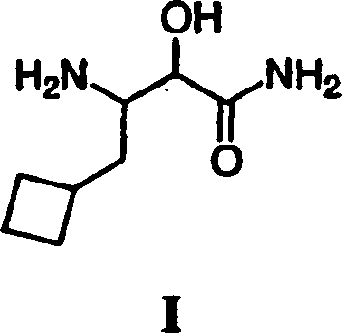
Scalable Synthesis of HCV Protease Inhibitor Intermediates via Optimized TEMPO Oxidation
Scalable Synthesis of HCV Protease Inhibitor Intermediates via Optimized TEMPO Oxidation
The pharmaceutical industry's relentless pursuit of effective Hepatitis C Virus (HCV) treatments has placed significant demand on the supply chain for high-quality protease inhibitor intermediates. Patent CN1805920A introduces a robust and commercially viable methodology for preparing 3-(amino)-3-cyclobutylmethyl-2-hydroxy-propionamide, a critical building block for next-generation antiviral therapies. This technical disclosure outlines a seven-step synthetic route that prioritizes safety, scalability, and impurity control, addressing the historical challenges associated with synthesizing sterically hindered amino acid derivatives. By leveraging mild oxidation conditions and efficient protection group strategies, this process offers a distinct advantage over traditional methods that often rely on hazardous reagents or complex chromatographic separations. For procurement leaders and R&D directors, understanding the nuances of this pathway is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting the rigorous quality standards of modern antiviral drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclobutyl-substituted amino acid intermediates has been plagued by inefficiencies related to harsh reaction conditions and poor atom economy. Traditional routes often necessitate the use of stoichiometric heavy metal oxidants, such as chromium-based reagents, to achieve the necessary oxidation states, which generates substantial toxic waste streams and complicates downstream purification. Furthermore, earlier methodologies frequently struggled with the control of stereochemistry at the alpha-carbon, leading to difficult-to-separate diastereomeric mixtures that drastically reduce overall yield. The reliance on expensive chiral auxiliaries or resolution steps in conventional processes also inflates the cost of goods sold (COGS), making the final API economically challenging to produce at a commercial scale. These factors collectively create bottlenecks in the supply chain, increasing lead times and introducing variability that is unacceptable for GMP manufacturing environments.
The Novel Approach
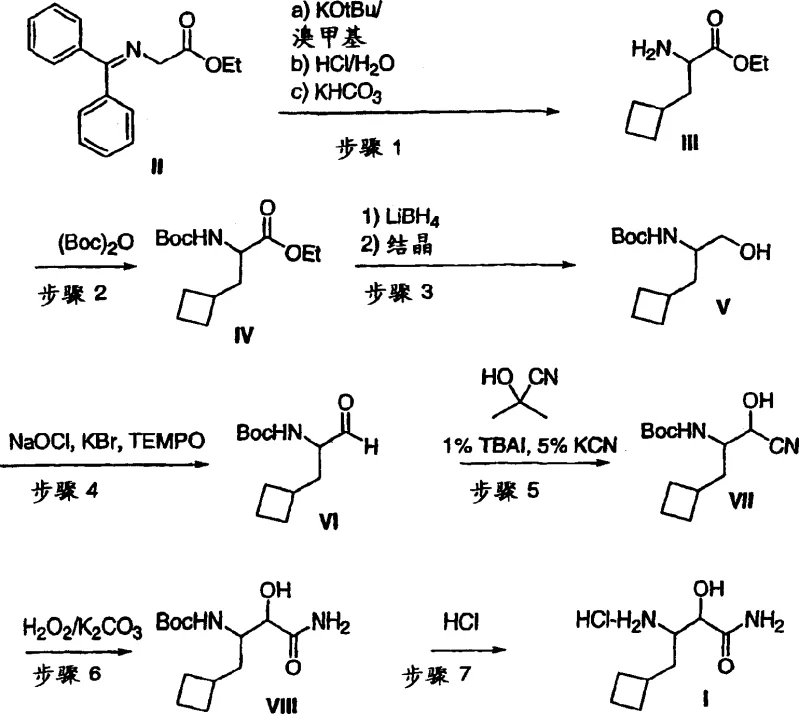
In contrast, the methodology described in CN1805920A employs a strategic sequence of protection, reduction, and catalytic oxidation to circumvent these traditional pitfalls. The process initiates with a highly efficient alkylation of a glycine equivalent, followed by a selective Boc-protection strategy that stabilizes the amine functionality throughout the subsequent transformations. A key innovation lies in the use of lithium borohydride for ester reduction, which offers superior chemoselectivity compared to bulkier hydride sources, minimizing side reactions. Most notably, the adoption of a TEMPO-mediated oxidation system replaces toxic heavy metals with a catalytic cycle driven by inexpensive bleach (NaOCl), aligning the process with green chemistry principles. This novel approach not only simplifies the workup procedures but also enhances the overall throughput, providing a clear pathway for cost reduction in API manufacturing without compromising on product integrity.
