Advanced Synthesis of Cyano-Substituted Resveratrol Analogues for Oncology Applications
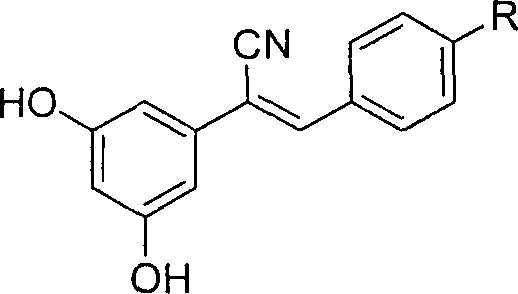
The pharmaceutical industry continuously seeks structural modifications of natural products to enhance therapeutic efficacy while mitigating toxicity, a challenge prominently addressed in patent CN100368388C. This pivotal intellectual property discloses a novel class of cyano-substituted resveratrol derivatives, specifically designed to overcome the limited potency of natural trans-resveratrol in oncology applications. The patent outlines a robust synthetic methodology that transforms simple aromatic precursors into highly active anticancer agents through a streamlined three-step sequence. By introducing a nitrile grouping substituent into the stilbene backbone, the inventors have created compounds that demonstrate superior proliferation inhibition rates against lung carcinoma cell lines compared to the parent molecule. This technological breakthrough represents a significant advancement for reliable pharmaceutical intermediates supplier networks aiming to diversify their oncology portfolios with next-generation candidates. The structural versatility allows for various R-group substitutions, including hydrogen, nitro, hydroxyl, and carboxymethoxyl moieties, enabling fine-tuning of pharmacokinetic properties for specific therapeutic targets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional sourcing of resveratrol relies heavily on extraction from botanical sources such as grape skins or Polygonum cuspidatum, a process inherently plagued by seasonal variability, low yields, and complex purification challenges. Natural extraction often results in batch-to-batch inconsistency regarding impurity profiles, which poses severe risks for regulatory compliance in Good Manufacturing Practice (GMP) environments. Furthermore, native trans-resveratrol suffers from rapid metabolic clearance and relatively weak anticancer potency, necessitating high dosages that can lead to off-target effects. Previous synthetic attempts to modify the stilbene core often involved harsh reaction conditions, expensive transition metal catalysts, or multi-step protection-deprotection sequences that drastically increased the cost of goods sold. These conventional pathways frequently struggled with stereochemical control, producing mixtures of E and Z isomers that required difficult chromatographic separation, thereby reducing overall process efficiency and scalability for commercial manufacturing.
The Novel Approach
The methodology described in CN100368388C circumvents these historical bottlenecks by employing a direct and atom-economical route centered on the Knoevenagel condensation reaction. This innovative approach utilizes readily available commodity chemicals like 3,5-dimethoxybenzyl bromide and substituted benzaldehydes, ensuring a stable and cost-effective supply chain foundation. The process achieves high stereoselectivity for the Z-isomer directly during the condensation step, eliminating the need for downstream isomer separation and significantly simplifying the purification workflow. By integrating a nucleophilic substitution to install the nitrile group early in the sequence, the synthesis builds molecular complexity efficiently without requiring exotic reagents. The final demethylation step uses boron tribromide under mild conditions, preserving the sensitive vinyl cyanide motif while revealing the crucial phenolic hydroxyl groups required for biological activity. This strategic design ensures cost reduction in pharmaceutical intermediates manufacturing by minimizing unit operations and maximizing yield at each stage.
Mechanistic Insights into Knoevenagel Condensation and Demethylation
The core of this synthetic strategy lies in the precise execution of the Knoevenagel condensation between 3,5-dimethoxyphenylacetonitrile and para-substituted benzaldehydes. In this mechanism, sodium methylate acts as a base to deprotonate the active methylene group of the nitrile, generating a resonance-stabilized carbanion that attacks the carbonyl carbon of the aldehyde. The subsequent elimination of water drives the equilibrium toward the formation of the conjugated vinyl cyanide system. The reaction conditions, specifically maintained between 45°C and 60°C in methanol, are critical for favoring the Z-configuration, likely due to steric interactions and thermodynamic stability during the elimination phase. This level of control is essential because the biological activity of stilbene derivatives is often highly dependent on their geometric configuration, with the Z-isomer frequently exhibiting distinct binding affinities to cellular targets compared to the E-isomer. The use of methanol as a solvent not only facilitates the dissolution of reactants but also participates in the proton transfer steps necessary for the catalytic cycle to proceed smoothly without side reactions.
Following the construction of the stilbene backbone, the demethylation step serves as the final activation of the pharmacophore. The patent specifies the use of boron tribromide (BBr3) in anhydrous dichloromethane, a reagent choice that offers superior selectivity for cleaving aryl methyl ethers compared to alternatives like hydroiodic acid or aluminum chloride. The mechanism involves the coordination of the Lewis acidic boron atom to the ether oxygen, followed by nucleophilic attack by bromide ions to release methyl bromide and generate the phenolic hydroxyl groups. Conducting this reaction at room temperature for 24 hours ensures complete conversion while preventing potential degradation of the electron-rich aromatic rings or the nitrile functionality. This mild condition is particularly advantageous for scaling up, as it reduces energy consumption and minimizes the formation of thermal decomposition byproducts. The resulting crude product is then subjected to a simple aqueous quench and recrystallization, yielding high-purity materials suitable for preclinical evaluation without the need for extensive chromatographic purification.
