing power of solid trichloroisocyanuric acid (TCCA), which contains over 90% available chlorine. This approach utilizes a biphasic solvent system of dichloromethane and water, facilitating efficient mass transfer while maintaining a mild thermal profile. The introduction of sodium bromide and TEMPO as catalysts allows for precise control over the oxidation state, enabling the selective formation of either ketones or carboxylic acids depending on the stoichiometric ratio of the oxidant. As illustrated in the conversion of ethylbenzene to acetophenone, this system achieves high yields without the need for toxic heavy metals. 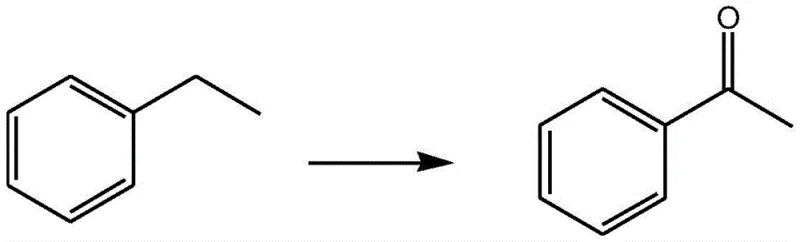
Mechanistic Insights into TCCA/TEMPO Catalytic Oxidation
The efficacy of this oxidation protocol lies in the synergistic interaction between the stable chlorinating agent TCCA and the nitroxyl radical catalyst TEMPO. Mechanistically, the sodium bromide serves as a co-catalyst, likely generating active bromine species in situ upon reaction with TCCA. These active species then interact with the TEMPO catalyst to form the active oxoammonium ion, which is the true oxidizing agent responsible for abstracting hydrogen from the benzylic position. This radical-mediated pathway is highly selective for benzylic C-H bonds due to their relatively lower bond dissociation energy compared to other aliphatic C-H bonds. The mild temperature range of 0-40°C is critical; it provides sufficient energy to drive the catalytic cycle while preventing the thermal decomposition of sensitive functional groups or the over-oxidation of the product, ensuring a clean impurity profile that is essential for pharmaceutical applications.
Furthermore, the biphasic nature of the reaction medium plays a crucial role in impurity control and product isolation. The use of dichloromethane and water allows for the effective partitioning of organic products and inorganic byproducts. The solid nature of TCCA ensures a controlled release of oxidizing equivalents, especially when added in portions as described in the patent examples. This controlled addition minimizes local hotspots of high oxidant concentration that could lead to side reactions or chlorination of the aromatic ring. The general scope of this transformation, capable of handling diverse substrates to yield either ketones or acids, underscores its versatility as a platform technology for synthesizing high-purity OLED materials or agrochemical intermediates. 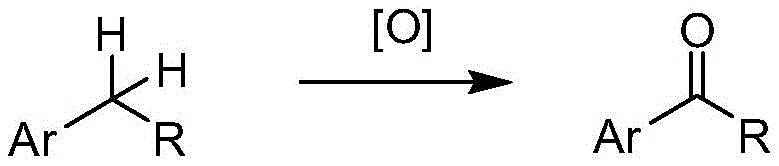
How to Synthesize Benzylic Ketones and Acids Efficiently
Implementing this oxidation strategy requires careful attention to reagent stoichiometry and addition rates to maximize yield and safety. The patent provides a robust framework for scaling this reaction, emphasizing the portion-wise addition of the solid oxidant to manage exotherms effectively. The process is designed to be operationally simple, avoiding the need for inert atmosphere techniques or anhydrous conditions, which significantly lowers the barrier to entry for commercial adoption. For detailed operational parameters, the standardized synthesis steps below outline the precise molar ratios and temperature controls necessary to replicate the high efficiencies reported in the patent data, ensuring a smooth transition from laboratory bench to pilot plant.
- Prepare the reaction mixture by combining the benzylic substrate, dichloromethane, water, sodium bromide (catalyst), and tetramethylpiperidine oxynitride (TEMPO catalyst) in a reactor.
