Advanced Manufacturing of High-Purity Hydroxyfasudil Semihydrate for Global Pharma Supply Chains
Advanced Manufacturing of High-Purity Hydroxyfasudil Semihydrate for Global Pharma Supply Chains
The pharmaceutical industry continuously demands more efficient and robust synthetic routes for critical cardiovascular therapeutics, particularly for Rho kinase inhibitors like Fasudil. Patent CN103450157A introduces a groundbreaking preparation method for high-purity hydroxyfasudil semihydrate that addresses long-standing challenges in yield, purity, and operational complexity. This technical disclosure outlines a streamlined process that bypasses the instability issues associated with traditional sulfonyl chloride intermediates, offering a viable pathway for reliable pharmaceutical intermediate supplier networks to enhance their production capabilities. By leveraging a direct condensation strategy followed by a sophisticated acid-base extraction and controlled crystallization protocol, manufacturers can achieve product purity exceeding 99.9% while significantly reducing processing time. This innovation represents a pivotal shift towards greener and more economical API manufacturing, ensuring consistent supply quality for downstream drug formulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
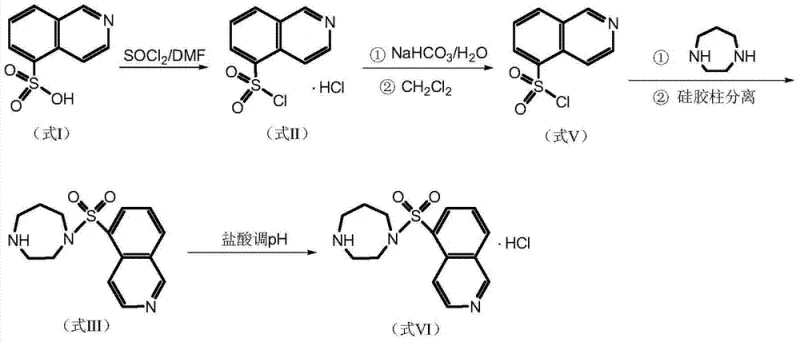
Traditional synthetic routes for Fasudil Hydrochloride, as documented in prior art such as US5942505 and CN102020636, typically involve a multi-step sequence that begins with the generation of the free base of 5-isoquinoline sulfonyl chloride. This intermediate is notoriously unstable and prone to rapid hydrolysis upon exposure to moisture, necessitating strict anhydrous conditions and immediate consumption. Furthermore, the conventional condensation reaction with homopiperazine often generates significant amounts of dimer impurities and colored by-products due to exothermic runaway and uncontrolled reaction kinetics. To mitigate these issues, existing processes rely heavily on column chromatography purification, a technique that is inherently batch-limited, solvent-intensive, and difficult to scale for industrial production. The reliance on chromatographic separation not only inflates manufacturing costs but also introduces bottlenecks in supply chain continuity, making it challenging to meet the high-volume demands of the global market.
The Novel Approach
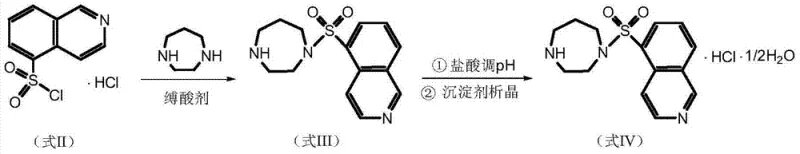
In stark contrast to legacy methods, the novel approach detailed in CN103450157A utilizes 5-isoquinoline sulfonyl chloride hydrochloride directly as the starting material, thereby circumventing the generation of the unstable free base species. This strategic modification allows the condensation reaction to proceed in the presence of an acid-binding agent within a dichloromethane solvent system, maintaining thermal control below 15°C to suppress side reactions. The subsequent workup involves a precise sequence of water washing, acid extraction, and alkalization, which effectively partitions impurities and pigments into aqueous phases without the need for chromatographic media. The final crystallization step employs a specific methanol-water solvent system with controlled pH adjustment to 5.9-6.1, ensuring the selective precipitation of the thermodynamically stable hemihydrate form. This methodology not only simplifies the operational workflow but also enhances the overall robustness of the manufacturing process, making it ideal for cost reduction in API manufacturing.
Mechanistic Insights into Sulfonylation and Crystallization Control
The core chemical transformation in this process is the nucleophilic substitution of the sulfonyl chloride group by homopiperazine, facilitated by the presence of a solid acid-binding agent such as potassium carbonate. Mechanistically, the acid-binding agent serves a dual purpose: it neutralizes the hydrochloric acid released during the formation of the sulfonamide bond and buffers the reaction medium to prevent the protonation of the nucleophilic amine, which would otherwise deactivate it. By maintaining the reaction temperature between 0°C and 15°C, the kinetic energy of the system is kept low enough to favor the mono-substitution product over the bis-substituted dimer impurity. The use of the hydrochloride salt of the sulfonyl chloride ensures that the electrophile remains stable until it encounters the activated amine base in the organic phase, thereby minimizing hydrolysis pathways that lead to 5-isoquinoline sulfonic acid contaminants.
