Advanced Multidentate Phosphite Ligands for High-Performance Hydroformylation Processes
The chemical industry continuously seeks advancements in catalytic processes to improve efficiency and sustainability, particularly in the production of essential aldehyde intermediates. Patent CN1374935A introduces a groundbreaking hydroformylation process utilizing novel multidentate phosphite ligands. These ligands are characterized by heteroatom-containing substituents positioned on the carbon attached to the ortho position of the terminal phenol group. This specific structural modification addresses long-standing challenges in catalyst stability and selectivity. By employing these advanced ligands in conjunction with transition metals, manufacturers can achieve superior performance in converting olefinically unsaturated compounds into valuable aldehydes. The technology represents a significant leap forward for producers of pharmaceutical intermediates and fine chemicals who require high-purity outputs with minimal byproduct formation.
Traditional hydroformylation methods often rely on simpler phosphine or phosphite ligands that may suffer from limited thermal stability or insufficient regioselectivity under rigorous industrial conditions. Conventional single phosphorus atom ligands, while effective in some contexts, frequently fail to maintain the precise steric and electronic environment required for optimal linear aldehyde production. Furthermore, the degradation of these simpler ligands can lead to catalyst deactivation, necessitating frequent replenishment and increasing operational costs. The limitations of these legacy systems become particularly pronounced when processing complex substrates or operating at elevated temperatures and pressures required for high throughput. Consequently, there is a persistent demand for more robust catalytic systems that can withstand these demands without compromising on yield or selectivity.
The novel approach disclosed in the patent overcomes these deficiencies through the strategic design of multidentate phosphite ligands. As illustrated in the core structural formulas, these ligands incorporate specific bridging units and ortho-substituents that lock the phosphorus atoms into a favorable conformation. 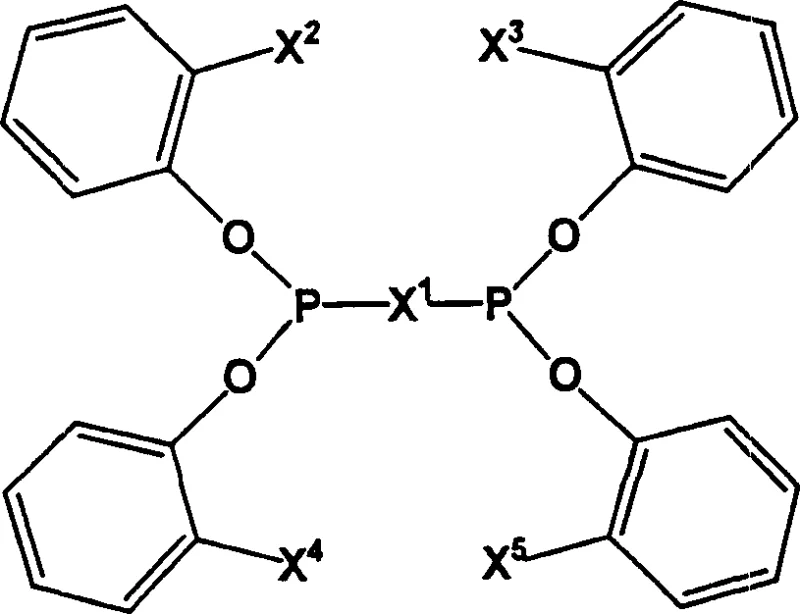 This structural rigidity enhances the stability of the metal-ligand complex, preventing premature decomposition. The presence of heteroatom-containing groups, such as acetals or ketals, further modulates the electronic properties of the phosphorus centers. This modulation allows for finer tuning of the catalytic activity, enabling the process to favor the formation of linear aldehydes over branched isomers. Such precision is critical for downstream processing, as linear aldehydes are often the desired precursors for plasticizers and detergents.
This structural rigidity enhances the stability of the metal-ligand complex, preventing premature decomposition. The presence of heteroatom-containing groups, such as acetals or ketals, further modulates the electronic properties of the phosphorus centers. This modulation allows for finer tuning of the catalytic activity, enabling the process to favor the formation of linear aldehydes over branched isomers. Such precision is critical for downstream processing, as linear aldehydes are often the desired precursors for plasticizers and detergents.
Mechanistic Insights into Rhodium-Catalyzed Hydroformylation
The efficacy of this hydroformylation process relies heavily on the interaction between the transition metal, typically rhodium, and the multidentate phosphite ligand. The catalytic cycle begins with the formation of an active hydrido-carbonyl species, where the ligand occupies coordination sites on the metal center. The unique bite angle and electronic donation of the multidentate ligand facilitate the oxidative addition of hydrogen and the subsequent insertion of the olefin substrate.  During the migratory insertion step, the steric bulk provided by the ortho-substituents on the ligand directs the incoming olefin to align in a manner that favors linear chain growth. This steric guidance is the mechanistic basis for the high linear-to-branched ratios observed in the experimental data. Furthermore, the electron-withdrawing nature of the phosphite moieties increases the electrophilicity of the metal center, accelerating the CO insertion step which is often rate-determining.
During the migratory insertion step, the steric bulk provided by the ortho-substituents on the ligand directs the incoming olefin to align in a manner that favors linear chain growth. This steric guidance is the mechanistic basis for the high linear-to-branched ratios observed in the experimental data. Furthermore, the electron-withdrawing nature of the phosphite moieties increases the electrophilicity of the metal center, accelerating the CO insertion step which is often rate-determining.
