Advanced Ionic Liquid Catalysis for Scalable Production of High-Purity Thiazolidinedione Derivatives
The escalating global prevalence of Type II diabetes has intensified the demand for effective therapeutic agents, particularly thiazolidinedione derivatives which serve as critical active pharmaceutical ingredients (APIs) and intermediates. Patent CN101293877A introduces a groundbreaking methodology for synthesizing these vital compounds, addressing significant bottlenecks in traditional manufacturing processes. This innovation utilizes the functionalized ionic liquid 1-butyl-3-methylimidazolium hydroxide ([Bmim]OH) as a dual-purpose catalyst and solvent, marking a paradigm shift towards sustainable and efficient chemical production. By replacing hazardous volatile organic solvents with a recyclable ionic medium, this technology not only aligns with rigorous green chemistry principles but also offers a robust pathway for the industrial scale-up of complex pharmaceutical intermediates. The strategic implementation of this synthesis route promises to enhance supply chain stability while delivering high-purity materials essential for downstream drug formulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-arylmethylene-2,4-thiazolidinedione derivatives has relied heavily on conventional organic solvents such as benzene, toluene, methanol, ethanol, acetic acid, DMF, and DMSO. These traditional protocols typically require the addition of separate organic acids, bases, or salts like piperidine, morpholine, or ammonium acetate to catalyze the condensation reaction. A major drawback of these legacy methods is the necessity for high reaction temperatures and extended reaction times, often ranging from 16 to 18 hours, which severely limits production throughput and energy efficiency. Furthermore, the reliance on volatile and toxic solvents creates substantial environmental hazards, necessitating complex waste treatment procedures and increasing the overall carbon footprint of the manufacturing process. The use of corrosive acids like acetic acid also poses equipment maintenance challenges and safety risks for operational personnel, while the difficulty in separating products from these solvent systems often leads to lower overall yields and compromised purity profiles.
The Novel Approach
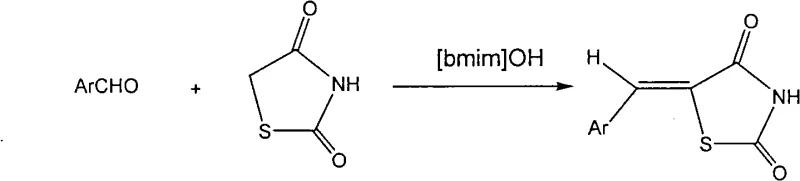
The novel approach detailed in the patent data revolutionizes this synthesis by employing the basic ionic liquid [Bmim]OH, which functions simultaneously as the reaction medium and the base catalyst. This eliminates the need for any additional catalytic additives, streamlining the reagent list and simplifying the reaction setup significantly. Operating at moderate temperatures between 50°C and 100°C, this method drastically reduces reaction times to a window of just 2 to 10 hours, thereby enhancing production capacity and reducing energy consumption. The unique physicochemical properties of [Bmim]OH, including its near-zero vapor pressure and thermal stability, ensure a non-volatile and safe working environment that complies with modern environmental regulations. Moreover, the product separation is remarkably straightforward; simply adding distilled water precipitates the crude product, allowing for easy filtration and washing, which translates to higher operational efficiency and reduced downtime between batches compared to traditional distillation-heavy workups.
Mechanistic Insights into Ionic Liquid Catalyzed Knoevenagel Condensation
The core of this technological advancement lies in the mechanistic efficiency of the [Bmim]OH ionic liquid during the Knoevenagel condensation reaction between aromatic aldehydes and 2,4-thiazolidinedione. The hydroxide anion in the ionic liquid provides the necessary basicity to deprotonate the active methylene group of the thiazolidinedione ring, generating a nucleophilic enolate intermediate that attacks the carbonyl carbon of the aromatic aldehyde. Unlike heterogeneous catalysts that may suffer from mass transfer limitations, the homogeneous nature of the ionic liquid ensures intimate contact between reactants, facilitating rapid reaction kinetics even at lower temperatures. The cationic imidazolium component stabilizes the transition states through electrostatic interactions, further lowering the activation energy required for the dehydration step that forms the final double bond. This synergistic effect results in a highly selective transformation that minimizes side reactions, such as polymerization or over-condensation, which are common pitfalls in acidic or poorly controlled basic environments.
Impurity control is inherently superior in this system due to the mild yet effective catalytic environment provided by the ionic liquid. Traditional strong bases or acids can often lead to the degradation of sensitive functional groups on the aromatic ring, such as nitro or hydroxyl groups, but the buffered basicity of [Bmim]OH preserves these moieties intact. The high solubility of the reactants in the ionic liquid phase ensures a homogeneous reaction mixture, preventing localized hot spots of high concentration that could trigger unwanted byproduct formation. Following the reaction, the addition of water disrupts the solvation shell of the product, causing it to crystallize out while the ionic liquid remains in the aqueous phase, effectively acting as a built-in extraction system. This physical separation mechanism prevents the entrapment of catalyst residues within the crystal lattice of the product, contributing to the consistently high purity levels of over 98% observed across various substrate scopes without the need for extensive chromatographic purification.
