Advanced Catalytic Synthesis of 2-(Cyclohexenylene) Malonic Acid Derivatives for Commercial Scale-Up
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more efficient and sustainable pathways to complex intermediates. Patent CN108264469B introduces a groundbreaking methodology for the preparation of 2-(cyclohexenylene) malonic acid derivatives, a class of compounds pivotal in the synthesis of advanced agrochemicals and pharmaceuticals. This innovation departs radically from conventional strategies by utilizing a catalytic cyclization between readily available olefins and 2-substituted malonic acid derivatives. For R&D directors and procurement specialists alike, this represents a significant opportunity to optimize supply chains for critical intermediates used in herbicides such as pinoxaden. The patent details a robust protocol that operates under mild conditions, utilizing common organic bases or acids as catalysts, thereby circumventing the severe limitations of prior art which often relied on scarce or unstable starting materials.
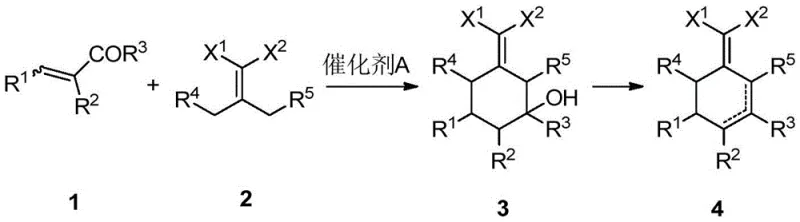
The significance of this technology extends beyond mere academic interest; it addresses fundamental bottlenecks in the manufacturing of sterically hindered cyclic structures. By enabling the efficient construction of the cyclohexene ring system from acyclic precursors, the method opens new avenues for structural diversity that were previously inaccessible or economically unviable. This is particularly relevant for the production of high-value agrochemical intermediates where purity and cost-efficiency are paramount. The ability to synthesize 2-(2,6-disubstituted cyclohexenylene) malonic acid derivatives with high regioselectivity and yield positions this technology as a cornerstone for next-generation process development in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(cyclohexenylene) malonic acid derivatives has been plagued by significant technical hurdles that impede industrial scalability. The first conventional approach relies on the Knoevenagel condensation of cyclohexenone derivatives with malonic acid esters. While conceptually straightforward, this method suffers from catastrophic yield drops when dealing with sterically hindered substrates, particularly those substituted at the 6-position. Literature indicates that yields can plummet to as low as 3% for such challenging substrates due to severe steric repulsion during the condensation step. Furthermore, the second traditional method involves the use of cyclohexadiene cobalt complexes reacting with dimethyl malonate under cryogenic conditions using strong bases like LDA. This organometallic route is not only prohibitively expensive due to the cost of cobalt reagents but also operationally complex, requiring ultra-low temperatures around -78°C and generating substantial heavy metal waste, rendering it unsuitable for green manufacturing initiatives.
The Novel Approach
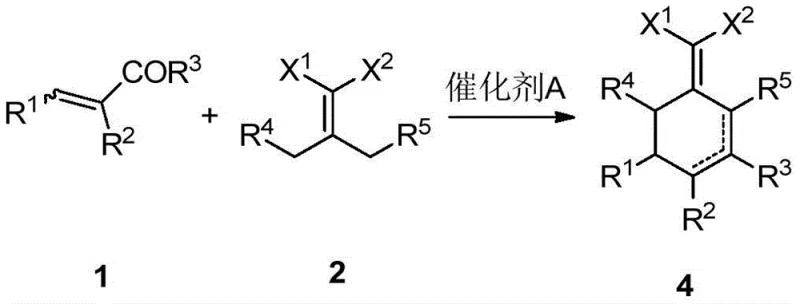
In stark contrast, the methodology disclosed in CN108264469B offers a paradigm shift by constructing the cyclic core through a catalytic cyclization of acyclic olefins and malonate derivatives. This strategy completely bypasses the need for pre-formed cyclohexenone rings or toxic cobalt complexes. The reaction proceeds efficiently using simple catalysts such as triethylamine or DABCO in common solvents like toluene at moderate temperatures ranging from 80°C to 130°C. This "one-pot" capability allows for the direct formation of the target 2-(cyclohexenylene) malonic acid derivatives without isolating unstable intermediates. The versatility of this approach is demonstrated by its ability to tolerate various functional groups and substituents, including bulky alkyl and aryl groups, achieving yields that are substantially higher than traditional methods. This robustness makes it an ideal candidate for reliable agrochemical intermediate supplier networks seeking to diversify their synthetic portfolios.
