Scalable Synthesis of Axially Chiral Imidazolium Salts for Advanced Asymmetric Catalysis
Scalable Synthesis of Axially Chiral Imidazolium Salts for Advanced Asymmetric Catalysis
The landscape of asymmetric catalysis is undergoing a significant transformation with the introduction of robust axially chiral imidazolium salts, as detailed in patent CN102070529A. This intellectual property outlines a sophisticated yet practical methodology for generating axially chiral N-heterocyclic carbene (NHC) precursors, which serve as superior alternatives to traditional phosphine ligands in various organic transformations. For R&D directors and procurement specialists in the fine chemical sector, understanding the nuances of this technology is critical for optimizing supply chains and enhancing catalytic efficiency. The patent describes a class of compounds featuring a binaphthyl backbone linked to an imidazolium ring, offering exceptional stability and stereocontrol. By leveraging this technology, manufacturers can access high-purity organocatalysts that maintain integrity under harsh oxidative conditions, a common failure point for legacy ligand systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of asymmetric catalysis has been dominated by chiral phosphine ligands, which, while effective in many contexts, suffer from inherent stability issues that complicate their commercial deployment. Chiral phosphines are notoriously sensitive to oxidation, often degrading rapidly when exposed to air or oxidizing agents, which severely limits their utility in asymmetric oxidation reactions. Furthermore, the synthesis of complex chiral phosphines often involves multi-step sequences with sensitive intermediates, leading to higher production costs and supply chain vulnerabilities. For procurement managers, this translates to higher prices and potential delays due to the stringent handling requirements of air-sensitive materials. The instability of phosphine-metal complexes also necessitates rigorous exclusion of moisture and oxygen during catalytic processes, increasing the operational complexity and capital expenditure required for reactor setups in pharmaceutical manufacturing.
The Novel Approach
In contrast, the novel approach presented in patent CN102070529A utilizes axially chiral imidazolium salts which offer a paradigm shift in ligand stability and versatility. These compounds function as precursors to N-heterocyclic carbenes (NHCs), which form metal complexes that are remarkably stable against air, water, and thermal stress. The synthetic route described involves a straightforward two-step process starting from readily available binaphthyl diamines, avoiding the need for expensive transition metals during the ligand synthesis phase itself. This method not only simplifies the manufacturing workflow but also ensures that the final products are stable solids that can be stored and transported without specialized inert atmosphere packaging. The ability to tolerate most oxidants allows these ligands to be deployed in asymmetric oxidation, coupling, and addition reactions where phosphine ligands would fail, thereby expanding the synthetic toolbox available to process chemists.
Mechanistic Insights into Binaphthyl Diamine Cyclization
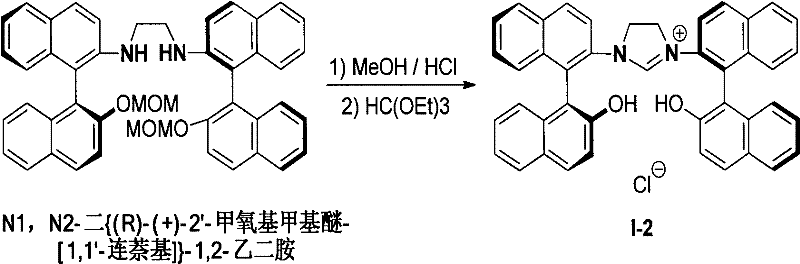
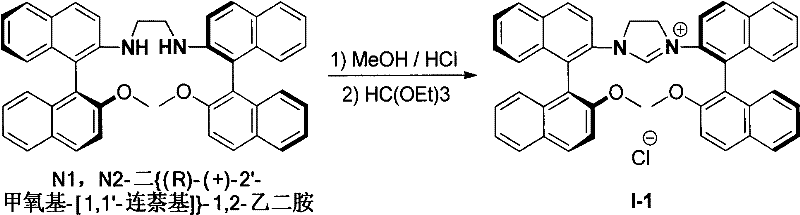
The core of this technological advancement lies in the efficient cyclization of binaphthyl axial chiral diamines to form the imidazolium ring system. The process begins with the acidification of the diamine substrate in an alcohol-acid mixed solvent, typically using anhydrous methanol or ethanol combined with a hydrohalic acid or strong acid like HBF4. This step protonates the amine groups, creating a reactive intermediate that is subsequently dried to remove residual moisture, a critical parameter for the success of the subsequent cyclization. The dried intermediate is then reacted with a trialkyl orthoformate under an inert atmosphere at elevated temperatures ranging from 80°C to 130°C. This cyclization step effectively bridges the two nitrogen atoms with a single carbon unit derived from the orthoformate, closing the imidazole ring while preserving the axial chirality of the binaphthyl backbone.
