Scalable Synthesis of Chiral 4-Halogenated Tryptophan Derivatives via Rh-Catalyzed Asymmetric Hydrogenation
The pharmaceutical industry's relentless pursuit of novel therapeutic agents has placed a premium on high-purity chiral building blocks, particularly those containing indole scaffolds which are ubiquitous in bioactive natural products. Patent CN112409235A, published in early 2021, introduces a groundbreaking synthesis method for chiral 4-halogenated tryptophan derivatives, addressing critical bottlenecks in the production of complex indole alkaloids. This technology represents a significant leap forward for manufacturers of pharmaceutical intermediates, offering a pathway to produce optically pure compounds that serve as essential precursors for drugs treating cardiovascular and cerebrovascular diseases. By leveraging a highly efficient rhodium-catalyzed asymmetric hydrogenation strategy, this patent outlines a process that not only simplifies the synthetic route but also drastically enhances the stereochemical purity of the final product, making it an invaluable asset for R&D teams focused on next-generation drug discovery.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of optically pure 4-halogenated tryptophan derivatives has been plagued by inefficient and costly methodologies that struggle to meet the rigorous demands of modern GMP manufacturing. Prior art often relied on palladium-mediated oxidative coupling reactions which necessitated the use of stoichiometric amounts of expensive palladium salts, leading to substantial metal waste and challenging downstream purification processes to remove residual heavy metals. Furthermore, alternative routes involving enzymatic kinetic resolution suffered from inherent theoretical yield limitations of 50%, while methods utilizing chiral auxiliaries introduced additional synthetic steps for installation and removal, thereby inflating both material costs and production timelines. These conventional approaches frequently resulted in moderate enantioselectivities, often capping at around 94% ee, which is insufficient for many high-value API applications requiring near-perfect optical purity.
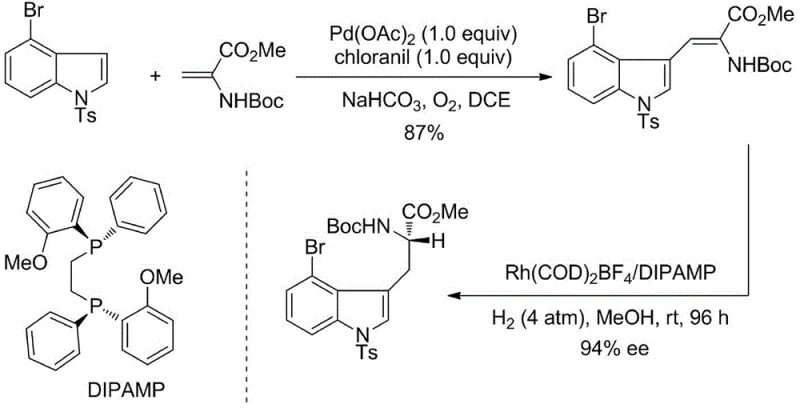
The Novel Approach
In stark contrast to these legacy methods, the invention disclosed in CN112409235A presents a streamlined three-step sequence that bypasses the need for stoichiometric metals or chiral auxiliaries. The process initiates with a condensation reaction to form an oxazolone intermediate, followed by a mild alcoholysis ring-opening to generate a dehydrotryptophan precursor. The cornerstone of this innovation is the final catalytic asymmetric hydrogenation step, which utilizes a specialized rhodium complex to install the chiral center with unprecedented precision. This novel route eliminates the cumbersome purification steps associated with older technologies, allowing for the direct isolation of the product via simple recrystallization. By shifting from stoichiometric reagents to a catalytic system, the method significantly reduces the environmental footprint and operational complexity, establishing a new benchmark for cost reduction in pharmaceutical intermediate manufacturing.
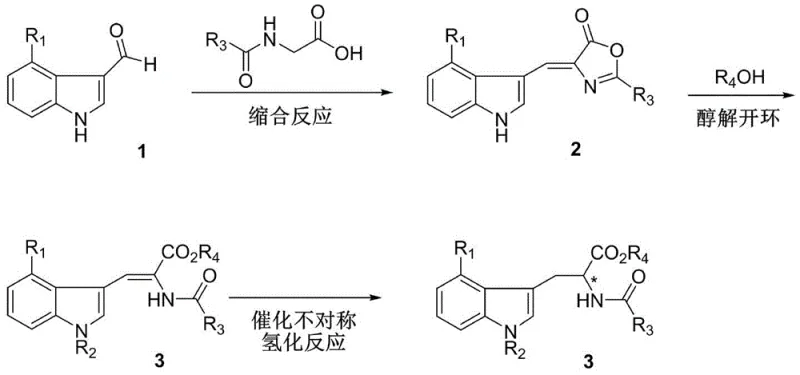
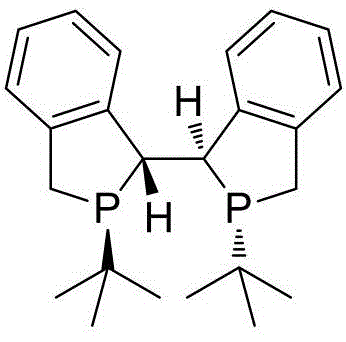
Mechanistic Insights into Rh-Catalyzed Asymmetric Hydrogenation
The exceptional performance of this synthesis hinges on the precise coordination between the rhodium catalyst and the chiral diphosphine ligand, specifically the (R,R,S,S)-Duanphos ligand. During the hydrogenation cycle, the rhodium center, typically introduced as Rh(NBD)2BF4, coordinates with the Duanphos ligand to form a chiral pocket that dictates the facial selectivity of hydrogen addition to the dehydrotryptophan double bond. The unique electronic and steric properties of the Duanphos ligand ensure that the substrate binds in a specific orientation, effectively shielding one face of the molecule and forcing hydrogen delivery to the other, which results in the observed 99.9% enantiomeric excess. This level of stereocontrol is critical for ensuring the biological efficacy of the downstream drug molecules, as even minor impurities of the wrong enantiomer can lead to toxicity or reduced potency.
