Revolutionizing Rosuvastatin Intermediate Production with Mild Phase-Transfer Catalysis
The pharmaceutical industry continuously seeks robust synthetic routes for high-value statins, particularly rosuvastatin calcium, a potent HMG-CoA reductase inhibitor widely prescribed for cardiovascular disease management. A critical bottleneck in the supply chain has historically been the efficient production of its key olefinic intermediate. Patent CN112521375A introduces a transformative preparation method that addresses long-standing challenges in yield, purity, and operational safety. This innovation shifts the paradigm from hazardous cryogenic chemistry to a mild, phase-transfer catalyzed protocol. By leveraging specific additives like tri-(3,6-dioxaheptyl) amine (TDA-1) and crown ethers, the process achieves exceptional stereocontrol and yield without the need for ultra-low temperatures. For global procurement teams and R&D directors, this represents a significant opportunity to optimize the supply of high-purity pharmaceutical intermediates while drastically reducing manufacturing complexity and environmental impact.
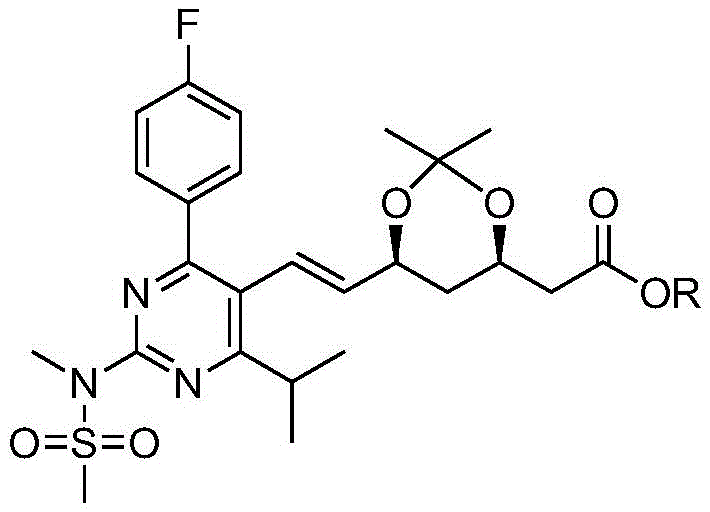
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the rosuvastatin skeleton has relied heavily on classical Wittig condensation reactions, as documented in early patents such as WO0185702 assigned to AstraZeneca. These conventional routes typically involve the reaction of a phosphonium salt with an aldehyde side chain under extremely harsh conditions. A major drawback is the requirement for cryogenic temperatures, often necessitating cooling to -78°C using dry ice or liquid nitrogen baths. Furthermore, these processes frequently employ highly reactive and pyrophoric bases such as n-butyl lithium or sodium bis(trimethylsilyl)amide (NaHMDS). Such reagents pose severe safety risks, including fire hazards and the need for specialized inert atmosphere handling equipment. Additionally, traditional methods often suffer from poor stereoselectivity, generating significant amounts of the unwanted Z-type isomer alongside the desired E-isomer. The separation of these geometric isomers is notoriously difficult and costly, often requiring complex chromatography or repeated crystallizations, which drives down the overall yield to levels as low as 34% in some reported variations. The generation of stoichiometric amounts of phosphine oxide by-products further complicates downstream purification and waste management.
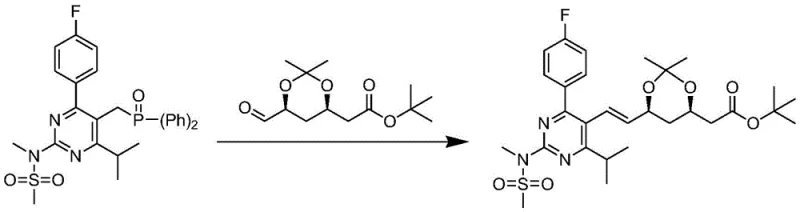
The Novel Approach
In stark contrast, the methodology disclosed in CN112521375A offers a streamlined and economically superior alternative. This novel approach utilizes a modified Wittig-type olefination that operates under remarkably mild thermal conditions, specifically between 30°C and 35°C. Instead of dangerous organolithium reagents, the process employs inexpensive and safe inorganic bases like potassium carbonate. The true innovation lies in the synergistic use of phase-transfer catalysts, specifically tri-(3,6-dioxaheptyl) amine (TDA-1) combined with crown ethers such as 18-crown-6. This catalytic system facilitates the deprotonation of the phosphonium salt in the organic phase, generating the reactive ylide species efficiently without extreme cold. Crucially, this specific combination of additives effectively suppresses the formation of Z-type isomers, ensuring high stereochemical purity of the desired E-alkene product. The result is a process that not only simplifies the operational workflow by eliminating cryogenic infrastructure but also boosts the isolated yield to over 86%, with purity levels exceeding 99.7%. This represents a substantial leap forward in process chemistry, aligning perfectly with the principles of green chemistry and industrial scalability.