From a mechanistic perspective, the proximity of the chiral binaphthyl axis to the reactive carbene center is a key design feature that enhances asymmetric induction. Unlike ligands where the chiral information is distal to the metal center, the rigid binaphthyl structure in these imidazolium salts ensures that the steric environment around the catalytic site is tightly controlled. This structural rigidity minimizes conformational freedom, leading to higher enantioselectivity in catalytic transformations. Furthermore, the resulting NHC-metal complexes exhibit irreversible coordination, preventing ligand dissociation during catalysis which is a common cause of catalyst deactivation in phosphine systems. The robustness of this coordination chemistry ensures consistent performance over extended reaction times, a crucial factor for industrial batch processes where catalyst turnover numbers directly impact cost efficiency.
How to Synthesize Axially Chiral Imidazolium Salts Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these high-value intermediates with consistent quality. The procedure is designed to be scalable, utilizing common reagents such as triethyl orthoformate and simple mineral acids, which are readily available in bulk quantities. The workup procedure is equally straightforward, involving cooling the reaction mixture to precipitate the product followed by filtration and washing with diethyl ether, eliminating the need for complex chromatographic purification in many cases. This simplicity is a major advantage for process development teams looking to minimize unit operations and solvent consumption.
- Dissolve the binaphthyl axial chiral diamine compound in an alcohol-acid mixed solvent (volume ratio 2: 1 to 20:1) and react at 25-80°C for 1-24 hours.
- Evaporate the solvent to obtain a solid intermediate, dry it in a P2O5 desiccator, and then dissolve in trialkyl orthoformate under inert gas.
- Heat the mixture to 80-130°C for 1-24 hours, cool to room temperature to precipitate the solid product, filter, and wash with diethyl ether.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the adoption of this synthetic route offers tangible benefits in terms of cost stability and logistical ease. The use of stable solid intermediates reduces the risk associated with transporting air-sensitive materials, lowering insurance and packaging costs significantly. Moreover, the high yields reported in the patent examples, ranging from 61% to 70%, indicate a material-efficient process that maximizes the output from raw materials. This efficiency is critical for maintaining competitive pricing in the global market for chiral intermediates. The elimination of sensitive phosphine chemistry also reduces the need for specialized glovebox infrastructure, allowing for production in standard chemical manufacturing facilities.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive transition metal catalysts during the ligand formation stage, relying instead on inexpensive organic reagents like orthoformates. This fundamental shift in reagent costs leads to substantial savings in the bill of materials. Additionally, the stability of the final imidazolium salts means they do not require cold chain logistics or inert gas blanketing during storage, further reducing overhead expenses associated with warehousing and distribution. The robust nature of the process also minimizes batch failures due to environmental exposure, ensuring a more predictable cost of goods sold.
- Enhanced Supply Chain Reliability: The starting materials, specifically binaphthyl diamines, are well-established commodities in the fine chemical industry, ensuring a steady supply of feedstock. The synthesis does not rely on rare earth metals or geopolitically constrained resources, mitigating supply risk. The ability to produce these ligands in a stable solid form allows for the creation of strategic inventory buffers without degradation concerns. This reliability is essential for pharmaceutical customers who require uninterrupted supply of key chiral building blocks to maintain their own production schedules for active pharmaceutical ingredients.
- Scalability and Environmental Compliance: The process utilizes standard solvents like methanol and ethanol, which are easier to recover and recycle compared to halogenated solvents often used in sensitive organometallic chemistry. The workup involves simple filtration and washing, generating less hazardous waste compared to extraction-heavy protocols. The high thermal stability of the products allows for processing at higher concentrations, potentially reducing solvent volumes per kilogram of product. These factors contribute to a greener manufacturing profile, aligning with increasingly stringent environmental regulations and corporate sustainability goals in the chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these axially chiral imidazolium salts. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these details is vital for technical teams evaluating the feasibility of integrating these ligands into existing catalytic processes.
Q: Why are axially chiral imidazolium salts preferred over chiral phosphine ligands?
A: Unlike chiral phosphine ligands which are highly sensitive to oxidants, axially chiral imidazolium salts and their resulting N-heterocyclic carbene (NHC) metal complexes exhibit exceptional stability towards air, water, and heat. This allows them to be used effectively in asymmetric oxidation reactions where phosphines would degrade.
Q: What is the typical yield for the synthesis of these imidazolium salts?
A: According to the provided patent data, the synthesis method achieves robust yields ranging from 61% to 70% across different substituted derivatives, demonstrating a reliable and efficient production process suitable for industrial scaling.
Q: Can these compounds be used for large-scale industrial catalysis?
A: Yes, the precursor imidazolium salts are stable solids that are easy to handle and store. Their stability and the simplicity of the synthesis route, which avoids expensive transition metals during the ligand formation itself, make them highly suitable for commercial scale-up and industrial catalytic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Imidazolium Salts Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of axially chiral imidazolium salts in modern asymmetric synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of chiral ligand meets the exacting standards required for pharmaceutical intermediate synthesis. We understand that consistency is key in catalysis, and our process controls are designed to deliver reproducible enantiomeric excess and chemical purity.
We invite you to collaborate with us to optimize your catalytic processes using these advanced ligand systems. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volume and purity requirements. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. By partnering with us, you gain access to a supply chain that prioritizes both technical excellence and commercial viability, securing your position in the competitive landscape of chiral drug manufacturing.