How to Synthesize 5-Arylmethylene-2,4-thiazolidinedione Efficiently
The synthesis protocol outlined in the patent offers a standardized and reproducible method for producing high-quality thiazolidinedione derivatives suitable for pharmaceutical applications. The process begins with the precise mixing of stoichiometric amounts of the chosen aromatic aldehyde and 2,4-thiazolidinedione with the [Bmim]OH ionic liquid, ensuring optimal molar ratios for maximum conversion. Reaction progress is easily monitored using thin-layer chromatography (TLC), allowing operators to determine the exact endpoint and prevent over-reaction. Upon completion, the workup procedure involves a simple aqueous quench followed by filtration, making it highly amenable to automation and large-scale batch processing. For a comprehensive understanding of the specific operational parameters and safety considerations, the detailed standardized synthesis steps are provided in the guide below.
- Mix aromatic aldehyde and 2,4-thiazolidinedione with ionic liquid [Bmim]OH in a reactor.
- Stir the mixture at 50-100°C for 2-10 hours until reaction completion monitored by TLC.
- Add distilled water to precipitate the product, filter, wash, and recrystallize from ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ionic liquid catalyzed process presents a compelling value proposition centered on cost optimization and risk mitigation. The elimination of volatile organic solvents removes the significant costs associated with solvent recovery, disposal, and regulatory compliance regarding VOC emissions. By integrating the catalyst and solvent into a single reusable component, the material bill of quantities is simplified, reducing the complexity of inventory management and sourcing logistics. The ability to recycle the [Bmim]OH ionic liquid multiple times without significant loss of activity means that the effective cost per kilogram of catalyst approaches zero over the long term, driving down the variable cost of goods sold. Furthermore, the shortened reaction cycles increase asset utilization rates, allowing existing reactor infrastructure to produce more volume in less time, which is a critical factor in meeting tight delivery schedules for downstream API manufacturers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the dual functionality of the ionic liquid, which negates the purchase of separate catalysts and expensive anhydrous solvents. Since the ionic liquid can be recovered from the filtrate by simple distillation to remove water, the recurring material cost is substantially lower than traditional methods that consume stoichiometric amounts of reagents. The simplified workup procedure, which relies on water precipitation rather than complex extractions or column chromatography, significantly reduces labor hours and utility consumption for solvent evaporation. Additionally, the high yield range of 85% to 92% minimizes raw material waste, ensuring that a greater proportion of the input cost is converted into saleable product, thereby improving overall gross margins for the manufacturing operation.
- Enhanced Supply Chain Reliability: Sourcing stability is improved because the key reagents, aromatic aldehydes and 2,4-thiazolidinedione, are commodity chemicals with robust global supply chains, unlike specialized catalysts that may have single-source dependencies. The robustness of the reaction conditions, which tolerate a wide range of temperatures and do not require strict inert atmospheres, reduces the risk of batch failures due to minor operational deviations. This reliability translates into more predictable lead times for customers, as production schedules are less likely to be disrupted by equipment corrosion issues or solvent quality fluctuations. The scalability of the process from gram to ton scale ensures that supply can be ramped up quickly to meet surges in demand for anti-diabetic medications without requiring significant capital investment in new specialized equipment.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers a distinct advantage by operating as a closed-loop system with minimal emissions. The non-volatile nature of the ionic liquid eliminates the risk of explosive vapor clouds, enhancing plant safety and potentially lowering insurance premiums. Waste generation is drastically reduced because the aqueous waste stream contains mainly the recyclable ionic liquid and minimal organic contaminants, simplifying wastewater treatment requirements. This alignment with green chemistry principles facilitates easier regulatory approval in markets with strict environmental standards, such as Europe and North America, removing a potential barrier to market entry. The process is inherently scalable, as heat transfer and mixing are more efficient in the ionic liquid medium compared to viscous traditional solvent mixtures, ensuring consistent quality during commercial scale-up.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and product quality. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The responses highlight the practical advantages concerning purity, recycling, and operational simplicity that distinguish this method from legacy technologies.
Q: What are the advantages of using [Bmim]OH over traditional solvents?
A: The ionic liquid [Bmim]OH serves a dual role as both solvent and catalyst, eliminating the need for volatile organic compounds like benzene or acetic acid and additional catalysts, thereby simplifying purification and reducing environmental impact.
Q: Can the ionic liquid catalyst be recycled?
A: Yes, the filtrate containing the ionic liquid can be distilled under reduced pressure to remove water, allowing the pure ionic liquid to be recovered and reused in subsequent batches, significantly lowering material costs.
Q: What is the typical purity and yield of this method?
A: This method consistently achieves yields between 85% and 92% with product purity exceeding 98% after simple recrystallization, meeting stringent pharmaceutical intermediate specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Arylmethylene-2,4-thiazolidinedione Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this ionic liquid catalyzed synthesis route for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry method are fully realized in an industrial setting. Our state-of-the-art facilities are equipped to handle ionic liquid chemistries safely and efficiently, maintaining stringent purity specifications through our rigorous QC labs and advanced analytical instrumentation. We are committed to delivering 5-arylmethylene-2,4-thiazolidinedione derivatives that meet the highest quality standards, supporting your drug development pipelines with reliable and compliant supply.
We invite you to collaborate with us to optimize your supply chain for anti-diabetic drug intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this novel synthesis method can reduce your total cost of ownership. Please contact us to request specific COA data for our current inventory or to discuss route feasibility assessments for your custom projects. By leveraging our expertise in green synthesis and scale-up, we can help you secure a competitive advantage in the global pharmaceutical market through superior quality and sustainable manufacturing practices.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
