Advanced Synthesis of C14 Aldehyde: Enhancing Purity and Scalability for Vitamin A Production
Introduction to Advanced Vitamin A Intermediate Synthesis
The global demand for Vitamin A acetate, a critical nutrient for visual health, growth, and epithelial tissue maintenance, continues to drive innovation in fine chemical manufacturing. As detailed in patent CN111484400A, a novel preparation method for 2-methyl-4-(2,6,6-trimethylcyclohexen-1-yl)-2-butenal, commonly known as C14 aldehyde, has emerged as a transformative technology for the industry. This key intermediate serves as the foundational building block for the subsequent synthesis of Vitamin A acetate, influencing the overall efficiency and cost structure of the final API production. The structural complexity of Vitamin A acetate necessitates precise synthetic strategies to ensure high purity and yield.
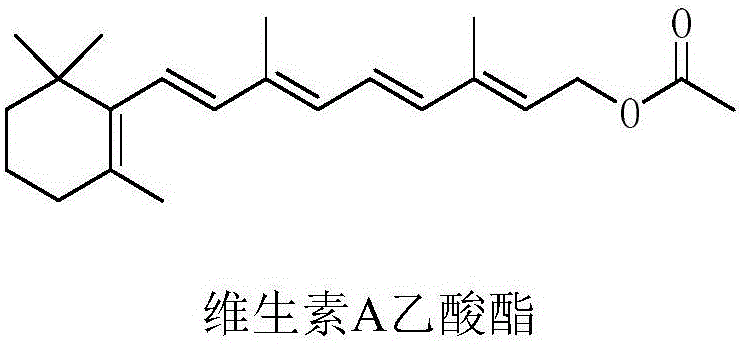
Traditional manufacturing pathways have long struggled with issues related to safety, environmental impact, and raw material costs. The new methodology described in the patent addresses these pain points by introducing a streamlined route that bypasses the need for expensive beta-ionone or hazardous acetylene gas. By leveraging advanced organic synthesis techniques involving acetal protection and classical coupling reactions, this process offers a robust alternative for reliable pharmaceutical intermediates supplier networks seeking to optimize their supply chains. The ability to produce high-purity C14 aldehyde with minimal impurities is paramount for downstream applications in medicine, food additives, and cosmetics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Vitamin A acetate has relied on three primary technical routes, each carrying significant drawbacks that hinder optimal cost reduction in vitamin manufacturing. The Roche C14+C6 route, for instance, involves a cumbersome six-step sequence starting from beta-ionone, including Darzens condensation and Grignard reactions, which often suffer from low atom economy and complex purification requirements. Similarly, the Rhone-Poulenc route necessitates harsh high-vacuum molecular distillation to purify key intermediates like C15 aldehyde and C18 ketone, a process that is energy-intensive and difficult to scale efficiently for large-volume production.
Furthermore, the BASF C15+C5 route, while shorter, imposes severe operational constraints such as the requirement for ultra-low temperatures and strictly anhydrous conditions during ethynylation. The use of lithium amide and acetylene gas in alternative acetylene-based routes introduces substantial safety risks and environmental hazards, including the generation of malodorous byproducts like dimethyl sulfide in sulfur ylide methods. These legacy processes often result in isomer mixtures that require extensive separation, thereby increasing lead time for high-purity intermediates and inflating the overall cost of goods sold. The reliance on expensive starting materials like beta-ionone further exacerbates the economic burden on manufacturers.
The Novel Approach
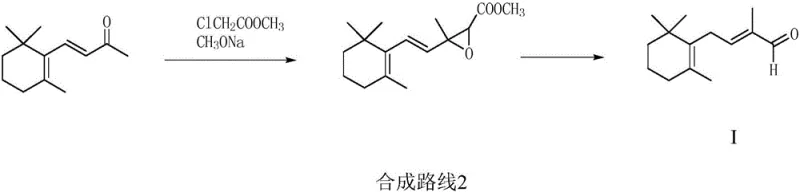
In stark contrast to these legacy methods, the invention disclosed in CN111484400A presents a groundbreaking strategy that utilizes 2-methyl-4-halogeno-2-butenal acetal as a primary raw material. This approach fundamentally shifts the synthetic paradigm by employing a protective group strategy that stabilizes the reactive aldehyde moiety during the critical carbon-carbon bond-forming steps. The process involves reacting the halogenated acetal with magnesium powder to form a Grignard reagent, or alternatively with triphenylphosphine or triester phosphite to generate Wittig or Horner-Wadsworth-Emmons reagents. This flexibility allows manufacturers to choose the most cost-effective and operationally simple pathway based on their existing infrastructure.
The subsequent reaction with 2,2,6-trimethylcyclohexanone proceeds under mild conditions, avoiding the extreme temperatures and pressures associated with older technologies. The final deprotection step under acidic conditions cleanly yields the target C14 aldehyde with exceptional purity, often exceeding 99% as demonstrated in the patent examples. This novel route not only eliminates the need for hazardous acetylene handling but also utilizes cheap and easily obtainable raw materials, significantly lowering the barrier to entry for commercial scale-up of complex intermediates. The high selectivity of the reaction minimizes the formation of isomers, simplifying the downstream purification process and enhancing overall process sustainability.
