Scalable Chiral Resolution Technology for High-Purity IRS Pharmaceutical Intermediates
The pharmaceutical industry's relentless pursuit of potent therapeutics for metabolic disorders has placed a premium on high-purity chiral intermediates, particularly those targeting Insulin Resistance Syndrome (IRS). Patent CN1433393A introduces a groundbreaking resolution methodology that fundamentally shifts the production paradigm for (S)-enantiomers of 2-ethoxy-3-(4-hydroxyphenyl)propanoic acids and their derivatives. Unlike traditional reliance on expensive chiral chromatography or low-yield enzymatic processes, this novel approach utilizes diastereomeric salt crystallization to achieve exceptional enantiomeric purity. The technical significance of this patent lies in its ability to transform a racemic mixture into a highly enriched chiral building block through a series of chemically robust and scalable unit operations. By leveraging specific chiral amines such as (S)-(-)-1-(1-naphthyl)-ethylamine, the process creates distinct physical property differences between diastereomers, enabling efficient separation via crystallization. This innovation is not merely a laboratory curiosity but a commercially viable pathway that addresses the critical bottlenecks of cost, scalability, and purity consistency required by global regulatory bodies for active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of enantiomerically enriched intermediates for complex pharmaceutical applications has been fraught with significant technical and economic challenges. Conventional techniques such as chiral high-performance liquid chromatography (HPLC), while highly effective for analytical separation, often fail to translate efficiently to industrial manufacturing scales due to prohibitive solvent consumption and column loading limitations. Furthermore, enzymatic resolution methods, though selective, frequently suffer from narrow substrate specificity and sensitivity to reaction conditions, leading to inconsistent batch-to-batch yields and extended processing times. The reliance on these traditional methods often results in a fragmented supply chain where the cost of goods sold (COGS) is inflated by the need for specialized equipment and extensive downstream purification steps. Additionally, the generation of substantial chemical waste from chromatographic mobile phases poses environmental compliance hurdles that modern green chemistry initiatives strive to eliminate. Consequently, procurement teams and process chemists have long sought a more robust alternative that balances high stereochemical control with operational simplicity and economic feasibility.
The Novel Approach
The methodology outlined in CN1433393A presents a transformative solution by employing classical diastereomeric salt formation coupled with modern crystallization engineering. This approach circumvents the limitations of chromatography by exploiting the inherent solubility differences between diastereomeric salts formed from the reaction of the racemic acid with a chiral amine. The process allows for the isolation of the desired (S)-enantiomer through straightforward filtration and washing steps, drastically reducing solvent usage and processing time. Moreover, the patent details versatile deprotection strategies, including the use of thiols for methyl ether cleavage and catalytic hydrogenation for benzyl groups, providing manufacturers with flexible options to integrate this chemistry into existing facilities. The ability to achieve enantiomeric excess (e.e.) values exceeding 95% through recrystallization demonstrates the high fidelity of this resolution technique. By shifting the separation burden from complex machinery to thermodynamic control via crystallization, this novel approach offers a streamlined, cost-effective, and environmentally friendlier route to high-value pharmaceutical intermediates.
Mechanistic Insights into Diastereomeric Salt Crystallization and Hydrogenation
The core mechanistic driver of this resolution process is the stereoselective interaction between the racemic carboxylic acid substrate and a chiral base, typically an amine. When the racemic mixture reacts with a pure enantiomer of a chiral amine, such as (S)-(-)-1-(1-naphthyl)-ethylamine, it generates a pair of diastereomeric salts. Unlike enantiomers, which possess identical physical properties in an achiral environment, diastereomers exhibit distinct melting points, solubilities, and crystallization behaviors. 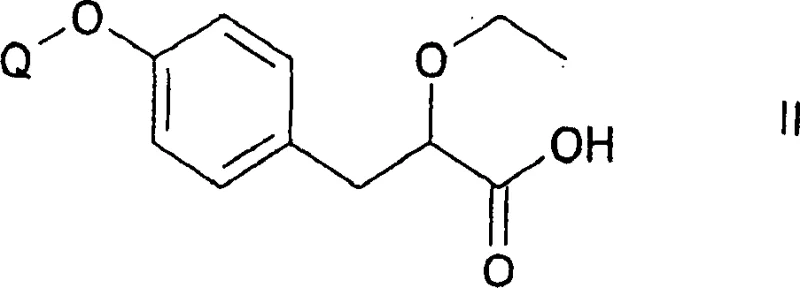 . This divergence in physical properties is the cornerstone of the separation strategy; by carefully selecting the solvent system (e.g., isopropyl acetate) and controlling the cooling profile, the less soluble diastereomeric salt precipitates out of the solution while the unwanted isomer remains in the mother liquor. The patent emphasizes that this crystallization can be iterative, allowing for successive enrichment of the target enantiomer to reach purity levels suitable for API synthesis. Following isolation, the chiral amine is removed via acid-base extraction, regenerating the free chiral acid without racemization, thus preserving the stereochemical integrity established during the salt formation step.