Mechanistic Insights into TEMPO-Mediated Oxidation and Nitrile Hydration
The core of this synthetic strategy relies on the precise execution of the TEMPO-catalyzed oxidation of the primary alcohol to the corresponding aldehyde. Mechanistically, the nitroxyl radical TEMPO acts as a hydrogen abstractor, facilitated by a co-oxidant like sodium hypochlorite and a bromide source such as lithium bromide to generate the active oxoammonium species in situ. This catalytic cycle is exceptionally mild, operating effectively at near-neutral pH and low temperatures (approximately -5°C to 5°C), which prevents the epimerization of the sensitive alpha-chiral center adjacent to the carbonyl group. Following oxidation, the aldehyde is immediately trapped as a cyanohydrin using acetone cyanohydrin and a phase transfer catalyst, a step that extends the carbon chain while installing the nitrogen functionality required for the final amide. The subsequent hydration of the nitrile to the primary amide using hydrogen peroxide and base is particularly elegant, avoiding the harsh acidic or basic conditions typically required for nitrile hydrolysis that could otherwise degrade the cyclobutyl ring or the protecting groups.
Impurity control is inherently built into the mechanistic design of this route, particularly through the management of diastereomers formed during the cyanohydrin addition and hydration steps. The patent data indicates that the hydration step yields a mixture of diastereomers (RR/SS and RS/SR), which can be effectively separated through fractional crystallization from specific solvent systems like heptane or butyl acetate. This physical separation capability is crucial for R&D teams focused on purity profiles, as it allows for the isolation of the desired stereoisomer with high enantiomeric excess prior to the final deprotection. By deferring the removal of the chiral auxiliary until the latest possible stage and utilizing crystallization rather than chromatography for purification, the process minimizes material loss and solvent consumption. This mechanistic understanding underscores the process's suitability for producing high-purity HCV intermediate grades that meet the stringent regulatory requirements for antiviral drug substances.
How to Synthesize 3-(Amino)-3-Cyclobutylmethyl-2-Hydroxy-Propionamide Efficiently
Executing this synthesis requires strict adherence to the temperature profiles and reagent stoichiometries outlined in the patent examples to ensure optimal yield and safety. The process begins with the generation of a nucleophilic glycine enolate using potassium tert-butoxide in THF, followed by alkylation with (bromomethyl)cyclobutane, a step that must be carefully monitored to prevent dialkylation. Subsequent steps involve standard unit operations such as aqueous workups, solvent swaps, and crystallizations, which are well-understood by process chemists but require precise control to maintain the integrity of the cyclobutyl moiety. The detailed标准化 synthesis steps见下方的指南 provide a granular view of the operational parameters necessary for successful replication.
- Alkylation of ethyl (diphenylmethylene)aminoacetate with (bromomethyl)cyclobutane followed by acidic hydrolysis to yield the free amine ester.
- Protection of the amine with Boc anhydride, followed by reduction of the ester to the primary alcohol using lithium borohydride.