Furthermore, the robustness of this catalytic system contributes significantly to impurity control throughout the manufacturing process. Unlike enzymatic resolutions which can be sensitive to trace contaminants or pH fluctuations, this metal-catalyzed hydrogenation operates under controlled conditions of 50 bar hydrogen pressure and 50°C, providing a stable environment that minimizes side reactions such as over-reduction or racemization. The use of methanol as a solvent further aids in maintaining the solubility of intermediates while facilitating the final crystallization of the product. For procurement managers and quality assurance teams, this mechanistic reliability translates to a consistent impurity profile and a simplified analytical workflow, ensuring that every batch meets the stringent specifications required for clinical-grade materials without the need for extensive reprocessing.
How to Synthesize Chiral 4-Halogenated Tryptophan Derivatives Efficiently
The practical implementation of this patented technology involves a straightforward sequence that is highly amenable to scale-up in standard chemical reactors. The process begins with the condensation of a 4-haloindole-3-carbaldehyde with an N-acyl glycine derivative in the presence of acetic anhydride and sodium acetate, followed by a ring-opening step using sodium methoxide in methanol. The resulting dehydrotryptophan ester is then subjected to the key hydrogenation step using the rhodium-Duanphos catalyst system. This methodology has been validated to produce high yields of up to 95% with exceptional optical purity, demonstrating its viability for industrial application. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined in the guide below to assist technical teams in replicating this high-efficiency route.
- Perform a condensation reaction between a 4-haloindole-3-carbaldehyde and an N-acyl glycine derivative using acetic anhydride and sodium acetate to form an oxazolone intermediate.
- Execute an alcoholysis ring-opening of the oxazolone intermediate using an alcohol and a metal alkoxide to generate the dehydrotryptophan precursor.
- Conduct asymmetric hydrogenation of the precursor using a Rhodium-Duanphos catalyst complex under hydrogen pressure to yield the final chiral product with high enantioselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this synthesis method offers transformative benefits that extend far beyond simple yield improvements. By replacing stoichiometric palladium reagents with a low-loading rhodium catalyst system, the process fundamentally alters the cost structure of production, removing the burden of expensive metal recovery and disposal. The elimination of column chromatography in favor of direct recrystallization not only accelerates the production cycle but also drastically reduces solvent consumption and waste generation, aligning perfectly with modern green chemistry initiatives. These operational efficiencies translate into a more resilient supply chain capable of delivering high-quality materials with greater consistency and reduced lead times.
- Cost Reduction in Manufacturing: The transition from stoichiometric metal mediators to a catalytic hydrogenation process inherently lowers the raw material costs per kilogram of product. By avoiding the use of large quantities of palladium and chiral auxiliaries, manufacturers can achieve substantial savings on input costs while simultaneously reducing the expenses associated with hazardous waste treatment. The simplified purification protocol further drives down operational expenditures by minimizing labor hours and solvent usage, creating a leaner and more economically viable production model for high-value chiral intermediates.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials, such as 4-bromoindole-3-carbaldehyde and common acyl glycines, ensures a stable supply base that is less susceptible to market volatility compared to specialized chiral pools. The robustness of the catalytic system allows for flexible manufacturing schedules, as the reaction is not constrained by the delicate handling requirements often associated with enzymatic or air-sensitive organometallic processes. This stability empowers supply chain heads to maintain continuous production flows, mitigating the risk of stockouts and ensuring timely delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of toxic heavy metal waste streams make this process exceptionally suitable for large-scale commercialization. The ability to isolate the product through crystallization rather than chromatographic separation significantly eases the burden on wastewater treatment facilities and reduces the overall environmental footprint of the manufacturing site. This alignment with strict environmental regulations facilitates smoother regulatory approvals and supports the long-term sustainability goals of global pharmaceutical partners seeking eco-friendly sourcing options.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational parameters and expected outcomes for potential adopters of this method.
Q: What enantiomeric excess (ee) can be achieved with this synthesis method?
A: The patented method utilizing a Rh(NBD)2BF4 catalyst with (R,R,S,S)-Duanphos ligand achieves an exceptional enantiomeric excess of up to 99.9% ee, significantly surpassing previous methods that typically capped at 94%.
Q: Does this process require complex purification techniques like column chromatography?
A: No, one of the key commercial advantages of this route is that the final product can be isolated simply through recrystallization from methanol, eliminating the need for costly and time-consuming column chromatography separation.
Q: What are the specific reaction conditions for the key hydrogenation step?
A: The asymmetric hydrogenation is conducted at a moderate temperature of 50°C under a hydrogen pressure of 50 bar, using methanol as the solvent, which balances reaction efficiency with operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 4-Halogenated Tryptophan Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity chiral intermediates play in the development of life-saving medications. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN112409235A can be seamlessly translated from the laboratory to the manufacturing plant. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced hydrogenation reactors capable of handling high-pressure processes safely, guaranteeing that every batch of chiral 4-halogenated tryptophan derivative meets stringent purity specifications and regulatory standards.
We invite global pharmaceutical partners to collaborate with us to leverage this cutting-edge synthesis technology for your pipeline projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this efficient route can optimize your overall COGS. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply of these vital building blocks for your future success.
