Advanced Epoxide Carbonylation Technologies for Commercial Scale-up of Complex Pharmaceutical Intermediates
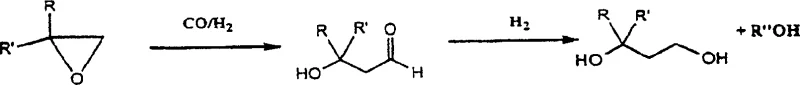
The chemical industry continuously seeks more efficient pathways for synthesizing bifunctional compounds that serve as critical building blocks for polymers and active pharmaceutical ingredients. Patent CN1188215C introduces a groundbreaking carbonylation method for epoxide derivatives that addresses long-standing challenges in reactivity and selectivity. This technology enables the high-yield synthesis of 3-hydroxyaldehyde and 3-hydroxyester derivatives, which are pivotal intermediates for producing 1,3-alkanediols. By leveraging a sophisticated catalyst system comprising Group 8B transition metals combined with cyclopentadienyl ligands alongside cobalt compounds, the process achieves superior performance compared to conventional techniques. The innovation lies not only in the catalyst composition but also in the strategic manipulation of reaction parameters such as temperature and pressure to maximize product purity. For R&D teams focused on process optimization, this patent offers a robust framework for developing scalable synthetic routes that minimize byproduct formation. The ability to convert simple epoxides into valuable diols through stable ester intermediates represents a significant leap forward in fine chemical manufacturing efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing 3-hydroxyaldehyde derivatives often rely on cobalt catalysts promoted by phosphine oxide ligands to achieve acceptable selectivity at moderate temperatures. However, these legacy systems suffer from significant drawbacks, primarily concerning the complexity of catalyst recovery and regeneration. The use of phosphine-based promoters frequently leads to difficult separation processes that increase operational costs and waste generation. Furthermore, alternative approaches utilizing expensive rhodium or ruthenium catalysts for hydroesterification have historically yielded disappointing results, with methyl 3-hydroxypropionate yields stagnating around 60 percent. These low yields are often accompanied by the formation of substantial byproducts due to the isomerization of raw materials. Additionally, when 3-hydroxyaldehydes are used as intermediates for 1,3-alkanediol synthesis, their inherent instability can lead to oligomer formation and acetal byproducts, ultimately compromising the quality of the final polyester or coating material. These technical bottlenecks have hindered the economic viability of large-scale production for many years.
The Novel Approach
The novel approach detailed in the patent overcomes these hurdles by introducing a dual-catalyst strategy that enhances both activity and selectivity without relying on problematic phosphine ligands. By employing transition metal compounds containing cyclopentadienyl groups in conjunction with cobalt catalysts, the process facilitates a highly selective hydroformylation reaction. Alternatively, for hydroesterification, the use of cobalt catalysts promoted by imidazole derivatives provides a cost-effective solution that avoids the high expense of noble metals. This method allows for the efficient conversion of epoxide derivatives into 3-hydroxy esters under controlled temperature and pressure conditions. The resulting esters are stable intermediates that can be subsequently hydrogenated to produce high-purity 1,3-alkanediols. This pathway effectively bypasses the instability issues associated with aldehyde intermediates, ensuring a cleaner reaction profile. The versatility of this system is evident in its ability to handle various epoxide substrates, making it a powerful tool for cost reduction in fine chemical manufacturing where feedstock flexibility is crucial.
Mechanistic Insights into Cobalt-Catalyzed Hydroesterification
The core of this technological advancement lies in the precise interaction between the cobalt catalyst and the promoter within the reaction medium. In the hydroesterification pathway, the cobalt compound, typically Co2(CO)8, interacts with promoters such as imidazole or pyridine derivatives to form an active catalytic species. This species facilitates the insertion of carbon monoxide into the epoxide ring, followed by nucleophilic attack by an alcohol molecule. The reaction mechanism is highly sensitive to the electronic environment created by the promoter, which stabilizes the transition state and directs the regioselectivity of the ring opening. Unlike traditional systems that might favor beta-lactone formation, this optimized catalyst system preferentially yields linear 3-hydroxy esters. The presence of specific solvents, such as water-saturated methyl tert-butyl ether (MTBE) or methanol, further influences the reaction kinetics by solvating the ionic intermediates effectively. Understanding these mechanistic nuances is essential for R&D directors aiming to replicate these results, as slight deviations in promoter concentration or solvent purity can impact the turnover frequency. The patent data indicates that maintaining the correct molar ratio between the cobalt source and the promoter is critical for sustaining high catalytic activity over extended reaction times.
Impurity control is another critical aspect governed by the reaction mechanism and conditions. The patent highlights that side reactions, such as the isomerization of epoxides to ketones or the formation of acetals, are minimized by carefully regulating the reaction temperature between 30°C and 130°C. At higher temperatures, while conversion rates may increase, the risk of byproduct formation also rises, necessitating a balanced approach. The use of imidazole promoters specifically helps suppress the formation of beta-lactones, which are common unwanted byproducts in cobalt-catalyzed carbonylation. Furthermore, the stability of the 3-hydroxy ester intermediate prevents the oligomerization issues seen with aldehyde counterparts. This inherent stability simplifies downstream purification, as the ester can be isolated via water extraction or vacuum distillation depending on the solvent system used. For quality control teams, this means a more consistent impurity profile and reduced burden on purification columns. The ability to recycle the catalyst solution after product separation further ensures that metal contamination in the final product remains within stringent limits, a key requirement for pharmaceutical grade intermediates.
