Scalable Synthesis of Novel Stilbene Carboxylic Acid Derivatives for Advanced Oncology Applications
Introduction to Patent CN100338008C and Novel Stilbene Derivatives
The pharmaceutical industry is constantly seeking robust synthetic pathways for next-generation antitumor agents, particularly those mimicking the biological activity of natural polyphenols like resveratrol. Patent CN100338008C introduces a significant advancement in this domain by disclosing a series of novel 1-carboxy-1-(3,5-dimethoxyphenyl)-2-(4-R group phenyl) ethylene compounds. These molecules are structurally related to pterostilbene but feature a carboxylic acid moiety that potentially enhances their pharmacokinetic profile and solubility characteristics. The patent outlines a comprehensive methodology for synthesizing these derivatives where the R group can be varied to include hydrogen, hydroxyl, nitro, or alkoxyl functionalities, allowing for a diverse library of candidates for oncology research. This technology represents a strategic shift from relying solely on plant extraction to precise chemical synthesis, offering greater control over impurity profiles and batch-to-batch consistency.
For R&D directors and procurement specialists, the significance of this patent lies in its ability to generate stable, scalable intermediates that serve as precursors for potent antioxidant and anticancer drugs. The described compounds are designed to overcome the metabolic limitations often associated with simple stilbenes, providing a more durable scaffold for drug development. By establishing a reliable synthetic route, manufacturers can secure a steady supply of high-value chemical building blocks essential for preclinical and clinical studies. The following analysis delves into the technical specifics of this synthesis, highlighting its operational feasibility and commercial potential for global supply chains.
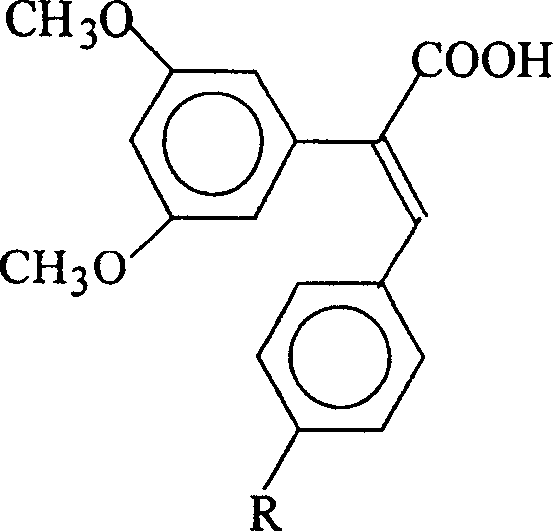
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the sourcing of stilbene-based bioactive compounds has relied heavily on extraction from botanical sources such as grape skins or Polygonum cuspidatum. This conventional approach suffers from inherent volatility in supply chain continuity due to seasonal variations, geographical constraints, and fluctuating agricultural yields. Furthermore, natural extracts often contain complex matrices of impurities that require extensive and costly downstream purification processes to meet pharmaceutical grade standards. The structural diversity achievable through extraction is also limited to what nature provides, restricting medicinal chemists from exploring specific analogues that might possess superior potency or reduced toxicity. Additionally, the isolation of specific isomers, such as the trans-form of resveratrol, can be technically challenging and yield inconsistent results on a commercial scale.
The Novel Approach
The methodology presented in CN100338008C circumvents these biological constraints by employing a fully synthetic organic chemistry route. This novel approach utilizes readily available industrial chemicals like 3,5-dimethoxybenzyl bromide and substituted benzaldehydes as starting materials, ensuring a stable and predictable raw material supply. The synthesis is designed to be modular, allowing for the easy interchange of the para-substituent on the phenyl ring to generate a variety of derivatives without altering the core reaction conditions significantly. This flexibility accelerates the structure-activity relationship (SAR) studies crucial for drug discovery. Moreover, the synthetic pathway avoids the use of exotic catalysts or hazardous high-energy conditions, making it inherently safer and more environmentally compliant than many alternative synthetic strategies.
Mechanistic Insights into the Three-Step Synthetic Sequence
The core of this patented technology is a logical three-step sequence that builds the target stilbene scaffold with high precision. The first stage involves a nucleophilic substitution reaction where 3,5-dimethoxybenzyl bromide reacts with sodium cyanide. This cyanation step extends the carbon chain by one unit, converting the benzyl halide into 3,5-dimethoxybenzeneacetonitrile. This transformation is critical as it installs the nitrile group which serves as the precursor for the carboxylic acid functionality found in the final product. The reaction is typically conducted in a mixed solvent system of water and ethanol at elevated temperatures around 65°C, facilitating the dissolution of inorganic salts and promoting efficient nucleophilic attack.
Following cyanation, the nitrile intermediate undergoes alkaline hydrolysis to yield 3,5-dimethoxyphenylacetic acid. This step utilizes sodium hydroxide under reflux conditions to cleave the carbon-nitrogen triple bond, converting it into a carboxylate salt which is subsequently acidified to precipitate the free acid. The final and most crucial step is a condensation reaction, mechanistically similar to the Perkin reaction, between the newly formed phenylacetic acid and a para-substituted benzaldehyde. In the presence of acetic anhydride and a weak base catalyst (referred to as 'salt of wormwood' or likely potassium acetate in standard chemical contexts), the acid and aldehyde condense to form the double bond characteristic of the stilbene structure. This step creates the conjugated system essential for the molecule's biological activity.