. This divergence in physical properties is the cornerstone of the separation strategy; by carefully selecting the solvent system (e.g., isopropyl acetate) and controlling the cooling profile, the less soluble diastereomeric salt precipitates out of the solution while the unwanted isomer remains in the mother liquor. The patent emphasizes that this crystallization can be iterative, allowing for successive enrichment of the target enantiomer to reach purity levels suitable for API synthesis. Following isolation, the chiral amine is removed via acid-base extraction, regenerating the free chiral acid without racemization, thus preserving the stereochemical integrity established during the salt formation step.
In addition to the resolution mechanics, the patent elucidates a critical reduction step involving the hydrogenation of unsaturated precursors to generate the saturated propanoic acid backbone. This transformation typically involves the reduction of a double bond in a 2-ethoxy-3-(4-substituted phenyl)acrylic acid derivative using a heterogeneous catalyst like palladium on carbon (Pd/C) under hydrogen pressure. 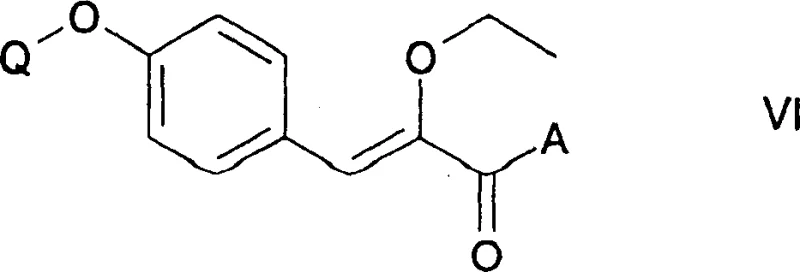 . The mechanistic nuance here lies in the compatibility of the catalyst with other functional groups present in the molecule, such as the ether linkage and the protected phenol. The process ensures that the reduction is chemoselective, targeting only the olefinic bond while leaving the rest of the molecular architecture intact. This step is pivotal for constructing the specific 2-ethoxy-3-phenylpropanoic acid scaffold required for IRS inhibitors. The combination of precise hydrogenation conditions and the subsequent chiral resolution creates a synergistic workflow that maximizes overall yield and minimizes impurity profiles, addressing the rigorous quality standards demanded by R&D directors for late-stage intermediate production.
. The mechanistic nuance here lies in the compatibility of the catalyst with other functional groups present in the molecule, such as the ether linkage and the protected phenol. The process ensures that the reduction is chemoselective, targeting only the olefinic bond while leaving the rest of the molecular architecture intact. This step is pivotal for constructing the specific 2-ethoxy-3-phenylpropanoic acid scaffold required for IRS inhibitors. The combination of precise hydrogenation conditions and the subsequent chiral resolution creates a synergistic workflow that maximizes overall yield and minimizes impurity profiles, addressing the rigorous quality standards demanded by R&D directors for late-stage intermediate production.
How to Synthesize (S)-2-Ethoxy-3-(4-Hydroxyphenyl)Propanoic Acid Efficiently
The synthesis of this high-value chiral intermediate requires a disciplined approach to reaction conditions and purification parameters to ensure optimal yield and stereochemical purity. The process begins with the preparation of the racemic precursor, followed by the critical resolution step where the choice of chiral amine and solvent dictates the success of the separation. Operators must maintain strict temperature control during the salt formation and crystallization phases to prevent oiling out or co-crystallization of impurities. Detailed standard operating procedures (SOPs) regarding the stoichiometry of the chiral amine (often used in sub-stoichiometric amounts to favor selective precipitation) and the rate of cooling are essential for reproducibility. This structured workflow ensures that the transition from bench-scale optimization to pilot plant production is seamless, mitigating the risks associated with scaling up chiral technologies.
- React the racemic 2-ethoxy-3-(4-hydroxyphenyl)propanoic acid derivative with a chiral amine such as (S)-(-)-1-(1-naphthyl)-ethylamine to form a diastereomeric salt mixture.
- Perform fractional crystallization from a suitable solvent system like isopropyl acetate to isolate the less soluble diastereomeric salt, thereby enriching the desired (S)-enantiomer.
