Advanced Synthesis of Cyano-Stilbene Derivatives for Oncology Applications
Advanced Synthesis of Cyano-Stilbene Derivatives for Oncology Applications
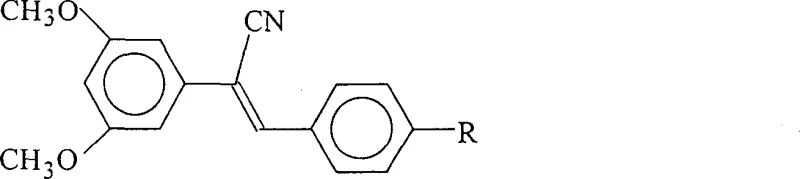
The pharmaceutical industry is constantly seeking novel scaffolds that can overcome the limitations of natural products while retaining their potent biological activities. Patent CN1301246C introduces a significant advancement in this domain by disclosing a series of 1-cyano-1-(3,5-dimethoxyphenyl)-2-(4-R-phenyl)ethylene compounds. These molecules represent a strategic evolution of the stilbene class, specifically designed to mimic and potentially surpass the antitumor and antioxidant capabilities of pterostilbene and resveratrol. By incorporating a cyano group into the ethylene bridge, the inventors have created a new chemical space that offers improved metabolic profiles and enhanced pharmacological potential. This technical insight report analyzes the synthetic methodology and commercial viability of these compounds for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the sourcing of bioactive stilbenes like resveratrol has relied heavily on extraction from natural sources such as grape skins or Polygonum cuspidatum, a process fraught with inconsistency and low yields. Even when synthesized chemically, standard stilbene derivatives often suffer from rapid metabolic clearance in vivo, limiting their therapeutic efficacy despite promising in vitro data. Furthermore, the structural rigidity of the unsubstituted ethylene bridge can sometimes lead to poor solubility characteristics or non-specific binding, which complicates drug formulation and delivery. Conventional synthetic routes often require harsh conditions or expensive transition metal catalysts that introduce heavy metal impurities, necessitating costly purification steps that drive up the final price of the active pharmaceutical ingredient. These factors collectively create a bottleneck for the reliable supply of high-purity stilbene-based therapeutics.
The Novel Approach
The methodology outlined in the patent data presents a streamlined, metal-free alternative that directly addresses these supply chain and efficacy challenges. By utilizing a Knoevenagel condensation strategy between 3,5-dimethoxyphenylacetonitrile and various para-substituted benzaldehydes, the process achieves high atom economy and structural diversity. The introduction of the electron-withdrawing cyano group not only stabilizes the double bond but also provides a handle for further functionalization if needed in downstream medicinal chemistry campaigns. This approach eliminates the need for transition metal catalysts entirely, thereby removing the risk of heavy metal contamination and simplifying the regulatory approval pathway for the resulting intermediates. The versatility of the 'R' group allows for the rapid generation of a library of analogues to optimize potency without altering the core synthetic workflow.
Mechanistic Insights into Knoevenagel Condensation and Cyanation
The synthesis begins with a nucleophilic substitution reaction where 3,5-dimethoxybenzyl bromide reacts with sodium cyanide in a mixed solvent system of water and ethanol. This step proceeds via an SN2 mechanism, where the cyanide ion attacks the benzylic carbon, displacing the bromide leaving group to form 3,5-dimethoxyphenylacetonitrile. The choice of a biphasic or mixed solvent system is critical here, as it ensures the solubility of the inorganic cyanide salt while maintaining the organic substrate in solution, facilitating efficient mass transfer. Temperature control at 65°C is maintained to accelerate the reaction kinetics without promoting the hydrolysis of the nitrile group or the degradation of the methoxy substituents. The resulting nitrile precipitates upon cooling, allowing for a simple filtration that yields a high-purity intermediate ready for the subsequent coupling step.
The second stage involves the base-catalyzed condensation of the newly formed nitrile with a para-substituted benzaldehyde using sodium methylate in methanol. The base deprotonates the alpha-carbon of the nitrile, generating a resonance-stabilized carbanion that acts as a nucleophile. This species attacks the carbonyl carbon of the benzaldehyde, followed by dehydration to establish the conjugated double bond characteristic of the stilbene framework. The use of sodium methylate is particularly advantageous as it is a strong, non-nucleophilic base that drives the equilibrium towards the product without causing side reactions such as ester hydrolysis if ester groups are present on the benzaldehyde. The final product crystallizes directly from the reaction mixture upon cooling, indicating a thermodynamically stable trans-configuration which is essential for biological activity.
