Advanced Microbial Transformation for High-Purity Chiral Pharmaceutical Intermediates
Advanced Microbial Transformation for High-Purity Chiral Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective routes to access critical chiral building blocks. A significant breakthrough in this domain is documented in patent CN102168036B, which details a novel microbial transformation process for the preparation of R-4-chloro-3-hydroxybutyrate and S-3-hydroxy-butyrolactone. This technology leverages a newly screened strain, Bacillus megaterium WZ009, to catalyze the asymmetric hydrolysis of racemic esters with exceptional stereoselectivity. Unlike traditional chemical synthesis which often relies on harsh conditions and expensive chiral auxiliaries, this biocatalytic approach operates under mild physiological conditions, offering a greener alternative for large-scale manufacturing. For R&D directors and procurement specialists, understanding the nuances of this biological route is essential for optimizing supply chains and reducing the overall cost of goods sold for complex API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure 4-chloro-3-hydroxybutyrate derivatives has relied heavily on chemical resolution or enzymatic kinetic resolution using isolated lipases. Traditional chemical methods often suffer from theoretical yield limitations, typically capping at 50% for the desired enantiomer unless dynamic kinetic resolution is employed, which adds complexity and cost. Furthermore, chemical routes frequently require toxic solvents, heavy metal catalysts, and extreme temperatures, generating significant hazardous waste that complicates environmental compliance. Even existing biocatalytic methods using commercial lipases, such as those from Rhizomucor miehei, often necessitate organic solvent systems like benzene or dioxane, posing safety risks and requiring extensive solvent recovery infrastructure. These legacy processes create bottlenecks in supply chain reliability and inflate the manufacturing costs of downstream pharmaceuticals like L-carnitine and statins.
The Novel Approach
The innovative method described in the patent utilizes whole-cell biocatalysis with the specific strain Bacillus megaterium WZ009 to overcome these historical inefficiencies. This biological system exhibits high enzyme activity and remarkable stereoselectivity, allowing for the simultaneous production of two valuable chiral intermediates from a single racemic substrate. The process operates in an aqueous buffer system, eliminating the need for volatile organic compounds and significantly reducing the environmental footprint. By employing a robust bacterial strain that can be cultivated via standard fermentation techniques, manufacturers can achieve high volumetric productivity. The ability to produce both the R-ester and the S-lactone simultaneously maximizes atom economy, turning what was once a waste stream in kinetic resolutions into a valuable co-product. This dual-production capability represents a paradigm shift in how we approach the synthesis of these critical pharma intermediates.
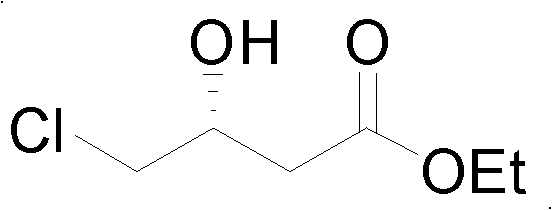
Mechanistic Insights into Microbial Asymmetric Hydrolysis
The core of this technology lies in the specific ester hydrolase activity inherent to the Bacillus megaterium WZ009 strain. The mechanism involves the selective hydrolysis of one enantiomer of the racemic 4-chloro-3-hydroxybutyrate ester, leaving the other enantiomer untouched while cyclizing the hydrolyzed acid into the corresponding lactone. This specificity is governed by the enzyme's active site geometry, which strictly accommodates the substrate in a specific orientation. The reaction proceeds optimally in a phosphate buffer system maintained at a pH between 6.5 and 7.5, with experimental data indicating peak performance at pH 7.2. Temperature control is also critical, with the biocatalyst showing highest efficiency at 30°C, although it remains active across a range of 20°C to 50°C. This robustness allows for flexible process control during scale-up, ensuring consistent quality even with minor fluctuations in reactor conditions.
From an impurity control perspective, the biological nature of the reaction ensures a very clean profile compared to chemical alternatives. The high enantiomeric excess (e.e. > 99% for the R-ester) minimizes the burden on downstream purification steps. The separation strategy capitalizes on the distinct physical properties of the products; the unreacted R-ester and the formed S-lactone have significantly different boiling points, allowing for efficient separation via vacuum distillation. This simplicity in downstream processing is a major advantage for commercial manufacturing, as it reduces the number of unit operations and solvent usage. The reaction scheme below illustrates the transformation where the racemic substrate is converted into the valuable chiral components with high fidelity.