- Oxidation of the alcohol to aldehyde using TEMPO/NaOCl, conversion to cyanohydrin, and subsequent hydration to the amide using hydrogen peroxide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly address the pain points of procurement managers and supply chain heads in the pharmaceutical sector. The elimination of heavy metal oxidants and the use of commodity chemicals like sodium hypochlorite and hydrogen peroxide significantly reduce the raw material costs and the environmental burden associated with waste disposal. Furthermore, the reliance on common solvents such as THF, ethyl acetate, and isopropanol ensures that the supply chain is resilient against shortages of exotic or highly regulated reagents. The process's inherent scalability, demonstrated by the patent's use of multi-kilogram batches, suggests that technology transfer to commercial manufacturing sites can be achieved with minimal risk, ensuring continuity of supply for critical antiviral medications.
- Cost Reduction in Manufacturing: The substitution of stoichiometric heavy metal oxidants with a catalytic TEMPO system drastically lowers the cost of reagents and eliminates the expensive downstream processing required to remove trace metals to ppm levels. Additionally, the ability to separate diastereomers via crystallization rather than preparative HPLC or column chromatography represents a massive saving in both time and silica/solvent costs. These efficiencies compound over large production runs, resulting in substantial cost savings that can be passed down the supply chain or reinvested into further process optimization. The overall atom economy is improved by avoiding unnecessary protection-deprotection sequences on the hydroxyl group, streamlining the step count and reducing labor hours.
- Enhanced Supply Chain Reliability: The starting materials, including ethyl (diphenylmethylene)aminoacetate and (bromomethyl)cyclobutane, are readily available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions, which tolerate slight variations in temperature and addition rates without catastrophic failure, adds a layer of operational security for contract manufacturing organizations (CMOs). This reliability is critical for maintaining just-in-time inventory levels for API production, reducing the need for excessive safety stock and freeing up working capital. The use of stable intermediates that can be isolated and stored, such as the Boc-protected alcohol, further decouples production steps, allowing for flexible scheduling and batch management.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, utilizing aqueous workups and generating benign byproducts like sodium chloride and water, which simplifies wastewater treatment and regulatory compliance. The avoidance of cryogenic conditions (below -40°C) for the majority of the steps reduces energy consumption for cooling, contributing to a lower carbon footprint for the manufacturing facility. Scalability is evidenced by the linear progression from gram to kilogram scales described in the examples, indicating that the heat transfer and mixing dynamics remain manageable in large reactors. This environmental and operational profile makes the process highly attractive for companies aiming to meet corporate sustainability goals while expanding production capacity for high-demand antiviral therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent's experimental data and claims. Understanding these details helps stakeholders evaluate the feasibility of adopting this technology for their specific manufacturing needs. The answers reflect the balance between chemical efficiency and practical operability that defines this invention.
Q: What are the key advantages of the TEMPO oxidation step in this process?
A: The use of TEMPO/NaOCl avoids toxic heavy metal oxidants like chromium, significantly reducing environmental waste and purification costs while maintaining high selectivity for the aldehyde intermediate.
Q: How is stereochemical purity managed during the synthesis?
A: The process utilizes crystallization techniques during the reduction and hydration steps to separate diastereomers, ensuring the final API intermediate meets stringent enantiomeric excess specifications required for HCV inhibitors.
Q: Is this process suitable for multi-ton commercial production?
A: Yes, the patent explicitly demonstrates scalability using kilogram-scale inputs (e.g., 35kg starting material) and common solvents like THF and ethyl acetate, making it highly viable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(Amino)-3-Cyclobutylmethyl-2-Hydroxy-Propionamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against Hepatitis C, and we are uniquely positioned to support your development and commercialization efforts. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3-(amino)-3-cyclobutylmethyl-2-hydroxy-propionamide meets or exceeds pharmacopeial standards. Our commitment to quality is matched by our dedication to technical excellence, making us a trusted partner for complex amino acid derivative synthesis.
We invite you to engage with our technical procurement team to discuss how this optimized synthetic route can be tailored to your specific volume and timeline requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how implementing this TEMPO-based methodology can improve your project's economics. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on real-world performance metrics rather than theoretical projections. Let us collaborate to secure your supply chain and accelerate the delivery of life-saving antiviral therapies to patients worldwide.
