Advanced Catalytic Synthesis of 2-Hydrazinopyridine Derivatives for Commercial Scale Production
Advanced Catalytic Synthesis of 2-Hydrazinopyridine Derivatives for Commercial Scale Production
The global demand for high-purity heterocyclic intermediates continues to surge, driven by the expanding pharmaceutical and agrochemical sectors. A pivotal advancement in this domain is detailed in patent CN106588758B, which discloses a robust synthesis process for 2-hydrazinopyridine derivatives. This technology represents a significant departure from legacy manufacturing methods, introducing a streamlined catalytic hydrogen substitution pathway that addresses critical bottlenecks in yield and environmental compliance. For R&D directors and procurement strategists, understanding this shift is essential for securing a reliable agrochemical intermediate supplier capable of meeting stringent quality specifications. The process leverages a sophisticated dual-catalyst system and a specialized solvent environment to achieve exceptional selectivity, positioning it as a cornerstone for modern fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of key intermediates like 3-chloro-2-hydrazinopyridine has relied on cumbersome multi-step sequences that are increasingly untenable in a green chemistry landscape. Traditional routes often commence with the Hofmann degradation of nicotinamide to yield 3-aminopyridine, followed by a series of hazardous transformations including chlorination, diazotization, and Sandmeyer reactions. These legacy processes are plagued by inherently low overall yields due to the accumulation of losses at each discrete step. Moreover, the diazotization and subsequent substitution steps generate voluminous amounts of acidic waste liquids and solid residues, creating severe environmental burdens and escalating disposal costs. The reliance on such inefficient pathways restricts the ability to scale production economically, making it difficult for manufacturers to offer cost reduction in pharmaceutical intermediates manufacturing without compromising on purity or safety standards.
The Novel Approach
In stark contrast, the novel approach outlined in the patent utilizes a direct hydrogen substitution reaction on a precursor compound, such as 2,3,6-trichloropyridine, to generate the necessary pyridine halide intermediate. This method bypasses the need for diazotization entirely, collapsing multiple synthetic steps into a single, highly efficient catalytic transformation. By employing a mixed catalyst system alongside a controlled dual-base environment, the process achieves remarkable selectivity, minimizing the formation of dehalogenated byproducts that typically plague hydrogenolysis reactions. The subsequent nucleophilic substitution with hydrazine hydrate is conducted in a specialized alcohol amine solvent that acts as an acid scavenger, further driving the reaction to completion. This streamlined architecture not only simplifies the operational workflow but also drastically reduces the generation of hazardous waste, aligning perfectly with modern sustainability mandates.
Mechanistic Insights into Catalytic Hydrogen Substitution and Hydrazinolysis
The core innovation of this synthesis lies in the precise orchestration of the hydrogen substitution reaction, where a precursor compound bearing multiple halogen atoms is selectively reduced. The patent specifies the use of a mixed catalyst comprising Palladium on Carbon (Pd/C) and Platinum on Carbon (Pt/C), which exhibits superior performance compared to single-metal catalysts. This synergistic effect is critical because halide ions, particularly chloride, can poison standard catalysts; the mixed system maintains high activity and selectivity even in the presence of these potential inhibitors. Concurrently, the reaction employs a composite base system involving both a weak organic base (pyridine) and a strong inorganic base (sodium hydroxide). Pyridine acts as a buffer to maintain an initial optimal pH, while sodium hydroxide is added dropwise to neutralize the hydrogen halide produced in situ, preventing the system from becoming too acidic which would deactivate the catalyst, or too alkaline which could introduce impurities.
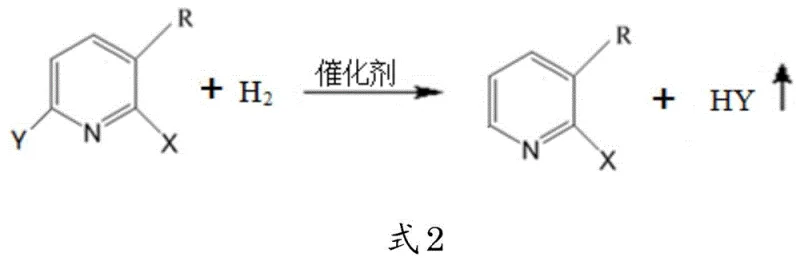
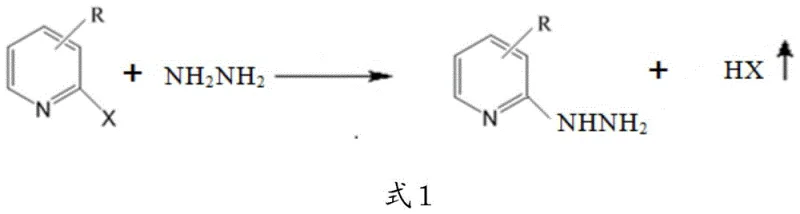
Following the formation of the pyridine halide intermediate, the process proceeds to the hydrazinolysis step, where the halogen atom at the 2-position is displaced by a hydrazine group. This nucleophilic aromatic substitution is facilitated by the use of N,N-dimethylpropanolamine as the reaction solvent. Unlike conventional solvents, this alcohol amine serves a dual purpose: it provides excellent solubility for both the organic substrate and the aqueous hydrazine hydrate, and its tertiary amine functionality acts as an internal acid binding agent. This effectively traps the hydrogen halide byproduct (HX) as it forms, shifting the chemical equilibrium towards the product side according to Le Chatelier's principle. The reaction is typically conducted at elevated temperatures between 100-150 °C under an inert nitrogen atmosphere to prevent oxidation of the hydrazine and ensure operator safety.
