Advanced Synthesis of Near-Infrared Phenazine Fluorescent Dyes for Commercial Scale-Up
Advanced Synthesis of Near-Infrared Phenazine Fluorescent Dyes for Commercial Scale-Up
The landscape of functional organic materials is undergoing a significant transformation with the advent of novel fluorophores designed for deep-tissue imaging and advanced optical applications. Patent CN108690032B introduces a groundbreaking class of phenazine condensed fluorescent dyes that address critical limitations in current bio-imaging technologies. Unlike traditional visible-light dyes, these compounds offer emission wavelengths in the near-infrared region, specifically ranging from 635nm to 829nm, which allows for deeper tissue penetration and reduced background interference. The structural versatility of these dyes, encompassing four distinct scaffolds labeled I through IV, provides a robust platform for tuning optical properties to specific application needs. 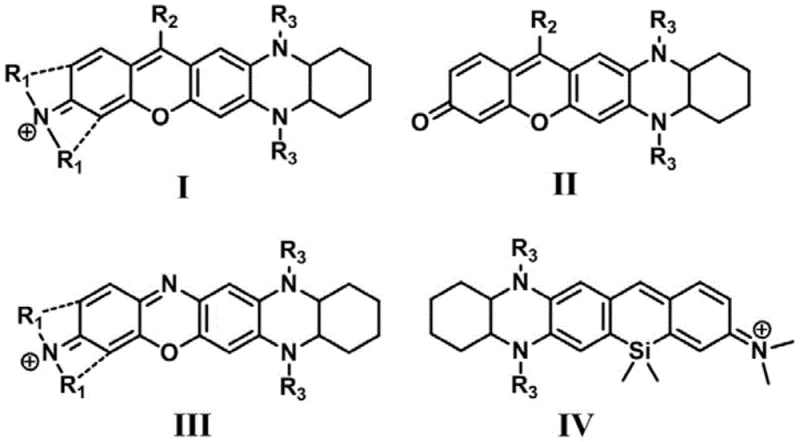 As a reliable fluorescent dye supplier, understanding the underlying chemistry of these structures is paramount for delivering high-purity materials that meet the rigorous demands of the pharmaceutical and electronic chemical sectors.
As a reliable fluorescent dye supplier, understanding the underlying chemistry of these structures is paramount for delivering high-purity materials that meet the rigorous demands of the pharmaceutical and electronic chemical sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional fluorescent probes, such as rhodamines, oxazines, and cyanine dyes, have long served as the standard for biological labeling; however, they suffer from inherent drawbacks that limit their efficacy in complex biological systems. A primary concern is their relatively small Stokes shift, which often leads to self-quenching and significant signal interference from excitation light, thereby reducing the signal-to-noise ratio in imaging applications. Furthermore, many classical dyes exhibit poor photostability, undergoing rapid bleaching under continuous laser irradiation, which compromises long-term observation studies. The synthesis of these conventional dyes often involves harsh conditions, expensive catalysts, or multi-step sequences that yield difficult-to-purify mixtures, creating bottlenecks in cost reduction in electronic chemical manufacturing and pharmaceutical intermediate production.
The Novel Approach
The innovative synthesis route disclosed in the patent overcomes these hurdles by leveraging a modular condensation strategy centered on a phenazine core. This approach utilizes accessible precursors like substituted o-phenylenediamines and cyclohexanedione to build the foundational scaffold under mild thermal conditions. By introducing specific substituents and employing a final cyclization step with tailored electrophiles, the method achieves large Stokes shifts of up to 122nm and high fluorescence quantum yields reaching 0.43. This strategic design not only enhances the optical performance but also simplifies the purification process, as the reaction byproducts are minimal and easily separated. The ability to tune the emission wavelength across the near-infrared spectrum simply by varying the R-groups offers unparalleled flexibility for developing specialized probes without redesigning the entire synthetic pathway.
Mechanistic Insights into Phenazine Core Construction and Functionalization
The heart of this technology lies in the efficient construction of the octahydrophenazine intermediate, which serves as the nucleophilic partner in the final dye assembly. The process begins with the condensation of 4-substituted o-phenylenediamine with cyclohexanedione in ethanol, forming a tetrahydrophenazine ring system through a double imine formation followed by tautomerization. 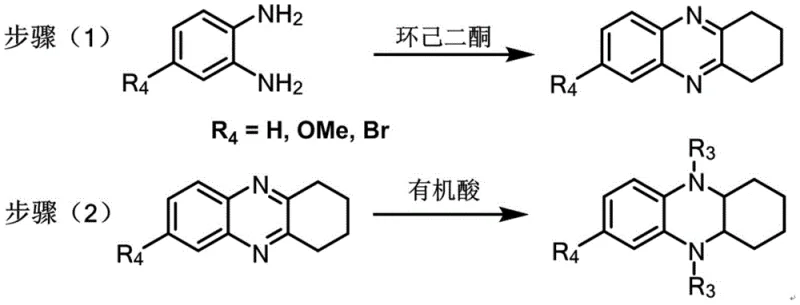 Subsequent reduction using sodium borohydride in the presence of an organic acid converts the aromatic character partially, yielding the 5,10-disubstituted-7-substituted-1,2,3,4,4a,5,10,10a-octahydrophenazine. This reduction step is critical as it activates the nitrogen centers for the final coupling reaction while maintaining the structural integrity of the fused ring system. The use of methanesulfonic acid as both solvent and catalyst in the final cyclization promotes the electrophilic aromatic substitution or condensation required to close the xanthene-like or sila-xanthene-like bridges, depending on the target dye type.
