Advanced Synthesis of Oxygen-Substituted Benzocycloheptenes for Commercial Scale-Up
Advanced Synthesis of Oxygen-Substituted Benzocycloheptenes for Commercial Scale-Up
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing complex active pharmaceutical ingredients (APIs) and their precursors. Patent CN100390143C introduces a groundbreaking methodology for the synthesis of oxygen-substituted benzocycloheptenes, which serve as critical intermediates in the preparation of tissue-selective estrogens. This innovation addresses the longstanding challenges associated with the production of Selective Estrogen Receptor Modulators (SERMs), such as raloxifene analogs, by drastically simplifying the synthetic route. Unlike previous methods that relied heavily on tedious purification techniques, this novel approach leverages cost-effective starting materials and robust reaction conditions to achieve high yields and exceptional purity. For R&D directors and procurement managers, this represents a pivotal shift towards more sustainable and economically viable manufacturing processes. The ability to produce these high-value intermediates without recourse to chromatographic purification steps is a significant technical breakthrough that directly impacts the bottom line.
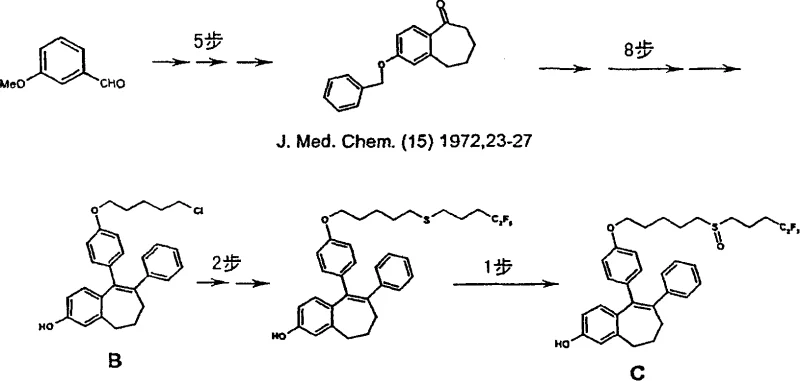
The limitations of conventional methods for synthesizing benzocycloheptene derivatives have long hindered large-scale production. As detailed in prior art such as WO 00/03979, the traditional synthesis of compounds like Formula A involves a cumbersome 16-step sequence. A major bottleneck in these legacy processes is the reliance on multiple chromatographic purification steps, which are not only time-consuming but also result in substantial material loss. The overall yield for these conventional routes is often dismally low, frequently falling below 6%. Furthermore, the complexity of the route makes scaling up to produce hundreds of kilograms of the final compound extremely difficult and risky. The presence of multiple purification stages increases the operational expenditure and extends the lead time, creating supply chain vulnerabilities for downstream drug manufacturers who require consistent, high-volume supplies of these critical intermediates.
In stark contrast, the novel approach described in CN100390143C offers a streamlined pathway that reduces the number of isolated intermediate steps to just 8. This reduction is achieved through the implementation of several one-pot reactions and the strategic selection of reagents that facilitate easier workup procedures. Most notably, the new method completely eliminates the need for chromatographic purification, relying instead on crystallization and extraction techniques that are far more amenable to industrial scale-up. The overall yield has been dramatically improved to approximately 36%, representing a six-fold increase over the prior art. This efficiency gain is not merely a laboratory curiosity but a fundamental restructuring of the synthesis logic, enabling the production of high-purity intermediates suitable for direct use in subsequent API manufacturing steps without further extensive cleaning.
Mechanistic Insights into Friedel-Crafts Cyclization and Suzuki Coupling
A cornerstone of this improved synthesis is the efficient construction of the benzocycloheptene core via a Friedel-Crafts ring closure. The process utilizes polyphosphoric acid (PPA) at elevated temperatures ranging from 80°C to 120°C to cyclize the pentanoic acid precursor. This step is critical for establishing the seven-membered ring structure characteristic of the target molecule. The use of PPA is particularly advantageous as it acts both as a solvent and a dehydrating agent, driving the equilibrium towards the desired ketone product. Following the cyclization, the resulting 7-methoxy-2-phenyl-1-benzocycloheptanone is converted into an enol nonafluorosulfonate. This activation strategy is superior to traditional halogenation methods because the nonafluorosulfonate group serves as an excellent leaving group for palladium-catalyzed cross-coupling reactions, offering enhanced stability and reactivity profiles.