Mechanistic Insights into Acetal-Protected Coupling Reactions
The core mechanistic advantage of this synthesis lies in the strategic use of the acetal functional group to mask the electrophilic aldehyde carbon. In conventional unprotected aldehyde syntheses, the presence of the carbonyl group often leads to competitive side reactions, such as self-condensation or polymerization, especially in the presence of strong bases or nucleophiles. By converting the aldehyde into a dimethyl or ethylene acetal (Compound II), the molecule becomes inert to nucleophilic attack at the carbonyl position, allowing the halogenated tail to selectively undergo metallation or phosphonium salt formation. This chemoselectivity is crucial for maintaining high reaction atom economy and ensuring that the stoichiometry of the reagents is utilized effectively for the desired C-C bond formation.
Once the nucleophilic species (Compound III), whether it be a Grignard reagent, a phosphonium ylide, or a phosphonate carbanion, is generated, it attacks the ketone carbonyl of 2,2,6-trimethylcyclohexanone (Compound IV). The steric environment of the cyclohexanone ring dictates the stereochemical outcome, but the robust nature of the acetal group ensures that the newly formed alcohol intermediate remains stable until the final workup. The final acidification step serves a dual purpose: it quenches any remaining basic reagents and catalyzes the hydrolysis of the acetal back to the aldehyde. This one-pot deprotection and isolation strategy streamlines the workflow, reducing the number of unit operations and solvent exchanges required, which is a key factor in reducing waste generation and improving the environmental profile of the manufacturing process.
How to Synthesize 2-Methyl-4-(2,6,6-trimethylcyclohexen-1-yl)-2-butenal Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology in a pilot or production setting. The process begins with the activation of magnesium powder or the quaternization of phosphine, followed by the controlled addition of the halogenated acetal. Temperature control is critical during the exothermic formation of the organometallic or phosphorus species, with optimal ranges identified between 30°C and 50°C for Grignard formation. The subsequent coupling with the ketone is performed at even lower temperatures, typically between 10°C and 30°C, to maximize selectivity and minimize side products. Detailed standardized synthesis steps see the guide below.
- React 2-methyl-4-halogeno-2-butenal acetal with magnesium powder, triphenylphosphine, or triester phosphite to generate the corresponding nucleophilic reagent.
- Couple the generated reagent with 2,2,6-trimethylcyclohexanone under controlled temperature conditions to form the protected intermediate.
- Perform acidification and deprotection under acidic conditions to remove the acetal group and yield the target C14 aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this new synthetic route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The shift away from beta-ionone, a commodity subject to price volatility and supply constraints, towards simpler petrochemical-derived precursors like 2-methyl-4-halogeno-2-butenal acetal creates a more resilient supply base. This diversification of raw material sources mitigates the risk of production stoppages due to upstream shortages, ensuring a more consistent flow of critical intermediates to downstream API manufacturers. Furthermore, the elimination of hazardous gases like acetylene reduces the regulatory burden and insurance costs associated with operating high-risk chemical plants.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive starting materials and the high efficiency of the reaction steps. By avoiding the multi-step sequences and complex distillations required by the Roche and Rhone-Poulenc routes, manufacturers can significantly reduce utility consumption and labor costs. The high yield reported in the patent examples, reaching up to 98% in optimized conditions, directly translates to lower raw material consumption per kilogram of product. Additionally, the ability to recover and reuse solvents like tetrahydrofuran and dichloromethane further enhances the cost-effectiveness of the operation, making it a superior choice for cost reduction in vitamin manufacturing.
- Enhanced Supply Chain Reliability: The operational simplicity of this method, which does not require cryogenic conditions or specialized high-pressure equipment, allows for broader adoption across different manufacturing sites. This flexibility increases the total addressable supply capacity in the market, reducing the likelihood of bottlenecks. The robustness of the reaction conditions means that production schedules are less susceptible to delays caused by equipment maintenance or stringent environmental controls needed for more hazardous processes. Consequently, partners can expect more reliable delivery timelines and greater consistency in product availability, which is essential for maintaining uninterrupted pharmaceutical production lines.
- Scalability and Environmental Compliance: From a sustainability perspective, this route aligns perfectly with modern green chemistry principles. The high atom economy ensures that a maximum proportion of the reactant mass ends up in the final product, minimizing waste generation. The absence of heavy metal catalysts and the reduction in hazardous byproducts simplify wastewater treatment and disposal procedures. This environmental friendliness facilitates easier permitting and compliance with increasingly strict global environmental regulations. The process is inherently scalable, allowing for seamless transition from laboratory benchtop to multi-ton commercial production without the need for fundamental process re-engineering, thus supporting the commercial scale-up of complex intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing portfolios. The answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making.
Q: What are the safety advantages of this new C14 aldehyde synthesis route compared to traditional methods?
A: Unlike traditional routes that require hazardous acetylene gas and lithium amide at ultra-low temperatures, this method utilizes stable halogenated acetals and operates at moderate temperatures (10-70°C), significantly improving operational safety and environmental compliance.
Q: How does the acetal protection strategy improve product purity?
A: The use of an acetal protecting group prevents the aldehyde functionality from participating in unwanted side reactions, such as self-condensation, during the Grignard or Wittig coupling steps, resulting in higher selectivity and easier purification.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process features high atom economy, uses cheap and readily available raw materials like 2,2,6-trimethylcyclohexanone, and avoids complex vacuum distillation steps, making it highly scalable for green industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-4-(2,6,6-trimethylcyclohexen-1-yl)-2-butenal Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates in the global vitamin supply chain. Our team of expert chemists has extensively evaluated the technology described in CN111484400A and possesses the technical capability to execute this advanced synthesis with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of C14 aldehyde meets the highest industry standards for pharmaceutical and feed additive applications.
We invite you to collaborate with us to leverage this innovative technology for your Vitamin A production requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how this greener route can optimize your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to drive efficiency and quality in your manufacturing operations.