- Heat the mixture to a mild temperature range of 0-40°C, preferably 40°C, to initiate the catalytic cycle without requiring harsh thermal conditions.
- Add trichloroisocyanuric acid (TCCA) in portions (typically 4 portions) to control the exotherm and ensure complete oxidation to the target ketone or carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this TCCA-mediated oxidation protocol offers profound strategic advantages over legacy technologies. The primary benefit stems from the replacement of hazardous, liquid oxidants with stable, solid reagents that are significantly easier to transport and store. This shift mitigates the risks associated with the degradation of oxidizing agents during transit, ensuring consistent raw material quality and reducing the frequency of supply disruptions. Moreover, the elimination of heavy metal catalysts removes the need for expensive and time-consuming metal scavenging steps during downstream processing. This simplification of the purification workflow translates directly into reduced manufacturing cycles and lower operational expenditures, making it a highly attractive option for cost reduction in fine chemical manufacturing.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the use of commodity-grade reagents like TCCA and sodium bromide, which are vastly cheaper than specialized metal catalysts or stoichiometric iodine reagents. By avoiding the use of chromium or other heavy metals, manufacturers eliminate the substantial costs associated with hazardous waste disposal and environmental remediation. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, further contributing to substantial cost savings in utility expenses. The high atom economy and selectivity of the reaction minimize the formation of byproducts, thereby increasing the overall yield of the target API intermediate and reducing the cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: The stability of the key reagents ensures a more resilient supply chain. Unlike liquid bleach which degrades over time, solid TCCA maintains its potency during long-term storage, allowing for strategic stockpiling without the risk of potency loss. This reliability is critical for maintaining continuous production schedules and meeting tight delivery deadlines for global clients. The simplicity of the workup procedure, involving standard extraction and crystallization, reduces the dependency on specialized equipment or scarce skilled labor, thereby enhancing the overall robustness of the manufacturing line against operational bottlenecks.
- Scalability and Environmental Compliance: Scaling this reaction from grams to tons is facilitated by the absence of high-pressure requirements and the use of common solvents. The process aligns perfectly with green chemistry principles by avoiding toxic heavy metals, thus simplifying the regulatory approval process for new drug filings. The reduction in hazardous waste generation not only lowers disposal costs but also enhances the corporate sustainability profile of the manufacturer. This environmental compliance is increasingly becoming a prerequisite for partnerships with major multinational pharmaceutical companies who prioritize green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzylic oxidation technology. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, offering a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using TCCA over traditional chromium oxidants?
A: TCCA is a solid, stable oxidant with over 90% available chlorine, eliminating the transportation and storage hazards associated with liquid hypochlorites. Crucially, it avoids the generation of toxic heavy metal waste (like chromium salts), significantly reducing environmental compliance costs and downstream purification burdens.
Q: Can this method selectively produce both ketones and carboxylic acids?
A: Yes, the patent demonstrates high versatility. By adjusting the stoichiometry of the oxidant (TCCA) relative to the substrate, the reaction can be tuned to stop at the ketone stage (e.g., ethylbenzene to acetophenone) or proceed to full oxidation to the carboxylic acid (e.g., o-nitrotoluene to o-nitrobenzoic acid).
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The reaction operates under mild conditions (0-40°C) and atmospheric pressure, removing the need for specialized high-pressure equipment. The use of inexpensive, solid reagents and a simple biphasic workup makes it highly scalable for commercial manufacturing of fine chemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzylic Oxidation Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the TCCA/TEMPO oxidation system for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of benzylic ketone or acid meets the exacting standards required for drug substance manufacturing. We are committed to delivering not just a product, but a optimized supply solution that balances quality, cost, and speed.
We invite you to leverage our technical expertise to evaluate the feasibility of this green oxidation route for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this metal-free protocol. We encourage you to contact us today to discuss your requirements, obtain specific COA data for similar compounds, and receive comprehensive route feasibility assessments tailored to your commercial goals.