Subsequent reduction using sodium borohydride in the presence of an organic acid converts the aromatic character partially, yielding the 5,10-disubstituted-7-substituted-1,2,3,4,4a,5,10,10a-octahydrophenazine. This reduction step is critical as it activates the nitrogen centers for the final coupling reaction while maintaining the structural integrity of the fused ring system. The use of methanesulfonic acid as both solvent and catalyst in the final cyclization promotes the electrophilic aromatic substitution or condensation required to close the xanthene-like or sila-xanthene-like bridges, depending on the target dye type.
Impurity control is meticulously managed through the selection of reaction conditions that favor thermodynamic stability of the desired product. For instance, the use of neutral alumina columns during the intermediate stage effectively removes polar byproducts and unreacted diamines before they can interfere with the final cyclization. In the case of the Type IV dye, which incorporates a silicon bridge, the mechanism involves a lithiation-silylation sequence that requires strict temperature control at -78°C to prevent side reactions such as polymerization or over-lithiation. This precision ensures that the final product possesses the intended conjugation length and electronic properties, resulting in the observed high photostability and specific emission profiles required for high-purity OLED material or bio-probe applications.
How to Synthesize Phenazine Fluorescent Dyes Efficiently
The synthesis of these advanced fluorophores is designed for scalability, moving seamlessly from gram-scale laboratory optimization to kilogram-level production. The protocol emphasizes the use of common industrial solvents like ethanol, toluene, and dichloromethane, which facilitates easy solvent recovery and waste management. The initial condensation step is robust, tolerating a range of substituents on the phenylenediamine ring, which allows for the generation of diverse libraries of dyes from a single set of operating parameters.  The final purification typically involves column chromatography or recrystallization, techniques that are well-established in commercial manufacturing environments. Detailed standardized synthesis steps see the guide below.
The final purification typically involves column chromatography or recrystallization, techniques that are well-established in commercial manufacturing environments. Detailed standardized synthesis steps see the guide below.
- Synthesize 7-substituted-1,2,3,4-tetrahydrophenazine by reacting 4-substituted o-phenylenediamine with cyclohexanedione in ethanol at 50-70°C.
- Reduce the tetrahydrophenazine intermediate using sodium borohydride and organic acid in toluene, followed by reflux to obtain the octahydrophenazine derivative.
- Perform the final condensation cyclization with specific substrates like 2-substituted ketone-4-disubstituted aminophenol or via lithiation-silylation for Type IV structures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel phenazine dye platform offers substantial strategic benefits beyond mere performance metrics. The reliance on commodity chemicals such as cyclohexanedione and substituted anilines mitigates the risk of supply chain disruptions often associated with exotic or proprietary reagents. This accessibility ensures a consistent flow of raw materials, reducing lead time for high-purity fluorescent dyes and enabling manufacturers to maintain steady production schedules even during market fluctuations. Furthermore, the elimination of precious metal catalysts in the primary bond-forming steps drastically lowers the cost of goods sold, making these high-performance dyes economically viable for widespread adoption in diagnostic kits and display technologies.
- Cost Reduction in Manufacturing: The synthetic route avoids the use of expensive transition metal catalysts like palladium or platinum, which are not only costly but also require complex removal processes to meet regulatory standards for biological applications. By utilizing organic acids and simple hydride reducers, the process significantly reduces the expense associated with catalyst procurement and heavy metal scavenging resins. Additionally, the high reaction yields and straightforward workup procedures minimize solvent consumption and energy usage, contributing to a leaner and more cost-effective manufacturing profile that enhances overall profit margins.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are bulk chemicals available from multiple global suppliers, preventing single-source dependency risks. The robustness of the reaction conditions, which do not require ultra-high pressures or cryogenic temperatures for the majority of steps, allows for production in standard stainless steel reactors found in most fine chemical facilities. This compatibility with existing infrastructure accelerates the timeline for technology transfer and commercial scale-up of complex fluorescent dyes, ensuring that supply commitments can be met reliably without the need for specialized equipment investments.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, as the primary byproducts are water and simple salts that are easily treated in standard effluent processing systems. The use of recyclable solvents like toluene and ethanol aligns with green chemistry principles, reducing the environmental footprint of the manufacturing operation. This compliance with environmental regulations simplifies the permitting process for new production lines and supports corporate sustainability goals, making the supply of these materials more resilient against tightening environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these phenazine-based fluorophores. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on performance and handling.
Q: What are the key optical properties of these phenazine dyes compared to traditional rhodamines?
A: These novel phenazine dyes exhibit near-infrared emission (635-829nm), significantly larger Stokes shifts (up to 122nm), and superior photostability compared to classical dyes like rhodamine or oxazine, making them ideal for deep-tissue imaging.
Q: Are the raw materials for this synthesis commercially available?
A: Yes, the synthesis utilizes readily available starting materials such as substituted o-phenylenediamines, cyclohexanedione, and common solvents like ethanol and toluene, ensuring a robust and scalable supply chain.
Q: How is the purity of the final fluorescent dye ensured during production?
A: The process incorporates rigorous purification steps including neutral alumina column chromatography and recrystallization, allowing for the removal of side products and unreacted intermediates to meet stringent purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenazine Fluorescent Dye Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of these near-infrared phenazine dyes in next-generation diagnostic and optical applications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from research to market is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of dye meets the exacting standards required for sensitive biological imaging and electronic material formulations. Our commitment to quality assurance means that you receive materials with consistent optical properties, batch after batch.
We invite you to collaborate with us to optimize your supply chain for these advanced materials. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your innovation pipeline while driving down costs and ensuring supply continuity.