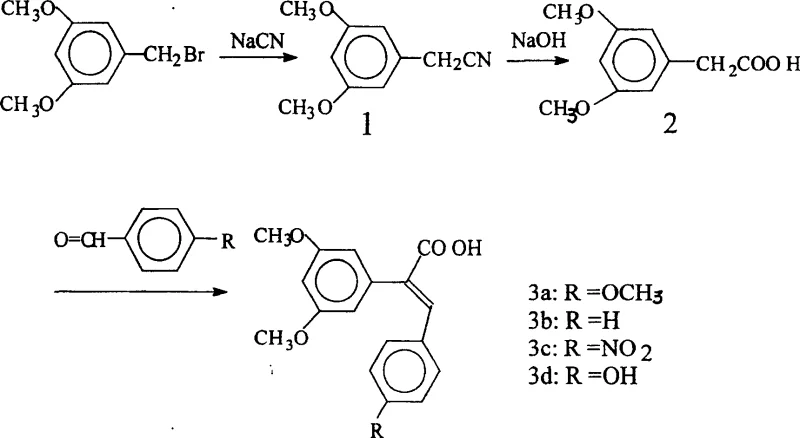
How to Synthesize 1-carboxy-1-(3,5-dimethoxyphenyl)-2-(4-R phenyl) ethylene Efficiently
The synthesis of these valuable pharmaceutical intermediates requires careful control of reaction parameters to maximize yield and purity. The process begins with the preparation of the key acid intermediate, followed by the condensation with various aldehydes to generate the specific derivative required. Operational simplicity is a key feature, as the reactions proceed in common solvents and utilize standard workup procedures like filtration and recrystallization. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Perform cyanation of 3,5-dimethoxybenzyl bromide with sodium cyanide in ethanol/water at 65°C to form 3,5-dimethoxybenzeneacetonitrile.
- Hydrolyze the nitrile intermediate using sodium hydroxide under reflux conditions, followed by acidification to isolate 3,5-dimethoxyphenylacetic acid.
- Conduct a condensation reaction between the phenylacetic acid and para-substituted benzaldehyde using acetic anhydride and a weak base catalyst at 40°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for procurement managers and supply chain heads looking to optimize costs and ensure reliability. The reliance on commodity chemicals rather than scarce natural resources significantly de-risks the supply chain, protecting against market volatility caused by crop failures or geopolitical issues affecting botanical sourcing. The modular nature of the synthesis means that a single production line can be adapted to manufacture multiple variants of the compound simply by changing the aldehyde feedstock, thereby maximizing asset utilization and reducing capital expenditure requirements for dedicated equipment.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive transition metal catalysts or specialized enzymatic systems, relying instead on inexpensive inorganic bases and acetic anhydride. This simplification of the reagent profile directly translates to lower raw material costs and reduced waste disposal expenses associated with heavy metal removal. Furthermore, the use of standard recrystallization techniques for purification avoids the need for costly chromatographic separations, streamlining the production workflow and lowering the overall cost of goods sold.
- Enhanced Supply Chain Reliability: By shifting production from agriculture-dependent extraction to chemical synthesis, manufacturers can guarantee consistent year-round availability of the intermediate. The starting materials, such as benzyl bromides and benzaldehydes, are produced on a massive global scale, ensuring that supply disruptions are minimal. This stability allows for long-term contracting and better inventory planning, which is critical for maintaining uninterrupted drug development timelines and commercial manufacturing schedules.
- Scalability and Environmental Compliance: The reaction conditions are mild, with temperatures generally kept below 100°C, which reduces energy consumption and simplifies reactor engineering requirements. The aqueous workups and alcohol-based recrystallizations generate waste streams that are easier to treat compared to processes involving chlorinated solvents or toxic heavy metals. This alignment with green chemistry principles facilitates regulatory approval and supports corporate sustainability goals, making the process attractive for modern, eco-conscious manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these stilbene derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals.
Q: What is the primary advantage of this synthetic route over natural extraction?
A: This synthetic route allows for the production of specific structural analogues with defined R-groups (such as nitro, methoxy, or hydroxyl) that may offer superior stability and bioavailability compared to naturally occurring resveratrol, ensuring consistent supply independent of agricultural variables.
Q: Are the reaction conditions suitable for large-scale manufacturing?
A: Yes, the process utilizes moderate temperatures ranging from 40°C to 65°C and standard reagents like sodium cyanide and acetic anhydride, avoiding extreme high-pressure or cryogenic conditions that typically hinder commercial scalability.
Q: How is product purity ensured in this protocol?
A: The protocol incorporates multiple recrystallization steps using solvents such as methanol and 95% ethanol at critical stages, effectively removing unreacted starting materials and side products to achieve high-purity crystalline solids suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-carboxy-1-(3,5-dimethoxyphenyl)-2-(4-R phenyl) ethylene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving antitumor therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1-carboxy-1-(3,5-dimethoxyphenyl)-2-(4-R phenyl) ethylene meets the highest international standards for pharmaceutical use.
We invite you to collaborate with us to leverage this innovative synthetic technology for your drug discovery programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your transition from laboratory research to commercial manufacturing.