The subsequent Suzuki coupling reaction allows for the introduction of diverse aromatic substituents, providing flexibility in the synthesis of various analogs. By reacting the enol nonafluorosulfonate with functionalized phenylboronic acids in the presence of palladium catalysts such as Pd(PPh3)4, the biaryl bond is formed with high fidelity. Another key mechanistic innovation is the selective demethylation of the aromatic methyl ether. Traditionally, removing a methyl ether in the presence of other sensitive functional groups can be challenging. However, this patent discloses the use of boron tribromide (BBr3) in combination with 2,6-lutidine. The addition of 2,6-lutidine is crucial; it modulates the Lewis acidity of the boron species, allowing for the selective cleavage of the aromatic methyl ether while leaving higher alkyl ethers intact. This selectivity ensures that the final phenolic product is obtained in high purity without the formation of side products that would otherwise complicate purification.
How to Synthesize Benzocycloheptene Intermediates Efficiently
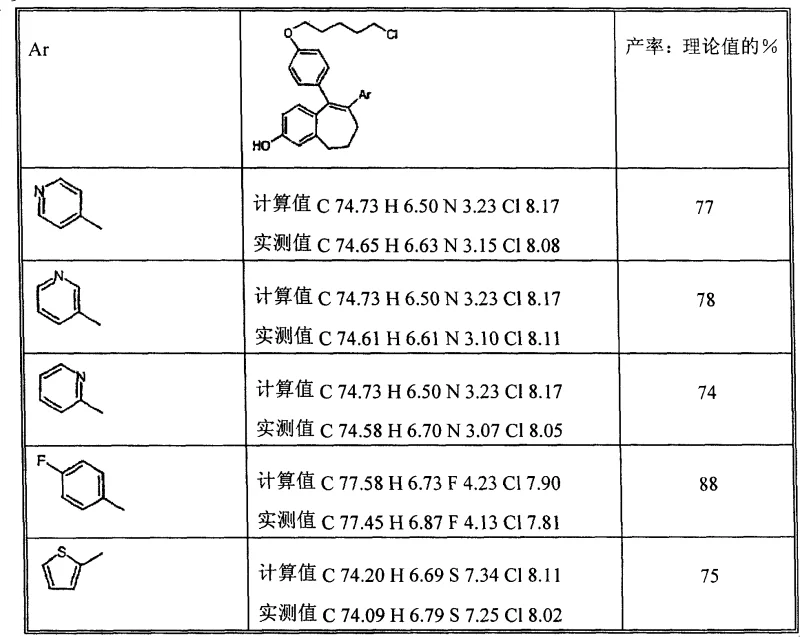
The synthesis of these valuable pharmaceutical intermediates begins with the preparation of a dienoic acid precursor through a condensation reaction involving 3-methoxybenzaldehyde and arylacetic acid. This initial step sets the stage for the subsequent ring-closing operations. The process is designed to be robust, tolerating variations in reaction conditions while maintaining high conversion rates. Following the formation of the carbon skeleton, the material undergoes hydrogenation and cyclization to form the core structure. The detailed operational parameters, including specific stoichiometry, temperature controls, and workup procedures, are critical for achieving the reported yields and purity levels. For process chemists looking to implement this route, understanding the nuances of the one-pot sequences is essential for maximizing throughput.
- Perform a one-pot condensation of 3-methoxybenzaldehyde with acetaldehyde and arylacetic acid to form the dienoic acid precursor.
- Execute catalytic hydrogenation followed by Friedel-Crafts cyclization using polyphosphoric acid at 80-120°C to form the benzocycloheptanone core.