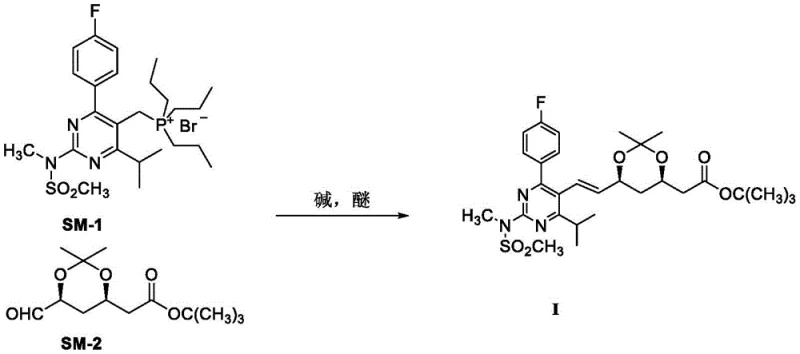
Mechanistic Insights into Phase-Transfer Catalyzed Olefination
The success of this novel synthesis hinges on the sophisticated interplay between the phase-transfer catalysts and the reaction medium. In traditional heterogeneous systems, the inorganic base (potassium carbonate) and the organic phosphonium salt reside in different phases, leading to slow reaction kinetics and incomplete conversion. The addition of 18-crown-6 ether acts as a complexing agent for the potassium cations. By encapsulating the potassium ion within its hydrophobic cavity, the crown ether renders the associated carbonate anion "naked" and highly nucleophilic within the organic solvent, typically toluene. This dramatically increases the basicity of the carbonate in the organic phase, allowing it to effectively abstract the acidic proton from the phosphonium salt (SM-1) to form the phosphorus ylide. Simultaneously, TDA-1, a lipophilic amine, further enhances the solubility of the ionic species in the organic medium. This dual-catalyst system ensures a high concentration of the active ylide species is maintained at mild temperatures. The controlled generation of the ylide is critical for stereoselectivity; rapid or uncontrolled ylide formation at low temperatures often favors kinetic products (Z-isomers), whereas the thermodynamic control afforded by this mild, catalyzed system favors the more stable E-isomer configuration required for biological activity.
Furthermore, the choice of the tributyl phosphonium group in SM-1, as opposed to the triphenyl groups used in many prior art references, plays a pivotal role in impurity control. The background art notes that triphenylphosphine-based routes can generate difficult-to-remove by-products like diphenyl metaphosphoric acid derivatives. The tributyl variant utilized in this patent produces tributylphosphine oxide as a by-product, which generally exhibits different solubility characteristics and is easier to separate from the polar product during the aqueous workup and crystallization steps. The reaction mechanism proceeds through a betaine intermediate which collapses to form the alkene and the phosphine oxide. The specific solvent system, preferably toluene, aids in this separation by allowing the product to crystallize out upon cooling while keeping impurities in solution. This mechanistic understanding underscores why the process achieves such high purity (99.70% - 99.90%) without the need for extensive chromatographic purification, making it ideally suited for GMP manufacturing environments where impurity profiles must be strictly controlled.
How to Synthesize Rosuvastatin Calcium Intermediate Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of scale-up. The procedure begins by establishing an inert atmosphere, typically using nitrogen, to prevent moisture interference which could quench the ylide. The phosphonium salt SM-1 is dissolved in the chosen organic solvent, followed by the sequential addition of the base and the catalytic system. It is critical to allow sufficient stirring time (approximately 30 minutes) for the catalyst-base complex to form before introducing the aldehyde component SM-2. The aldehyde is added slowly to manage the exotherm and maintain the optimal temperature window of 30-35°C. Following the reaction completion, confirmed by TLC, the workup involves simple filtration to remove inorganic salts, concentration, and a straightforward crystallization from toluene. This simplicity stands in contrast to the multi-step extractions and column chromatography often required by older methods. For detailed laboratory protocols and precise stoichiometric ratios, please refer to the standardized synthesis guide below.
- Dissolve the phosphonium salt precursor (SM-1) in an organic solvent such as toluene under inert gas protection.
- Add a mild inorganic base like potassium carbonate along with tri-(3,6-dioxaheptyl) amine (TDA-1) and 18-crown-6 ether to the mixture.