How to Synthesize 3-Hydroxy Esters Efficiently
Implementing this synthesis route requires careful attention to the preparation of the catalyst system and the control of reaction parameters to ensure optimal yield and selectivity. The process begins with the dissolution of the cobalt catalyst and the chosen promoter in a suitable solvent, followed by the introduction of the epoxide substrate under an inert atmosphere. Operators must then pressurize the reactor with carbon monoxide and heat the mixture to the specified range, monitoring the pressure drop to gauge reaction progress. Detailed standard operating procedures are essential to maintain safety given the use of high-pressure carbon monoxide and flammable solvents. The following guide outlines the standardized synthesis steps derived from the patent examples to assist technical teams in replicating this high-efficiency pathway.
- Prepare the catalyst system by dissolving a cobalt compound such as Co2(CO)8 and an effective promoter like imidazole or a cyclopentadienyl transition metal complex in a suitable solvent such as methanol or MTBE.
- Charge the reactor with the epoxide derivative and pressurize with carbon monoxide to a range of 690 to 20684 kPa, maintaining a reaction temperature between 30°C and 130°C.
- After the hydroesterification reaction reaches completion, separate the 3-hydroxy ester intermediate and subject it to hydrogenation using a Cu-Cr or Pd/C catalyst to yield the final 1,3-alkanediol product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift towards this novel carbonylation technology offers substantial opportunities for optimizing the cost structure of intermediate production. The replacement of expensive noble metal catalysts like rhodium and ruthenium with abundant cobalt compounds significantly lowers the initial capital outlay for catalyst procurement. Moreover, the elimination of complex phosphine oxide ligands reduces the dependency on specialized chemical suppliers and simplifies the supply chain for raw materials. The ability to recycle the catalyst system multiple times without significant loss of activity translates directly into reduced operating expenses over the lifecycle of the plant. For supply chain heads, the robustness of this process under moderate pressure and temperature conditions enhances operational safety and reduces the energy footprint associated with high-severity reactions. The versatility of the feedstock, allowing for the use of various epoxides, provides flexibility in sourcing strategies, mitigating risks associated with single-source dependencies. These factors collectively contribute to a more resilient and cost-effective manufacturing model.
- Cost Reduction in Manufacturing: The substitution of high-cost noble metal catalysts with economical cobalt-based systems drives down the variable cost per kilogram of the final product. By avoiding the use of expensive phosphine ligands and reducing the need for complex purification steps to remove metal residues, the overall production cost is significantly optimized. The high selectivity of the reaction minimizes waste generation, leading to better atom economy and lower disposal costs. Furthermore, the potential for catalyst recycling extends the useful life of the catalytic material, amortizing the initial investment over a larger production volume. These cumulative efficiencies result in a more competitive pricing structure for the final 1,3-alkanediol intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: Utilizing widely available cobalt salts and simple organic promoters like imidazole reduces the risk of supply disruptions compared to relying on scarce noble metals. The simplified catalyst formulation means fewer specialized components are required to keep the production line running, streamlining inventory management. The process tolerance for various solvent systems, including common ethers and alcohols, allows for flexible sourcing of consumables based on market availability. Additionally, the stability of the intermediate 3-hydroxy esters facilitates safer storage and transportation if semi-finished goods need to be moved between facilities. This operational flexibility ensures consistent delivery schedules and strengthens the reliability of the supply chain for downstream customers requiring just-in-time inventory.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are well-suited for scale-up from pilot plants to commercial production facilities without requiring exotic high-pressure equipment. The use of less toxic promoters and the avoidance of heavy metal contaminants align with increasingly strict environmental regulations regarding effluent discharge. Efficient catalyst recovery systems minimize the release of heavy metals into the environment, supporting sustainability goals. The high conversion rates reduce the volume of unreacted starting materials that need to be treated or recycled, lowering the overall environmental burden. This alignment with green chemistry principles not only ensures regulatory compliance but also enhances the brand reputation of manufacturers committed to sustainable production practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this epoxide carbonylation technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to provide accurate guidance for potential adopters. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this process into their existing manufacturing portfolios. The insights cover catalyst performance, substrate scope, and downstream processing considerations.
Q: How does the novel cobalt-imidazole catalyst system improve upon traditional rhodium-based hydroformylation?
A: The novel system utilizes inexpensive cobalt compounds promoted by imidazole derivatives instead of costly rhodium or ruthenium catalysts. This substitution significantly reduces raw material costs while maintaining high selectivity for 3-hydroxy esters, avoiding the formation of unstable aldehyde intermediates common in older methods.
Q: What are the primary advantages regarding catalyst recovery in this carbonylation process?
A: Unlike processes using phosphine oxide ligands which complicate regeneration, the cyclopentadienyl transition metal and cobalt catalysts described in the patent allow for easier separation. The catalyst solution can often be recycled multiple times with minimal loss of activity, enhancing overall process sustainability.
Q: Can this method be scaled for the production of diverse 1,3-alkanediol derivatives?
A: Yes, the patent demonstrates high conversion rates and selectivity across a broad substrate scope including ethylene oxide, propylene oxide, and various substituted epoxides. The mild reaction conditions and robust catalyst system support commercial scale-up for producing high-purity intermediates used in polyesters and pharmaceuticals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxy Ester Derivatives Supplier
The technological advancements outlined in patent CN1188215C represent a significant opportunity for the production of high-value chemical intermediates with improved efficiency and purity. NINGBO INNO PHARMCHEM stands ready to leverage this expertise to support your development and commercialization goals. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to the marketplace. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for pharmaceutical and specialty chemical applications. We understand the critical nature of supply continuity and are committed to delivering consistent quality through our robust process control systems.
We invite you to engage with our technical procurement team to discuss how this advanced carbonylation technology can be tailored to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits applicable to your specific product portfolio. We encourage potential partners to contact us for specific COA data and route feasibility assessments to validate the performance of these intermediates in your downstream processes. Together, we can drive innovation and efficiency in the global supply chain for fine chemicals and pharmaceutical ingredients.
