Scalable Production of High-Performance Aggregation-Induced Emission Compounds for Advanced Optical Applications
Scalable Production of High-Performance Aggregation-Induced Emission Compounds for Advanced Optical Applications
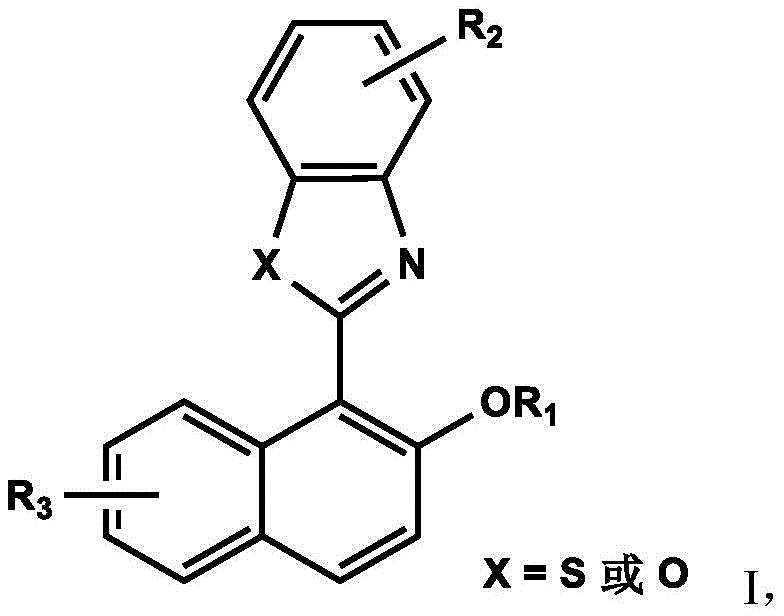
The rapid evolution of optoelectronic materials and biological imaging technologies has created an urgent demand for fluorescent compounds that defy the limitations of traditional organic emitters. Patent CN112480025B introduces a groundbreaking class of naphthalene-fused heterocyclic compounds exhibiting robust Aggregation-Induced Emission (AIE) properties. Unlike conventional fluorophores that suffer from concentration quenching, these novel structures demonstrate significantly enhanced luminescence in aggregated states, addressing a critical bottleneck in OLED efficiency and bio-probe sensitivity. The disclosed synthesis pathway leverages a streamlined two-step protocol involving Schiff base condensation followed by oxidative cyclization, offering a viable route for high-purity manufacturing. As a leading chemical partner, we recognize the immense potential of this technology to revolutionize sectors ranging from display manufacturing to clinical diagnostics, providing a stable supply of next-generation optical materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional organic fluorescent materials have long been plagued by the Aggregation-Caused Quenching (ACQ) effect, where emission intensity drastically diminishes at high concentrations or in solid films due to detrimental pi-pi stacking interactions. This phenomenon severely restricts their utility in solid-state devices like Organic Light-Emitting Diodes (OLEDs) and limits the concentration of probes usable in biological assays without signal loss. Furthermore, existing biological markers such as Green Fluorescent Protein (GFP) often exhibit poor photostability, small Stokes shifts, and susceptibility to enzymatic degradation, complicating long-term cell tracking and in vivo imaging. The reliance on complex transfection processes for protein-based markers also introduces significant time delays and potential interference with normal cellular functions, creating a pressing need for small-molecule alternatives that offer superior stability and ease of use.
The Novel Approach
The methodology outlined in the patent data presents a transformative solution by synthesizing compounds that inherently possess AIE characteristics through a restricted intramolecular motion mechanism. By fusing a naphthalene ring with a benzothiazole or benzoxazole moiety, the resulting molecular architecture prevents non-radiative decay pathways in the aggregated state, thereby turning aggregation into an asset rather than a liability. This approach not only eliminates the need for complex bulky groups often used to prevent stacking but also simplifies the molecular design, allowing for easier functionalization. The synthesis avoids harsh conditions and expensive noble metal catalysts, utilizing mild oxidative cyclization to construct the rigid planar core essential for efficient fluorescence. This strategic shift enables the production of materials that maintain high quantum yields in solid films and aqueous environments, directly addressing the efficiency roll-off issues prevalent in current organic emitter technologies.
Mechanistic Insights into Oxidative Cyclization and ESIPT Effects
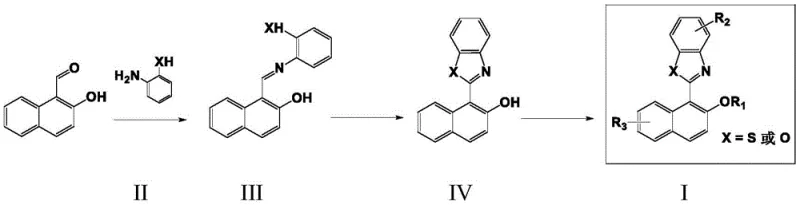
The core of this synthetic strategy lies in the precise construction of the heterocyclic ring system via an oxidative dehydrogenation mechanism. The process initiates with the condensation of 2-hydroxy-1-naphthaldehyde and an ortho-substituted aniline derivative, such as 2-aminothiophenol or 2-aminophenol, to form a Schiff base intermediate. This intermediate is then subjected to oxidative cyclization using reagents like 2,3-dichloro-5,6-dicyano-p-benzoquinone (DDQ) or 2-iodoxybenzoic acid (IBX). The oxidant facilitates the removal of hydrogen atoms, closing the ring to form the stable benzothiazole or benzoxazole structure fused to the naphthalene backbone. This rigidification is crucial as it restricts the intramolecular rotation of the aromatic rings, which is the fundamental physical basis for the observed AIE phenomenon, ensuring that energy is released as photons rather than heat.