Impurity control is another critical aspect managed by this sophisticated ligand design. In conventional systems, ligand dissociation can lead to the formation of inactive metal clusters or non-selective catalytic species that generate unwanted byproducts. The chelating effect of the multidentate structure significantly reduces the likelihood of ligand dissociation, thereby maintaining a homogeneous and stable active species throughout the reaction duration. This stability minimizes the formation of heavy ends and saturated hydrocarbons, which are common impurities that complicate purification. By ensuring that the catalyst remains intact and selective, the process inherently produces a cleaner crude product stream. This reduction in impurity load translates directly to simplified downstream distillation and purification steps, enhancing the overall economic viability of the manufacturing process.
How to Synthesize Multidentate Phosphite Ligands Efficiently
The synthesis of these high-performance ligands involves a multi-step procedure that requires careful control of reaction conditions to ensure high purity and yield. The general methodology typically starts with the preparation of a phosphorus chloride intermediate by reacting specific substituted phenols or acetals with phosphorus trichloride in the presence of a base. This intermediate is then coupled with a divalent bridging unit, such as a binaphthyl derivative, to form the final multidentate structure. The detailed standardized synthesis steps are provided in the guide below.
- Preparation of Phosphorus Chloride Intermediate: React substituted phenols or acetals with phosphorus trichloride (PCl3) in the presence of a base like triethylamine at low temperatures.
- Coupling with Bridging Unit: React the resulting phosphorus chloride species with a divalent bridging compound, such as 2,2'-dihydroxy-1,1'-binaphthyl derivatives.
- Purification: Filter the reaction mixture to remove salts, evaporate solvents, and purify the final ligand product via crystallization or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this advanced ligand technology offers substantial strategic benefits beyond mere technical performance. The primary value proposition lies in the potential for significant cost reduction in aldehyde manufacturing. By achieving higher selectivity for the desired linear aldehyde products, the process minimizes the generation of worthless branched isomers and heavy byproducts. This efficiency gain means that less raw material is wasted, and the yield of valuable product per batch is maximized. Additionally, the enhanced stability of the catalyst system reduces the frequency of catalyst makeup or replacement. This longevity lowers the consumption of expensive precious metals like rhodium, which represents a major portion of the variable costs in hydroformylation operations.
- Cost Reduction in Manufacturing: The implementation of these multidentate ligands drives down production costs through improved atom economy and reduced catalyst consumption. The high selectivity ensures that a greater proportion of the feedstock is converted into saleable product rather than waste, effectively lowering the cost per kilogram of output. Furthermore, the robustness of the catalyst allows for longer run times without significant loss of activity, reducing downtime associated with catalyst change-outs. These factors combine to create a more lean and cost-effective manufacturing operation that is better insulated against fluctuations in raw material prices.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the versatility and stability of the catalyst system. The ligands are designed to be effective across a range of substrates, from simple alpha-olefins to functionalized internal olefins. This flexibility allows manufacturers to adapt quickly to changes in feedstock availability without needing to requalify entirely new catalyst systems. Moreover, the patent describes embodiments where the ligands can be immobilized on polymeric supports. This capability facilitates catalyst recovery and reuse, mitigating risks associated with the supply of fresh catalyst and reducing dependency on continuous external sourcing of sensitive catalytic materials.
- Scalability and Environmental Compliance: Scaling this technology from pilot to commercial production is streamlined by the well-defined synthesis routes and robust performance characteristics of the ligands. The process operates effectively under standard hydroformylation conditions, meaning existing infrastructure can often be utilized with minimal modification. From an environmental perspective, the higher selectivity and catalyst stability result in less chemical waste and lower energy consumption for separation processes. This aligns with increasingly stringent global regulations regarding industrial emissions and waste disposal, ensuring that the manufacturing process remains compliant and sustainable in the long term.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of multidentate phosphite ligands in hydroformylation processes. These answers are derived from the specific technical disclosures and experimental data found within the patent documentation. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this technology into their current production lines.
Q: What is the primary advantage of using multidentate phosphite ligands in hydroformylation?
A: These ligands significantly improve regioselectivity towards linear aldehydes and enhance catalyst stability under industrial reaction conditions compared to monodentate ligands.
Q: Can these ligands be used with polymer supports?
A: Yes, the patent describes embodiments where the ligands are loaded onto polymeric resins like Merrifield resin, facilitating catalyst recovery and reuse.
Q: What metals are compatible with these phosphite ligands?
A: While rhodium is preferred for high activity, the ligands are also compatible with other Group VIII metals such as cobalt, iridium, ruthenium, palladium, and platinum.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Multidentate Phosphite Ligands Supplier
NINGBO INNO PHARMCHEM stands at the forefront of catalytic innovation, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, ensuring that every batch of ligand meets the exacting standards required for high-performance hydroformylation. We understand the critical role that catalyst consistency plays in maintaining process stability and product quality. Our team of experts is ready to assist you in optimizing your specific application, whether it involves fine chemical synthesis or large-scale commodity production.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current operations. By leveraging our expertise in ligand design and process optimization, we can help you identify opportunities to enhance yield and reduce operational expenditures. Reach out today to obtain specific COA data and route feasibility assessments that will demonstrate the tangible value of partnering with us for your catalytic needs.