Mechanistic Insights into Catalytic Cyclization and Aromatization
The core of this innovation lies in the mechanistic elegance of the cyclization step, which likely proceeds via a Michael addition-initiated cascade. The nucleophilic attack of the activated methylene group of the malonic acid derivative onto the electron-deficient olefin triggers a sequence of intramolecular reactions that close the six-membered ring. The use of organic bases like triethylamine facilitates the generation of the necessary enolate species while maintaining a mild reaction environment that preserves sensitive functional groups. This mechanism is distinct from radical or organometallic pathways, offering superior control over stereochemistry and regioselectivity. The subsequent dehydration or elimination steps lead to the formation of the exocyclic double bond characteristic of the 2-(cyclohexenylene) motif. Understanding this mechanism is crucial for process chemists aiming to optimize reaction parameters for commercial scale-up of complex polymer additives or pharmaceutical intermediates.
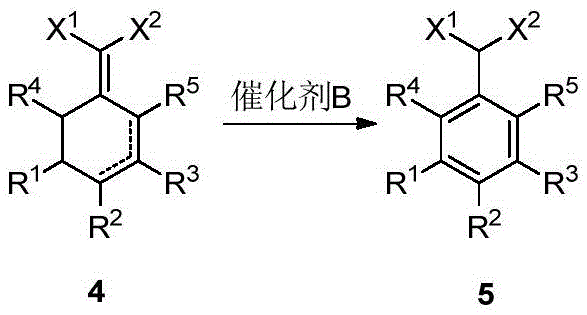
Furthermore, the patent elucidates a valuable downstream application involving the aromatization of these cyclic derivatives to form 2-arylmalonic acid derivatives. This transformation is achieved using a heterogeneous metal catalyst, preferably Pd/C, at elevated temperatures between 180°C and 220°C. This step effectively converts the cyclohexene ring into a benzene ring, a transformation of immense value in the synthesis of biaryl structures found in many active pharmaceutical ingredients and herbicides. The ability to seamlessly transition from the cyclic intermediate to the aromatic product in a controlled manner highlights the versatility of this synthetic platform. It provides a streamlined route to complex molecular architectures that would otherwise require multiple protection-deprotection sequences, thereby enhancing the overall atom economy of the process.
How to Synthesize 2-(Cyclohexenylene) Malonic Acid Derivatives Efficiently
Implementing this synthesis requires careful attention to reagent quality and thermal management to ensure consistent results. The process begins with the precise mixing of the olefinic substrate and the malonic acid derivative in a dry, oxygen-free environment to prevent side reactions. The choice of solvent plays a critical role, with toluene being identified as the optimal medium due to its ability to dissolve both reactants and withstand the required reflux temperatures. Detailed standardized operating procedures are essential for maintaining safety and efficiency, particularly when scaling from laboratory benchtop to pilot plant reactors. The following guide outlines the critical operational phases derived from the patent examples to assist technical teams in replicating this high-yielding pathway.
- Combine the olefin substrate and 2-substituted malonic acid derivative in a suitable organic solvent such as toluene.
- Add a catalytic amount of organic base like triethylamine or DABCO and heat the mixture to reflux temperatures between 80-130°C.