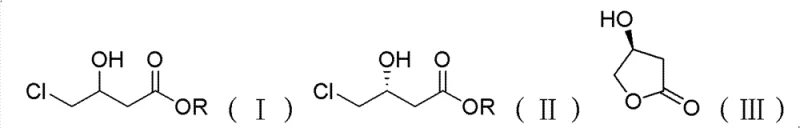
How to Synthesize R-4-chloro-3-hydroxybutyrate Efficiently
Implementing this biocatalytic route requires precise control over fermentation and bioconversion parameters to maximize yield and optical purity. The process begins with the cultivation of the bacterial strain in a nutrient-rich medium followed by the bioconversion step in a buffered aqueous system. Operators must carefully monitor the substrate concentration, as high loads can inhibit enzyme activity, while low loads reduce volumetric efficiency. The patent data suggests an optimal substrate concentration around 100 mmol/L to balance these factors. Following the reaction, a straightforward extraction and distillation protocol isolates the high-purity products. For detailed operational parameters and standard operating procedures, please refer to the technical guide below.
- Cultivate Bacillus megaterium WZ009 in a fermentation medium containing glucose and yeast extract at 30°C for 12 hours to obtain wet thallus.
- Suspend the freeze-dried bacterial powder in a phosphate buffer system (pH 7.2) and add racemic 4-chloro-ethyl 3-hydroxybutanoate substrate.
- React at 30°C for 12 hours, then separate the organic phase via ethyl acetate extraction and purify products through vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this microbial transformation technology offers tangible strategic benefits beyond mere technical feasibility. The shift from chemical resolution to whole-cell biocatalysis fundamentally alters the cost structure of producing these chiral intermediates. By utilizing a renewable biological catalyst that can be produced via fermentation, companies can decouple their supply from fluctuating prices of synthetic chiral reagents and precious metal catalysts. The process simplifies the manufacturing workflow, reducing the number of synthetic steps and the associated labor and utility costs. Furthermore, the aqueous nature of the reaction aligns with increasingly stringent global environmental regulations, mitigating the risk of regulatory fines and shutdowns associated with hazardous waste disposal.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolving agents and organic solvents leads to substantial cost savings in raw material procurement. The ability to recycle the biocatalyst for multiple batches significantly amortizes the cost of the enzyme preparation over a larger volume of product. Additionally, the high selectivity reduces the loss of starting material, improving the overall mass balance and lowering the effective cost per kilogram of the final API intermediate. These factors combine to create a more resilient and cost-competitive supply chain for high-value pharmaceutical ingredients.
- Enhanced Supply Chain Reliability: Relying on a robust bacterial strain like Bacillus megaterium ensures a consistent and scalable source of catalytic activity. Unlike isolated enzymes which may have stability issues during storage and transport, whole cells or freeze-dried powders offer superior shelf life and ease of handling. This reliability translates to reduced lead times and fewer production delays caused by catalyst degradation. The simplicity of the fermentation process also means that production can be easily scaled up or diversified across different manufacturing sites to mitigate geopolitical or logistical risks.
- Scalability and Environmental Compliance: The process is inherently green, operating in water at mild temperatures, which drastically reduces energy consumption compared to high-temperature chemical processes. The absence of toxic solvents simplifies waste treatment and lowers the environmental compliance burden. This sustainability profile is increasingly important for multinational corporations aiming to meet carbon neutrality goals. The scalability of fermentation technology is well-established in the industry, allowing for seamless transition from pilot scale to multi-ton commercial production without significant process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic process. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing platforms. The answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for decision-makers.
Q: What is the enantiomeric excess achieved with this biocatalytic method?
A: The patented process utilizing Bacillus megaterium WZ009 achieves an enantiomeric excess (e.e.) of greater than 99% for the R-4-chloro-3-hydroxybutyrate product and approximately 95% for the S-3-hydroxy-butyrolactone byproduct.
Q: Can the biocatalyst be reused to lower production costs?
A: Yes, the wet thallus or freeze-dried powder of Bacillus megaterium WZ009 demonstrates excellent stability and can be recycled for multiple batches while maintaining over 80% of its original enzymatic activity.
Q: What are the optimal reaction conditions for this transformation?
A: The optimal conditions involve a phosphate buffer system at pH 7.2, a reaction temperature of 30°C, and a substrate concentration of roughly 100 mmol/L to maximize both conversion rate and stereoselectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable R-4-chloro-3-hydroxybutyrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalysis in modern pharmaceutical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the Bacillus megaterium transformation are successfully translated into robust industrial operations. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs that guarantee every batch meets the highest international standards. We understand that consistency is key in the pharmaceutical supply chain, and our state-of-the-art facilities are designed to deliver reliable volumes of high-purity intermediates without compromise.
We invite you to collaborate with us to optimize your supply chain for chiral intermediates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how this biocatalytic route can enhance your bottom line. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. Let us help you navigate the complexities of chiral synthesis with confidence and efficiency.