- Treat the isolated salt with acid or base to liberate the free chiral acid, followed by optional deprotection steps using thiols or hydrogenation to yield the final pure intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the resolution technology described in CN1433393A represents a strategic opportunity to optimize the cost structure and reliability of the pharmaceutical supply chain. The shift from chromatographic separation to crystallization-based resolution eliminates the need for expensive chiral columns and the vast quantities of high-grade solvents associated with HPLC, leading to a substantial reduction in raw material costs. Furthermore, crystallization is a unit operation that is inherently easier to scale than chromatography, allowing manufacturers to increase batch sizes significantly without proportional increases in capital expenditure or facility footprint. This scalability directly translates to enhanced supply chain reliability, as production bottlenecks related to column capacity or solvent recovery are effectively removed. The robustness of the process also implies shorter lead times for order fulfillment, as the cycle time per batch is reduced and the risk of failed batches due to column degradation is nullified. Additionally, the use of common reagents like palladium on carbon and standard amines ensures that the supply of inputs remains stable and less susceptible to market volatility compared to specialized enzymatic kits or custom chromatography media.
- Cost Reduction in Manufacturing: The elimination of chiral chromatography removes one of the most cost-intensive steps in fine chemical manufacturing, replacing it with energy-efficient crystallization and filtration. This structural change in the process flow significantly lowers the variable cost per kilogram of the intermediate, allowing for more competitive pricing models in long-term supply agreements. The ability to recover and recycle the chiral amine resolving agent further amplifies these savings, creating a closed-loop system that minimizes waste and maximizes resource utilization. By reducing the dependency on specialized consumables, manufacturers can allocate capital towards capacity expansion rather than ongoing operational overheads, driving long-term economic sustainability.
- Enhanced Supply Chain Reliability: Crystallization processes are well-understood and widely supported by standard chemical engineering infrastructure, reducing the risk of technical failures that can disrupt supply. Unlike enzymatic processes that may require strict cold-chain logistics for enzyme storage or chromatography that suffers from column fouling, this chemical resolution method offers consistent performance across diverse manufacturing environments. This reliability ensures that downstream API producers receive their critical intermediates on schedule, preventing costly delays in drug development timelines. The robustness of the chemistry also allows for multi-sourcing strategies, as the process can be easily transferred between different contract manufacturing organizations (CMOs) without significant re-validation burdens.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by minimizing solvent intensity and avoiding the generation of hazardous waste streams associated with chromatographic mobile phases. The ability to scale from gram to tonne quantities using standard reactors facilitates rapid response to market demand surges without the need for bespoke equipment fabrication. Moreover, the simplified waste profile simplifies effluent treatment requirements, ensuring compliance with increasingly stringent environmental regulations. This alignment with sustainability goals not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the supply chain, a factor of growing importance to global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this intermediate into their drug development pipelines. The answers are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for technical decision-makers. These insights provide a clear framework for assessing the operational readiness and strategic value of adopting this novel synthetic route.
Q: What is the primary advantage of using diastereomeric salt crystallization over chiral chromatography for this intermediate?
A: Diastereomeric salt crystallization offers superior scalability and significantly lower operational costs compared to chiral chromatography. While chromatography is effective for analytical separation, it is often prohibitively expensive and difficult to scale for multi-kilogram production due to high solvent consumption and column costs. Crystallization leverages standard industrial equipment and allows for continuous processing, making it the preferred method for commercial manufacturing of pharmaceutical intermediates.
Q: Which chiral resolving agents are most effective for this specific resolution process?
A: The patent data identifies (S)-(-)-1-(1-naphthyl)-ethylamine as the most preferred chiral amine for forming the diastereomeric salt. Other effective agents include quinine, quinidine, and pseudoephedrine derivatives. The choice of amine is critical as it dictates the solubility differences between the diastereomers, which drives the efficiency of the crystallization and the final enantiomeric excess (e.e.) of the product.
Q: How is the phenolic hydroxyl group protected and deprotected during this synthesis?
A: The phenolic hydroxyl group can be protected as a methyl ether or a benzyl ether depending on the specific synthetic route. For methyl ethers, deprotection is achieved using thiols like octanethiol at elevated temperatures (100-140°C). For benzyl ethers, catalytic hydrogenation using palladium on carbon (Pd/C) is employed. This flexibility allows manufacturers to choose the protection strategy that best aligns with their existing infrastructure and safety protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Ethoxy-3-(4-Hydroxyphenyl)Propanoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity chiral intermediates play in the development of next-generation therapeutics for metabolic diseases. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee that every batch meets the exacting standards required for pharmaceutical applications. Our commitment to quality is matched by our dedication to process innovation, continuously optimizing synthetic routes to enhance yield and reduce environmental impact while maintaining the highest levels of safety and compliance.
We invite potential partners to engage with our technical procurement team to discuss how our capabilities align with your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of sourcing this intermediate through our optimized manufacturing channels. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your development timeline. Let us collaborate to accelerate your drug development programs with reliable, high-quality chemical solutions that drive success from early-stage research through to commercial launch.