- Upon completion, perform an acidic workup to remove basic catalysts, followed by concentration and purification to isolate the target cyclohexenylene derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this catalytic cyclization technology offers profound advantages for supply chain resilience and cost management. Traditional methods relying on specialized cyclohexenones or organometallic reagents create single points of failure in the supply chain due to the limited number of global suppliers for such niche starting materials. By shifting to commodity chemicals like simple olefins and malonates, manufacturers can leverage existing bulk chemical infrastructure, significantly reducing raw material costs and lead times. This strategic pivot enhances supply chain reliability, ensuring that production schedules for critical agrochemical intermediates are not disrupted by the scarcity of exotic reagents. Moreover, the elimination of cryogenic operations and heavy metal catalysts in the primary step simplifies facility requirements and reduces capital expenditure on specialized cooling and waste treatment systems.
- Cost Reduction in Manufacturing: The economic impact of this method is driven primarily by the substitution of expensive, low-yield starting materials with abundant, low-cost commodity chemicals. The avoidance of stoichiometric cobalt reagents and strong bases like LDA removes a major cost center associated with reagent procurement and hazardous waste disposal. Additionally, the high yields reported in the patent examples mean that less raw material is required per unit of product, directly improving the cost of goods sold. The simplified workup procedures, which often involve basic aqueous washes rather than complex chromatographic separations, further reduce operational expenses related to solvent consumption and labor. These factors collectively contribute to substantial cost savings in herbicide manufacturing without compromising product quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of universally available feedstocks that are not subject to the geopolitical or logistical constraints often associated with specialty organometallics. The robustness of the reaction conditions allows for manufacturing in a wider range of facilities, increasing the potential for geographic diversification of production sites. This flexibility is crucial for mitigating risks associated with regional disruptions or regulatory changes affecting specific chemical classes. By establishing a synthesis route based on stable and widely traded chemicals, companies can secure long-term supply agreements with greater confidence, ensuring a steady flow of high-purity intermediates to downstream formulation plants.
- Scalability and Environmental Compliance: The environmental profile of this process aligns perfectly with modern sustainability goals, facilitating easier regulatory approval and community acceptance. The absence of heavy metal residues in the initial cyclization step simplifies the purification process and reduces the burden on wastewater treatment facilities. The use of heterogeneous catalysts in the subsequent aromatization step allows for easy catalyst recovery and recycling, minimizing solid waste generation. These green chemistry attributes make the process highly scalable, as the engineering challenges associated with heat transfer and mixing are manageable with standard reactor designs. Consequently, this technology supports the commercial scale-up of complex agrochemical intermediates while maintaining strict adherence to environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. They are designed to provide clarity on the practical aspects of the synthesis, helping stakeholders evaluate its fit for their specific production needs. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: How does this new method overcome steric hindrance issues in 2,6-disubstituted derivatives?
A: Unlike traditional Knoevenagel condensations which struggle with sterically hindered cyclohexenones, this catalytic cyclization builds the ring from acyclic precursors, bypassing the steric bottleneck and achieving high yields even for 2,6-disubstituted targets.
Q: Is the aromatization step compatible with large-scale herbicide production?
A: Yes, the aromatization utilizes heterogeneous Pd/C catalysts at elevated temperatures without requiring exotic reagents, making it highly scalable and suitable for the industrial synthesis of herbicides like pinoxaden.
Q: What are the environmental benefits compared to organometallic methods?
A: This process eliminates the need for stoichiometric amounts of expensive and toxic cobalt complexes and cryogenic conditions, significantly reducing heavy metal waste and energy consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(Cyclohexenylene) Malonic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic routes described in CN108264469B for the global agrochemical and pharmaceutical markets. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle the specific thermal and catalytic requirements of this cyclization chemistry, guaranteeing stringent purity specifications for every batch. With rigorous QC labs and a commitment to continuous improvement, we are positioned to be your trusted partner in bringing these advanced intermediates to market efficiently and safely.
We invite you to explore how our expertise can accelerate your project timelines and optimize your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact us today to request specific COA data and route feasibility assessments for 2-(cyclohexenylene) malonic acid derivatives and related herbicide intermediates. Let us collaborate to build a more resilient and cost-effective supply chain for your critical chemical needs.