How to Synthesize 3-Chloro-2-hydrazinopyridine Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry and sequence of reagent addition to maximize the benefits of the catalytic system. The process begins with the hydrogenation of the trichloropyridine precursor, where the ratio of the mixed catalyst to the substrate is strictly controlled to ensure complete conversion without over-reduction. Following the isolation of the dichloropyridine intermediate, the hydrazinolysis step demands a molar excess of hydrazine hydrate, typically in a ratio of 1:1.5 to 1:1.8 relative to the halide, to drive the substitution to completion while minimizing side reactions with other functional groups. Detailed standardized operating procedures regarding temperature ramping, pressure control, and work-up protocols are essential for reproducibility.
- Perform hydrogen substitution on a precursor compound (e.g., 2,3,6-trichloropyridine) using a mixed Pd/C and Pt/C catalyst with a dual-base system (pyridine and sodium hydroxide) to generate pyridine halide A.
- React the resulting pyridine halide A with hydrazine hydrate in a solvent such as N,N-dimethylpropanolamine at 100-150°C under an inert atmosphere.
- Execute post-treatment including cooling crystallization, centrifugation, washing, and drying to isolate the final 2-hydrazinopyridine derivative product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this catalytic methodology offers profound strategic advantages beyond mere technical elegance. The elimination of the diazotization and Sandmeyer steps removes significant logistical complexities associated with handling unstable diazonium salts and managing large volumes of corrosive waste streams. This simplification translates directly into a more resilient supply chain, as the process relies on stable, commercially available starting materials like 2,3,6-trichloropyridine rather than custom-synthesized amines that may have limited supplier bases. Furthermore, the high selectivity of the mixed catalyst system reduces the burden on downstream purification units, allowing for faster batch turnover and improved asset utilization in manufacturing facilities.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of the synthetic route and the recovery of valuable materials. By avoiding the multi-step degradation and substitution sequence, manufacturers save significantly on labor, energy, and raw material consumption. The ability to recover and reuse the N,N-dimethylpropanolamine solvent through distillation further lowers the variable cost per kilogram. Additionally, the high yield and purity achieved in the crude reaction mixture reduce the need for extensive recrystallization or chromatographic purification, leading to substantial cost savings in processing time and solvent usage.
- Enhanced Supply Chain Reliability: Securing a reliable pharmaceutical intermediates supplier often hinges on the robustness of the underlying chemistry. This process utilizes robust heterogeneous catalysts that can be filtered and potentially regenerated, reducing dependency on scarce homogeneous metal complexes. The mild reaction conditions and the use of common solvents like methanol in the hydrogenation step ensure that the process can be easily replicated across different manufacturing sites, mitigating the risk of supply disruptions caused by facility-specific bottlenecks or regulatory hurdles associated with more hazardous chemistries.
- Scalability and Environmental Compliance: From a regulatory perspective, the reduction in three-waste discharge is a critical enabler for commercial scale-up of complex pharmaceutical intermediates. The closed nature of the hydrogenation reaction and the efficient absorption of hydrogen halide gases mean that the process generates minimal volatile organic compounds (VOCs) and acidic emissions. This green profile facilitates easier permitting for capacity expansion and aligns with the increasingly strict environmental, social, and governance (ESG) criteria demanded by global multinational corporations, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on the practical aspects of scaling this route. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this process for their specific portfolio of active ingredients.
Q: Why is a mixed catalyst system preferred over single metal catalysts in this synthesis?
A: The patent data indicates that a mixed catalyst system comprising both Palladium on Carbon (Pd/C) and Platinum on Carbon (Pt/C) significantly enhances both the selectivity and reaction rate of the hydrogen substitution step compared to using either catalyst independently. This synergy prevents catalyst poisoning by halide ions and ensures higher conversion efficiency.
Q: What role does N,N-dimethylpropanolamine play in the reaction mechanism?
A: N,N-dimethylpropanolamine serves a dual function as both the primary solvent and an acid binding agent. As a tertiary amine, it stabilizes the pH of the reaction system by neutralizing the hydrogen halide byproduct generated during the nucleophilic substitution, thereby driving the equilibrium towards the formation of the desired 2-hydrazinopyridine derivative.
Q: How does this process improve environmental compliance compared to traditional methods?
A: Unlike traditional routes involving Hofmann degradation and Sandmeyer reactions which generate substantial waste liquids and solids, this catalytic hydrogen substitution route drastically reduces three-waste discharge. Furthermore, the closed-loop solvent recovery system and the ability to absorb hydrogen halide byproducts minimize volatile emissions and hazardous waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydrazinopyridine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch meets the exacting standards required for API synthesis. Our infrastructure is designed to handle the specific safety requirements of hydrazine chemistry and high-pressure hydrogenation, providing a secure and compliant manufacturing environment.
We invite you to collaborate with us to leverage this innovative synthesis route for your supply chain needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both cost and quality for your final products.