How to Synthesize 1-Cyano-1-(3,5-dimethoxyphenyl)-2-phenylethylene Efficiently
The operational simplicity of this two-step sequence makes it an ideal candidate for technology transfer from laboratory to pilot plant. The process relies on standard unit operations such as stirred tank reactors, filtration units, and recrystallization vessels, all of which are ubiquitous in fine chemical manufacturing facilities. The critical parameters to monitor include the stoichiometric ratio of the base to ensure complete conversion of the aldehyde, and the cooling rate during crystallization to control particle size distribution. For detailed standard operating procedures regarding reagent grades, specific agitation speeds, and drying protocols, please refer to the standardized synthesis guide below.
- Perform nucleophilic substitution by reacting 3,5-dimethoxybenzyl bromide with sodium cyanide in an ethanol-water mixture at 65°C to form 3,5-dimethoxyphenylacetonitrile.
- Conduct Knoevenagel condensation by mixing the resulting nitrile with a para-substituted benzaldehyde and sodium methylate in methanol at 60°C.
- Isolate the final cyano-stilbene product by cooling the reaction mixture to room temperature, filtering the precipitated solid, and recrystallizing from methanol.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial advantages over extraction-based methods or complex multi-step syntheses involving precious metals. The reliance on commodity chemicals such as benzyl bromides, sodium cyanide, and benzaldehydes ensures that raw material costs remain low and predictable, shielding the supply chain from the volatility associated with agricultural harvests or rare earth mining. The elimination of chromatographic purification in favor of crystallization significantly reduces solvent consumption and processing time, leading to a drastically simplified manufacturing workflow. This efficiency translates directly into a more competitive cost structure for the final pharmaceutical intermediate, enabling better margin management for downstream drug developers.
- Cost Reduction in Manufacturing: The absence of expensive transition metal catalysts such as palladium or rhodium removes a major cost driver from the bill of materials. Additionally, the avoidance of chromatography means that solvent recovery is more straightforward and energy-intensive distillation columns are not required for product isolation. The high yield of the precipitation step implies that less raw material is wasted, further optimizing the overall material balance and reducing the cost of goods sold significantly.
- Enhanced Supply Chain Reliability: Because all starting materials are bulk industrial chemicals available from multiple global vendors, the risk of single-source supply disruption is minimized. The synthetic nature of the process decouples production from seasonal variations or geopolitical issues affecting botanical sourcing regions. This consistency allows for accurate demand forecasting and the maintenance of strategic inventory levels, ensuring that clinical trial materials and commercial batches can be delivered with short lead times.
- Scalability and Environmental Compliance: The reaction conditions are mild and operate at atmospheric pressure, reducing the safety risks associated with high-pressure hydrogenation or exothermic runaway reactions. The waste stream primarily consists of aqueous salts and recoverable alcohols, which are easier to treat and dispose of compared to heavy metal-laden waste from cross-coupling reactions. This environmental profile facilitates easier permitting for manufacturing sites and aligns with the increasing regulatory pressure for greener pharmaceutical processes.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the stability, scalability, and application of these cyano-stilbene derivatives. Understanding these nuances is crucial for R&D teams evaluating this scaffold for their oncology pipelines. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation.
Q: What is the primary advantage of the cyano-substitution in these stilbene derivatives compared to natural resveratrol?
A: The introduction of the cyano group at the 1-position significantly alters the electronic distribution and steric environment of the stilbene backbone. This modification is designed to enhance metabolic stability and potentially increase binding affinity to biological targets involved in tumor suppression, addressing the limitation of rapid metabolism often seen with natural polyphenols like resveratrol.
Q: How does the patented synthesis method ensure high purity for pharmaceutical applications?
A: The process utilizes a robust precipitation and recrystallization protocol. By leveraging the differential solubility of the product in methanol at varying temperatures, impurities such as unreacted aldehydes or side-products are effectively removed. The use of sodium methylate as a mild base also minimizes harsh degradation pathways, preserving the integrity of the sensitive methoxy groups.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the reaction conditions are highly amenable to scale-up. The use of common solvents like methanol and ethanol, along with readily available starting materials like benzyl bromides and benzaldehydes, eliminates the need for exotic reagents. Furthermore, the isolation method relies on simple filtration rather than complex chromatography, which is a critical factor for cost-effective manufacturing at the metric ton scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Cyano-1-(3,5-dimethoxyphenyl)-2-phenylethylene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of next-generation antitumor agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from preclinical research to market launch is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 1-cyano-1-(3,5-dimethoxyphenyl)-2-phenylethylene meets the highest international standards for pharmaceutical intermediates. Our commitment to quality assurance ensures that the impurity profiles are tightly controlled, supporting your regulatory filings with confidence.
We invite you to collaborate with us to leverage this innovative chemistry for your drug discovery programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your project timelines and reduce overall development costs.
