Advanced Synthesis of Axial Chiral Imidazolium Salts for High-Selectivity Asymmetric Catalysis
Introduction to Next-Generation Chiral Catalyst Precursors
The landscape of asymmetric synthesis is continually evolving, driven by the demand for higher enantioselectivity and robust catalytic systems. Patent CN102491947A introduces a significant advancement in this field through the development of novel axial chiral imidazolium salt compounds. These compounds serve as critical precursors for N-heterocyclic carbenes (NHCs), which have emerged as superior ligands compared to traditional phosphines due to their exceptional stability and strong coordination capabilities with transition metals. For R&D directors and procurement specialists in the fine chemical sector, understanding the synthesis and application of these materials is vital for optimizing production lines. The technology detailed in this patent offers a pathway to generate chiral catalysts that are not only highly effective in inducing asymmetry but also possess the thermal and oxidative stability required for rigorous industrial processes. This report analyzes the technical merits and commercial implications of adopting this synthesis route for reliable catalyst precursor supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral nitrogen ligands, particularly those with axial chirality, has been fraught with challenges that hinder their widespread industrial adoption. Traditional methods often suffer from low overall yields and require harsh reaction conditions that can compromise the integrity of sensitive functional groups. A major bottleneck in the field has been the difficulty in synthesizing axially chiral imidazolium salts or their corresponding azacarbenes, which has severely limited the exploration of new catalytic applications. Furthermore, the dominant class of chiral ligands, organophosphines, exhibits significant instability when exposed to oxidizing agents. This sensitivity restricts their utility in asymmetric oxidation reactions, a critical transformation in the manufacture of many active pharmaceutical ingredients (APIs). The inability to maintain catalyst integrity under oxidative stress leads to inconsistent batch quality and increased costs associated with catalyst replacement and purification.
The Novel Approach
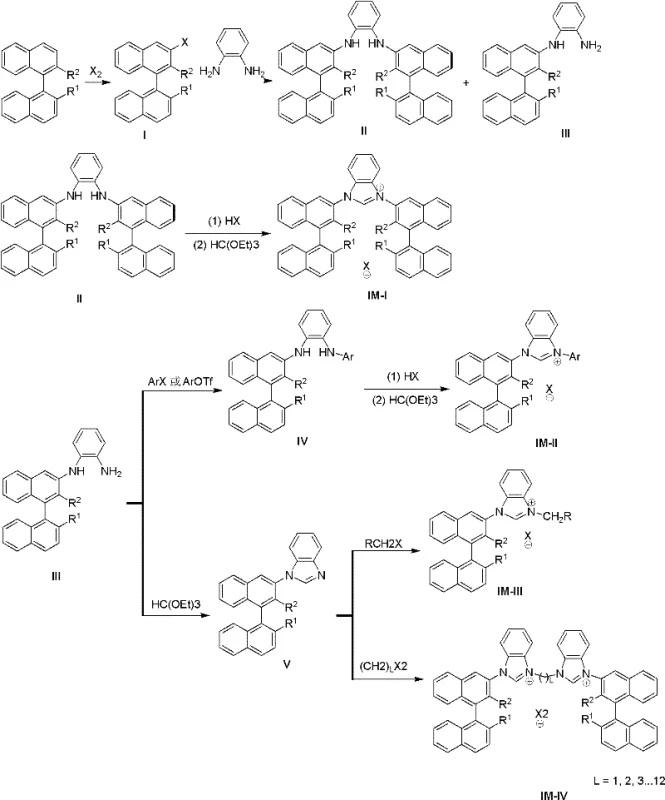
The methodology outlined in CN102491947A presents a robust solution to these longstanding issues by leveraging a modular synthesis strategy starting from binaphthol derivatives. This novel approach ensures that the chiral center is positioned in close proximity to the reactive site of the catalyst, a structural feature that is instrumental in achieving high asymmetric selectivity. By utilizing a palladium-catalyzed coupling reaction followed by cyclization with trialkyl orthoformates, the process achieves remarkably high yields, with specific examples demonstrating conversion rates exceeding 80% in key steps. Unlike phosphine-based systems, the resulting imidazolium salts and their metal complexes demonstrate excellent stability against air, humidity, and heat. This resilience not only simplifies storage and handling requirements but also expands the scope of applicable reactions to include asymmetric oxidations, thereby offering a versatile toolset for cost reduction in catalyst manufacturing and process optimization.
Mechanistic Insights into Palladium-Catalyzed Coupling and Cyclization
The core of this synthesis technology relies on a sophisticated sequence of organometallic transformations that ensure precise stereochemical control. The process initiates with the halogenation of binaphthol derivatives to generate 3-halo-binaphthyl intermediates, which serve as the electrophilic partners in the subsequent coupling step. In the presence of a palladium catalyst, such as Pd(OAc)2, and a phosphine ligand like BINAP, these intermediates undergo a Buchwald-Hartwig type amination with o-phenylenediamine. This step is critical as it establishes the C-N bond while preserving the axial chirality of the binaphthyl backbone. The reaction typically proceeds at temperatures between 50-150°C, allowing for tunable kinetics depending on the steric bulk of the substrates. Following the coupling, the resulting axial chiral o-diamine compounds are subjected to cyclization. This is achieved by reacting the diamine with a trialkyl orthoformate in the presence of an acid, facilitating the formation of the imidazole ring. The proximity of the bulky binaphthyl groups to the imidazolium core creates a well-defined chiral pocket, which is essential for discriminating between enantiomeric transition states during catalysis.