Beyond the structural rigidity, the photophysical performance is governed by the Excited State Intramolecular Proton Transfer (ESIPT) effect inherent in the molecular design. A weak intramolecular hydrogen bond forms between the hydroxyl hydrogen atom on the naphthalene ring and the nitrogen atom on the adjacent heterocyclic ring. Upon excitation, this proton transfers to the nitrogen, creating a tautomer that emits fluorescence at a significantly red-shifted wavelength compared to the absorption band. This mechanism results in a large Stokes shift, which is highly desirable for imaging applications as it minimizes self-absorption and background interference. The ability to tune this effect by modifying the substituents on the naphthalene or benzene rings allows for the customization of emission colors, making these compounds versatile candidates for full-color display technologies and multiplexed biological sensing.
How to Synthesize Naphthalene-Based AIE Compounds Efficiently
The preparation of these high-value fluorescent materials follows a robust and reproducible protocol designed for scalability. The process begins with the reflux of stoichiometric amounts of aldehyde and amine precursors in alcohol solvents, followed by a critical purification step to ensure the purity of the Schiff base intermediate before oxidation. The subsequent oxidative cyclization is performed in non-polar solvents like toluene or xylene, where temperature control and oxidant selection play pivotal roles in maximizing yield. For detailed operational parameters, safety guidelines, and specific workup procedures required to achieve pharmaceutical-grade purity, please refer to the standardized synthesis guide below.
- Condense 2-hydroxy-1-naphthaldehyde with 2-aminothiophenol or 2-aminophenol in ethanol under reflux to form the Schiff base intermediate.
- Purify the intermediate via recrystallization and subject it to oxidative cyclization using DDQ or IBX in toluene.
- Isolate the final AIE compound through silica gel chromatography and optional derivatization for specific probe applications.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits over traditional fluorophore manufacturing. The elimination of transition metal catalysts such as palladium or copper removes the need for costly and time-consuming metal scavenging steps, which are often required to meet strict residual metal specifications in electronic and pharmaceutical grades. This simplification of the downstream processing significantly reduces the overall production cycle time and lowers the consumption of specialized purification resins. Furthermore, the use of commodity chemicals like naphthaldehyde and aminothiophenol ensures a stable and resilient supply chain, mitigating risks associated with the scarcity of exotic reagents. The high thermal and photo-stability of the final product also translates to extended shelf life and reduced waste during storage and transportation, enhancing overall inventory management efficiency.
- Cost Reduction in Manufacturing: The synthetic pathway is designed to minimize operational expenditures by utilizing inexpensive oxidants like DDQ and common organic solvents that can be easily recovered and recycled. By avoiding the use of precious metal catalysts, the process eliminates a major cost driver associated with catalyst loading and recovery, leading to a leaner cost structure. Additionally, the high conversion rates and straightforward isolation methods reduce the volume of solvent waste generated per kilogram of product, lowering disposal costs and environmental compliance burdens. This economic efficiency makes the commercial production of high-performance AIE materials financially viable for mass-market applications in consumer electronics and diagnostic kits.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals ensures that production schedules are not disrupted by the volatility of niche reagent markets. The robustness of the reaction conditions, which tolerate mild heating and standard atmospheric pressure, allows for manufacturing in versatile facilities without the need for specialized high-pressure or cryogenic equipment. This flexibility enables rapid scale-up from pilot batches to multi-ton annual production capacities, ensuring consistent availability for long-term contracts. The stability of the intermediates also allows for stockpiling, providing a buffer against sudden spikes in demand and ensuring business continuity for downstream clients.
- Scalability and Environmental Compliance: The process generates minimal hazardous byproducts, primarily consisting of reduced forms of the oxidant which are manageable through standard waste treatment protocols. The absence of heavy metals simplifies the environmental impact assessment and facilitates easier regulatory approval for use in sensitive applications like in vivo imaging. The high atom economy of the cyclization step ensures that a significant portion of the reactant mass is incorporated into the final product, aligning with green chemistry principles. This environmental compatibility not only reduces regulatory risk but also enhances the brand value of end-products marketed as sustainable and eco-friendly.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these advanced AIE compounds. Our answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for integrating these materials into your specific R&D pipelines or procurement strategies effectively.
Q: What is the primary advantage of this AIE compound over traditional fluorophores?
A: Unlike traditional fluorophores that suffer from Aggregation-Caused Quenching (ACQ), this compound exhibits enhanced emission in the aggregated state, making it ideal for solid-state OLEDs and high-concentration biological imaging.
Q: Can this synthesis route be scaled for industrial production?
A: Yes, the process utilizes common solvents like ethanol and toluene and avoids expensive transition metal catalysts, facilitating straightforward scale-up from laboratory to multi-ton production.
Q: What are the key structural features enabling the large Stokes shift?
A: The molecule features an intramolecular hydrogen bond between the naphthalene hydroxyl group and the heterocyclic nitrogen, enabling Excited State Intramolecular Proton Transfer (ESIPT) which results in a large Stokes shift.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aggregation-Induced Emission Compound Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market deployment is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including detailed analysis of impurity profiles and residual solvent levels. We understand that consistency is key in the electronic and pharmaceutical industries, and our dedicated process engineering team works closely with clients to optimize reaction parameters for maximum yield and reproducibility. Whether you require custom derivatives for specific wavelength emissions or bulk quantities for OLED panel manufacturing, our infrastructure is ready to support your growth.
We invite you to engage with our technical procurement team to discuss your specific requirements and explore how our manufacturing capabilities can enhance your supply chain resilience. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized synthesis routes can reduce your overall material costs. We encourage potential partners to contact us for specific COA data and route feasibility assessments to validate the suitability of our AIE compounds for your next-generation optical applications.