How to Synthesize Cyano-Substituted Resveratrol Efficiently
The synthesis of these high-value anticancer intermediates follows a logical progression designed for reproducibility and safety in a pilot or production plant setting. The process begins with the preparation of the key nitrile building block, followed by the condensation with various aldehydes to introduce diversity, and concludes with the global deprotection of the phenolic groups. Each step has been optimized in the patent examples to provide clear guidance on stoichiometry, temperature profiles, and workup procedures, ensuring that technical teams can replicate the results with high confidence. The detailed standardized synthesis steps below outline the specific operational parameters required to achieve the reported yields and purity levels, serving as a foundational protocol for process development teams looking to implement this technology.
- Perform nucleophilic substitution between 3,5-dimethoxybenzyl bromide and sodium cyanide in ethanol at 65°C to generate 3,5-dimethoxyphenylacetonitrile.
- Execute Knoevenagel condensation using the nitrile intermediate and para-substituted benzaldehyde with sodium methylate catalyst in methanol at 45-60°C.
- Conduct demethylation using boron tribromide (BBr3) in dichloromethane at room temperature for 24 hours, followed by aqueous quenching and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial strategic benefits by relying entirely on bulk commodity chemicals that are widely available from multiple global vendors. The starting materials, such as 3,5-dimethoxybenzyl bromide and various substituted benzaldehydes, are established industrial intermediates with mature supply chains, effectively eliminating the risk of raw material shortages that often plague specialty syntheses. The avoidance of precious metal catalysts like palladium or rhodium not only reduces the direct material cost but also simplifies the regulatory burden associated with heavy metal residue testing in the final Active Pharmaceutical Ingredient (API). Furthermore, the use of common solvents like ethanol, methanol, and dichloromethane allows for efficient solvent recovery and recycling systems, contributing to a more sustainable and economically viable manufacturing process. These factors collectively drive significant cost reduction in pharmaceutical intermediates manufacturing, making the commercial production of these derivatives financially attractive compared to extraction-based or complex catalytic methods.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and chromatographic purification steps drastically lowers the operational expenditure per kilogram of product. By utilizing a precipitation-based isolation method where solids separate directly from the reaction mixture, the process minimizes solvent usage and energy-intensive distillation requirements. This streamlined workflow translates into a leaner cost structure, allowing for competitive pricing strategies in the global market for oncology research compounds. Additionally, the high yields reported in the patent examples indicate efficient atom economy, meaning less waste is generated per unit of product, further enhancing the overall economic efficiency of the production line.
- Enhanced Supply Chain Reliability: The reliance on non-proprietary, off-the-shelf reagents ensures that production schedules are not held hostage by single-source suppliers or geopolitical disruptions affecting rare earth elements. The robustness of the reaction conditions, which tolerate standard industrial equipment materials, means that manufacturing can be easily transferred between different facilities without requiring specialized hardware investments. This flexibility is crucial for maintaining supply continuity, especially when scaling up from laboratory grams to multi-ton annual production volumes to meet clinical trial demands. The simplified purification protocol also reduces the lead time for batch release, enabling faster response to market fluctuations and urgent customer requests for high-purity materials.
- Scalability and Environmental Compliance: The process operates at moderate temperatures and atmospheric pressure, reducing the safety risks associated with high-pressure hydrogenation or cryogenic reactions. The waste streams generated are primarily composed of standard organic solvents and inorganic salts, which can be treated using conventional wastewater treatment infrastructure found in most modern chemical parks. The absence of persistent organic pollutants or toxic heavy metals in the waste profile simplifies environmental permitting and compliance reporting. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturing entity, appealing to environmentally conscious partners and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these cyano-substituted resveratrol analogues. These answers are derived directly from the experimental data and technical specifications provided in the source patent documentation, ensuring accuracy and relevance for decision-makers evaluating this technology for their pipeline. Understanding these nuances is critical for assessing the feasibility of integrating these intermediates into broader drug discovery programs.
Q: What are the primary advantages of cyano-substituted resveratrol over natural trans-resveratrol?
A: According to patent CN100368388C, these derivatives exhibit significantly enhanced antitumor activity against lung carcinoma cells while maintaining lower toxicity profiles compared to the natural parent compound, addressing the potency limitations of standard resveratrol.
Q: Is the demethylation process scalable for industrial production?
A: Yes, the process utilizes boron tribromide in dichloromethane under mild room temperature conditions for 24 hours, avoiding extreme thermal stress and allowing for straightforward scale-up with standard stainless steel reactors and established safety protocols.
Q: How is the Z-isomer selectivity achieved in this synthesis?
A: The synthetic route employs a specific Knoevenagel condensation protocol using sodium methylate in methanol at controlled temperatures (45-60°C), which favors the formation of the thermodynamically stable Z-configuration vinyl cyanide structure essential for biological activity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyano Resveratrol Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic pathway detailed in CN100368388C for developing next-generation antitumor agents. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to clinical supply is seamless and compliant. Our state-of-the-art facilities are equipped to handle the specific reagents and conditions required for this chemistry, including safe handling of boron tribromide and efficient solvent recovery systems. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of cyano-substituted resveratrol derivative meets the exacting standards required for pharmaceutical research and development.
We invite you to collaborate with us to leverage this advanced technology for your oncology projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing expertise can accelerate your path to market with high-quality, cost-effective intermediates.