Impurity control is inherently managed through the high selectivity of the palladium-catalyzed steps and the crystallization properties of the final salts. The use of specific bases like Cs2CO3 and solvents such as 1,4-dioxane or toluene helps minimize side reactions such as homocoupling or over-alkylation. Furthermore, the final quaternization step, where alkyl halides are introduced to form the imidazolium cation, is conducted under mild conditions (25-130°C) to prevent racemization. The resulting compounds, characterized by their distinct NMR and mass spectrometry signatures, exhibit high purity levels suitable for direct use in sensitive catalytic cycles. This mechanistic robustness ensures that the high-purity catalyst precursors produced are consistent in performance, reducing the risk of batch-to-batch variability in downstream pharmaceutical synthesis.
How to Synthesize Axial Chiral Imidazolium Salts Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and optical purity. The process is divided into three main stages: the preparation of the halogenated binaphthyl scaffold, the palladium-catalyzed coupling with diamines, and the final cyclization and quaternization. Each stage utilizes commercially available reagents and standard laboratory equipment, making the transition from bench scale to pilot plant feasible. The detailed standardized synthesis steps below outline the specific conditions, stoichiometry, and workup procedures required to replicate the high efficiency reported in the patent data. Adhering to these protocols ensures the production of materials that meet the stringent specifications required for commercial scale-up of complex catalysts.
- React binaphthol derivatives with halogens to obtain 3-halo-binaphthyl compounds, followed by Pd-catalyzed coupling with o-phenylenediamine at 50-150°C.
- Purify the resulting axial chiral o-diamine compounds to isolate C2 symmetric or monosubstituted intermediates.
- React the diamine intermediates with trialkyl orthoformate and acid at 60-130°C to form the final imidazolium salt structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis technology offers tangible benefits that extend beyond mere technical performance. The reliance on binaphthol derivatives as starting materials leverages a supply chain that is well-established and cost-effective, mitigating the risks associated with sourcing exotic or proprietary reagents. The high yields observed in the experimental data, such as the 97% yield in the initial halogenation step and substantial conversion in the coupling phases, directly translate to reduced raw material consumption per kilogram of final product. This efficiency is a key driver for cost reduction in fine chemical manufacturing, as it lowers the overall cost of goods sold (COGS) and minimizes waste disposal expenses. Additionally, the stability of the final imidazolium salts reduces the need for specialized storage infrastructure, such as inert atmosphere gloveboxes or cryogenic facilities, further lowering operational overheads.
- Cost Reduction in Manufacturing: The synthesis route eliminates the need for expensive and sensitive phosphine ligands in the final product, replacing them with stable imidazolium salts that are cheaper to produce and store. The high atom economy of the coupling and cyclization steps ensures that a significant portion of the input mass is converted into the desired product, drastically reducing the volume of chemical waste generated. This efficiency allows for a more streamlined production process with fewer purification cycles, leading to substantial cost savings in energy and solvent usage without compromising on the quality of the chiral inducer.
- Enhanced Supply Chain Reliability: By utilizing common reagents like Pd(OAc)2, BINAP, and trialkyl orthoformates, the manufacturing process is less susceptible to supply disruptions caused by the scarcity of niche chemicals. The robustness of the reaction conditions, which tolerate a range of temperatures and standard solvents, means that production can be maintained across different facilities with minimal requalification effort. This flexibility ensures reducing lead time for high-purity catalyst precursors, allowing for faster response to market demands and more reliable inventory management for long-term projects.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from liter-scale reactors to multi-ton vessels. The absence of highly toxic or unstable intermediates simplifies safety protocols and environmental compliance measures. The ability to produce C2-symmetric and non-C2-symmetric variants from the same core intermediates provides a flexible platform for generating diverse catalyst libraries, supporting rapid innovation while maintaining a lean and compliant manufacturing footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these axial chiral imidazolium salts. The answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this technology integrates into existing workflows. Understanding these details is crucial for technical teams evaluating the feasibility of incorporating these catalysts into their synthesis pipelines.
Q: Why are axial chiral imidazolium salts preferred over traditional phosphine ligands?
A: Unlike organophosphine ligands which are highly sensitive to oxidizing agents, imidazolium salts and their resulting N-heterocyclic carbene (NHC) metal complexes exhibit superior stability against air, moisture, and heat. This stability allows them to be used in asymmetric oxidation reactions where phosphines would degrade, broadening their utility in complex synthetic pathways.
Q: What are the key advantages of the synthesis method described in CN102491947A?
A: The patented method utilizes readily available and inexpensive raw materials like binaphthol derivatives. It achieves high synthesis yields, such as 97% in the halogenation step and up to 83% in the coupling step, significantly reducing waste. Furthermore, the chiral center is positioned close to the reaction site, which enhances asymmetric selectivity in downstream catalytic applications.
Q: What types of catalytic reactions can these imidazolium salts facilitate?
A: These compounds serve as precursors for N-heterocyclic carbenes that form stable complexes with transition metals. They are widely applicable in asymmetric coupling reactions, asymmetric addition of metal reagents to carbonyls, asymmetric oxidation and hydrogenation reactions, and the asymmetric oxidative resolution of alcohols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Imidazolium Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance chiral catalysts play in the development of next-generation pharmaceuticals and fine chemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Whether you require custom modifications to the binaphthyl backbone or specific counterions for solubility optimization, our R&D experts are equipped to tailor solutions that meet your exact process requirements.
We invite you to collaborate with us to unlock the full potential of this advanced catalytic technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis for your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you accelerate your development timeline. Let us be your partner in achieving superior asymmetric selectivity and operational efficiency.