- Convert the ketone to a nonafluorosulfonate ester and perform Suzuki coupling with a functionalized phenylboronic acid.
- Conduct selective demethylation using boron tribromide and 2,6-lutidine to yield the final phenolic intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers tangible benefits that extend beyond simple yield improvements. The elimination of chromatographic purification is perhaps the most significant cost driver reduction. Chromatography on an industrial scale requires expensive stationary phases, large volumes of solvents, and specialized equipment, all of which contribute heavily to the cost of goods sold (COGS). By replacing these steps with crystallization and extraction, the manufacturing process becomes significantly more cost-effective and environmentally friendly due to reduced solvent waste. Furthermore, the reduction in the total number of steps from 16 to 8 inherently reduces the cumulative processing time and labor costs associated with each batch. This streamlining allows for faster turnover times and increased production capacity within existing facilities.
- Cost Reduction in Manufacturing: The primary economic advantage lies in the drastic simplification of the purification workflow. Removing chromatography not only saves on direct material costs but also reduces the energy consumption associated with solvent recovery and evaporation. The use of commercially available starting materials, such as 3-methoxybenzaldehyde and standard arylacetic acids, ensures that raw material costs remain stable and predictable. Additionally, the high overall yield of 36% means that less starting material is required to produce the same amount of final product, effectively lowering the unit cost of the intermediate. This efficiency makes the final API more competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the chemical transformations described. The reactions utilize reagents and catalysts that are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. The ability to produce intermediates with high purity without complex purification means that quality control failures are less likely to occur, ensuring a consistent flow of material to downstream customers. The process is designed to be scalable from kilogram to multi-ton quantities, providing the flexibility needed to respond to fluctuating market demands without compromising on delivery schedules or product quality specifications.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process aligns well with green chemistry principles. The reduction in solvent usage and the avoidance of silica gel waste from chromatography columns significantly lower the environmental footprint of the manufacturing process. This is increasingly important as regulatory bodies impose stricter limits on pharmaceutical waste disposal. The use of polyphosphoric acid and standard palladium catalysts allows for established waste treatment protocols to be employed. The scalability of the Friedel-Crafts and Suzuki coupling steps has been demonstrated to be effective for industrial production, ensuring that the technology can be transferred from the lab to the plant with minimal technical risk.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of benzocycloheptene intermediates. These answers are derived directly from the experimental data and process descriptions found in the patent literature. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this new manufacturing route. The focus is on practical implementation details, purity profiles, and the specific advantages over legacy methods.
Q: How does this new process improve upon the conventional WO 00/03979 method?
A: The novel process reduces the total step count from 16 to just 8 isolated steps and completely eliminates the need for chromatographic purification, resulting in a significant increase in overall yield from less than 6% to approximately 36%.
Q: What are the key conditions for the selective demethylation step?
A: The selective removal of the aromatic methyl ether is achieved using boron tribromide (BBr3) in combination with 2,6-lutidine at temperatures between -30°C and 50°C, ensuring high purity without affecting other ether groups.
Q: Is this synthesis suitable for industrial scale production?
A: Yes, the method is specifically designed for industrial application, utilizing cost-effective starting materials and avoiding complex purification techniques like chromatography, which facilitates scaling to hundreds of kilograms.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzocycloheptene Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the modern pharmaceutical landscape. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We are committed to delivering high-purity pharmaceutical intermediates that adhere to stringent purity specifications and rigorous QC labs standards. By leveraging advanced technologies like the one described in CN100390143C, we can offer cost-effective solutions that enhance your supply chain stability. Our state-of-the-art facilities are equipped to handle complex organic syntheses, including palladium-catalyzed couplings and sensitive demethylation reactions, guaranteeing consistent quality batch after batch.
We invite you to collaborate with us to optimize your supply chain for tissue-selective estrogen intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments. Let us help you reduce lead time for high-purity pharmaceutical intermediates and secure a competitive advantage in the global market through our reliable partnership and technical expertise.