- Slowly introduce the aldehyde side chain (SM-2) while maintaining the temperature between 30-35°C to ensure high E-isomer selectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates into tangible strategic benefits beyond mere chemical yield. The elimination of cryogenic requirements removes a significant capital expenditure barrier, as facilities no longer need specialized -78°C reactors or the continuous supply of dry ice and liquid nitrogen. This drastically simplifies the manufacturing infrastructure and reduces energy consumption. Moreover, the substitution of hazardous pyrophoric reagents like n-butyl lithium with stable potassium carbonate significantly lowers insurance premiums and safety compliance costs. The process generates less hazardous waste, aligning with increasingly stringent environmental regulations and reducing disposal fees. From a supply continuity perspective, the reagents used—toluene, potassium carbonate, and crown ethers—are commodity chemicals with robust global supply chains, mitigating the risk of raw material shortages that often plague specialty reagent-dependent processes. The high yield and purity directly reduce the cost of goods sold (COGS) by minimizing material loss and maximizing throughput per batch.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the removal of expensive and energy-intensive unit operations. By operating at near-ambient temperatures (30-35°C), the process eliminates the massive energy load associated with maintaining cryogenic conditions over long reaction times. The use of cheap, non-hazardous bases like potassium carbonate instead of premium-priced organometallic reagents results in substantial raw material cost savings. Furthermore, the high selectivity for the E-isomer minimizes the loss of valuable starting materials to the wrong stereoisomer, effectively increasing the atom economy of the process. The simplified purification strategy, relying on crystallization rather than chromatography, reduces solvent consumption and processing time, leading to a leaner, more cost-efficient production cycle that enhances overall profit margins for the final API.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available, non-controlled reagents. Unlike n-butyl lithium, which requires special shipping and storage protocols due to its pyrophoric nature, potassium carbonate and crown ethers can be sourced from multiple vendors globally without regulatory hurdles. This diversification of the supplier base reduces the risk of single-source bottlenecks. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in ambient temperature or humidity, leading to more consistent batch-to-batch quality and fewer failed campaigns. This reliability ensures a steady flow of intermediates to downstream API manufacturers, preventing production delays and ensuring that market demand for rosuvastatin calcium can be met consistently without interruption.
- Scalability and Environmental Compliance: Scaling chemical processes from the lab to the plant often reveals hidden thermal hazards, but this method is inherently safe for scale-up. The mild exotherm and lack of unstable intermediates mean that heat removal systems do not need to be oversized, allowing for larger batch sizes in standard reactors. Environmentally, the process is cleaner; it avoids the generation of heavy metal waste or difficult-to-treat organolithium quench by-products. The primary waste streams are aqueous salts and organic solvents that can be readily recovered and recycled. This "green" profile facilitates easier permitting for new manufacturing lines and supports corporate sustainability goals. The ability to run this process in common solvents like toluene, which has well-established recovery infrastructure in most chemical plants, further streamlines the path to commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method. They are derived from the specific advantages outlined in the patent literature and are intended to clarify the operational benefits for potential partners. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production lines. The answers reflect the consensus on best practices for implementing phase-transfer catalyzed olefinations in a GMP setting.
Q: How does the new method improve stereoselectivity compared to traditional Wittig reactions?
A: By utilizing specific phase-transfer catalysts like TDA-1 and crown ethers, the reaction suppresses the formation of Z-type isomers without requiring cryogenic temperatures (-78°C), achieving high E-isomer purity directly.
Q: What are the safety advantages of using potassium carbonate over n-butyl lithium?
A: Potassium carbonate is a stable, non-pyrophoric solid that eliminates the severe fire hazards and specialized handling equipment associated with pyrophoric bases like n-butyl lithium, significantly enhancing operational safety.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process operates at mild temperatures (30-35°C) using common solvents like toluene and avoids ultra-low temperature infrastructure, making it highly scalable and cost-effective for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Calcium Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to production requires more than just a recipe; it demands deep process engineering expertise. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of this mild Wittig protocol, ensuring that the stringent purity specifications demanded by global regulatory bodies are met consistently. Our rigorous QC labs utilize advanced HPLC methods to monitor the E/Z ratio and impurity profiles at every stage, guaranteeing that every batch of rosuvastatin intermediate delivered meets the highest standards of quality and safety. We are committed to being a reliable rosuvastatin calcium intermediate supplier that bridges the gap between innovative chemistry and commercial reality.
We invite pharmaceutical companies and generic manufacturers to collaborate with us to leverage this cost-effective synthesis route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments. Let us help you secure a competitive advantage in the statin market through superior supply chain efficiency and uncompromising quality.