Impurity control is further refined through the liquid-liquid extraction protocol, which exploits the differential solubility and ionization states of the reaction components. The initial acid extraction with dilute hydrochloric acid converts the basic fasudil product into its water-soluble salt form, leaving neutral organic impurities and unreacted starting materials in the dichloromethane layer. Subsequent basification of the aqueous layer regenerates the free base, which is then extracted back into the organic phase, effectively leaving inorganic salts and highly polar degradation products behind. The final crystallization mechanism relies on the precise control of supersaturation and pH; adjusting the pH to the narrow range of 5.9-6.1 ensures that the mono-hydrochloride salt precipitates selectively, while any potential di-hydrochloride species remain in solution. This rigorous control over the solid-state chemistry guarantees a consistent crystal lattice with the desired water content of 2.5% to 3.3%, critical for the stability and bioavailability of the final drug substance.
How to Synthesize Hydroxyfasudil Semihydrate Efficiently
The synthesis of high-purity hydroxyfasudil semihydrate requires strict adherence to the optimized reaction parameters and workup procedures defined in the patent to ensure reproducibility and quality. Operators must prioritize temperature control during the addition of the sulfonyl chloride salt to prevent exotherms that could degrade product quality. The detailed standardized synthesis steps, including specific reagent ratios, stirring times, and drying conditions, are outlined in the technical guide below to assist process engineers in implementing this route.
- Condense 5-isoquinoline sulfonyl chloride hydrochloride with homopiperazine in dichloromethane using an acid-binding agent at low temperature.
- Perform sequential acid-base extraction workup to remove impurities and isolate the fasudil base.
- Dissolve the base in methanol-water, adjust pH to 5.9-6.1 with hydrochloric acid, and crystallize using a precipitation agent to obtain the semihydrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers substantial strategic benefits that extend beyond mere technical feasibility. The elimination of column chromatography represents a paradigm shift in processing efficiency, removing a major bottleneck that typically constrains throughput and increases lead time for high-purity pharmaceutical intermediates. By replacing this batch-dependent purification step with continuous liquid-liquid extraction and crystallization, manufacturers can drastically increase batch sizes and reduce cycle times, leading to a more responsive supply chain capable of meeting fluctuating market demands. Furthermore, the reduction in solvent consumption and waste generation aligns with increasingly stringent environmental regulations, lowering the total cost of ownership for the production facility.
- Cost Reduction in Manufacturing: The removal of silica gel column chromatography eliminates the recurring cost of stationary phase materials and the extensive solvent volumes required for elution and regeneration. This qualitative improvement in process design translates directly to lower variable costs per kilogram of product, as the operation shifts from a labor-intensive purification method to a more automated extraction process. Additionally, the higher yield obtained by minimizing hydrolysis and dimerization side reactions means that less raw material is wasted, further enhancing the economic viability of the process for large-scale commercial production.
- Enhanced Supply Chain Reliability: Utilizing the stable hydrochloride salt of 5-isoquinoline sulfonyl chloride as a starting material mitigates the risk of raw material degradation during storage and transport, which is a common issue with the free base form used in older methods. This stability ensures a consistent quality of input materials, reducing the likelihood of batch failures and production delays. The robustness of the crystallization step also guarantees a uniform particle size and crystal form, which simplifies downstream handling and packaging, thereby securing a steady flow of high-quality product to customers.
- Scalability and Environmental Compliance: The unit operations employed in this process, such as agitation, filtration, and distillation, are standard equipment found in most multipurpose chemical plants, facilitating easy technology transfer and scale-up from pilot to commercial scale. The avoidance of complex purification techniques reduces the generation of hazardous solid waste associated with spent chromatography media, simplifying waste disposal protocols. This streamlined approach not only accelerates the timeline for commercial scale-up of complex pharmaceutical intermediates but also supports sustainability goals by minimizing the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of hydroxyfasudil semihydrate, based on the specific advancements disclosed in the patent literature. These insights are designed to clarify the operational advantages and quality standards associated with this improved manufacturing method for stakeholders evaluating potential supply partners.
Q: How does this novel process improve purity compared to conventional methods?
A: By utilizing the stable hydrochloride salt of the sulfonyl chloride directly and employing a specific acid-base extraction sequence, the process effectively removes dimer impurities and pigments without the need for column chromatography, achieving purity greater than 99.9%.
Q: What are the critical control points for obtaining the hemihydrate crystal form?
A: Critical parameters include maintaining the salification pH between 5.9 and 6.1 during crystallization and drying the final product under reduced pressure at 60-80°C for at least 8 hours to ensure the correct water content of 2.5% to 3.3%.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the elimination of column chromatography and the use of standard unit operations like extraction and crystallization make this route highly scalable, robust, and cost-effective for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxyfasudil Semihydrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest international standards.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific supply chain requirements. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate how our optimized processes can enhance your product portfolio. Contact us today to discuss how our expertise in high-purity API manufacturing can drive value and reliability for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
