Advanced Ruthenium Carbene Catalysts: Enabling Efficient Commercial Scale-Up of Complex Pharmaceutical Intermediates
Advanced Ruthenium Carbene Catalysts: Enabling Efficient Commercial Scale-Up of Complex Pharmaceutical Intermediates
The landscape of organometallic catalysis has been fundamentally transformed by the innovations detailed in Chinese Patent CN1990493A, which discloses a novel class of high metathesis activity ruthenium and osmium metal carbene complexes. These compounds represent a significant leap forward in synthetic efficiency, offering unprecedented stability in the presence of diverse functional groups while maintaining exceptional catalytic activity for olefin metathesis reactions. Unlike previous generations of catalysts that suffered from slow initiation rates and complex multi-step syntheses, these new complexes facilitate the rapid transformation of non-bent cyclic and acyclic olefins into valuable pharmaceutical intermediates and fine chemicals. The patent highlights a robust methodology for preparing these carbene compounds using diazo compounds and neutral electron donor ligand exchange, ensuring that the resulting catalysts are not only highly active but also practically viable for industrial applications. This technological breakthrough addresses critical bottlenecks in the production of active pharmaceutical ingredients (APIs) by enabling cleaner reaction profiles and higher throughput.
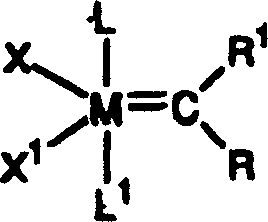
For research and development directors seeking to optimize synthetic routes, the implications of this patent are profound. The ability to catalyze ring-opening metathesis polymerization (ROMP), ring-closing metathesis (RCM), and cross-metathesis with such high fidelity allows for the construction of complex molecular architectures that were previously difficult or impossible to access efficiently. The structural versatility of these catalysts, defined by the general formula where M is osmium or ruthenium and ligands can be tuned for specific electronic and steric environments, provides a customizable platform for process chemists. By leveraging these high-activity species, manufacturers can achieve substantial cost reduction in pharmaceutical intermediate manufacturing through reduced catalyst loading and shorter reaction cycles. Furthermore, the stability of these complexes under ambient conditions simplifies handling and storage, removing the need for extreme inert atmosphere protocols that often complicate scale-up efforts in commercial facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the developments described in this patent, the field of olefin metathesis was largely dominated by vinyl alkylidene complexes, such as those depicted in earlier patents like US 5,312,940. These conventional catalysts, while functional, presented two distinct and severe shortcomings that hindered their widespread adoption in cost-sensitive commercial manufacturing. Firstly, the preparation of these vinyl alkylidene complexes was inherently inefficient, requiring rapid but multistep synthetic sequences that were time-consuming, expensive, and often resulted in low overall yields due to purification losses at each stage. Secondly, and perhaps more critically for process efficiency, the velocity of initiation for these traditional vinyl alkylidene complexes was remarkably low. This slow initiation kinetics led to the formation of polymers with broad molecular weight distributions in ROMP reactions and necessitated excessively long reaction times for RCM processes, thereby reducing reactor throughput and increasing energy consumption. The combination of difficult synthesis and sluggish performance made these older catalysts suboptimal for the rigorous demands of modern pharmaceutical supply chains.
The Novel Approach
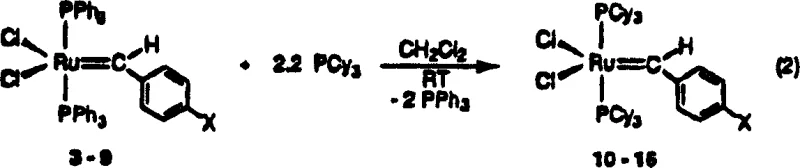
The novel approach introduced in CN1990493A overcomes these historical barriers by utilizing specific alkylidene moieties where R1 is hydrogen and R is selected from hydrogen, substituted or unsubstituted alkyl, or aryl groups. This structural modification results in complexes that possess unusually high initiation velocities, quantitatively demonstrated to be approximately 1000 times faster than their vinyl alkylidene counterparts. The synthesis strategy is elegantly simplified, often proceeding through a one-pot reaction method involving diazo compounds and neutral electron donors, which drastically streamlines the production workflow. For instance, the conversion of triphenylphosphine-stabilized precursors to the highly active tricyclohexylphosphine derivatives can be achieved at room temperature with high yields, as illustrated in the reaction schemes provided in the patent documentation. This shift from complex, multi-step preparations to streamlined, high-yield protocols directly translates to enhanced supply chain reliability and reduced lead times for high-purity catalysts.
Mechanistic Insights into Ruthenium-Catalyzed Olefin Metathesis
The superior performance of these ruthenium carbene complexes is rooted in their unique electronic and steric properties, which facilitate a highly efficient catalytic cycle. The mechanism involves the coordination of the olefin substrate to the ruthenium center, followed by the formation of a metallacyclobutane intermediate, which subsequently collapses to release the metathesized product and regenerate the active carbene species. The key to the high activity lies in the lability of the phosphine ligands; the substitution of triphenylphosphine with bulky tricyclohexylphosphine (PCy3) ligands creates a steric environment that promotes the dissociation of one phosphine ligand, generating the coordinatively unsaturated 14-electron species required for olefin binding. This dissociation step is the rate-determining factor in many metathesis reactions, and the specific ligand architecture described in the patent optimizes this equilibrium to favor the active species without compromising the thermal stability of the resting state. Consequently, the catalyst remains stable in solution even at elevated temperatures or in the presence of protic solvents, a feature that is rare for such reactive transition metal centers.
Furthermore, the mechanistic robustness of these complexes extends to their tolerance of functional groups, which is a critical parameter for the synthesis of complex pharmaceutical intermediates. Traditional early transition metal catalysts were often incompatible with polar functional groups such as alcohols, amines, and carboxylic acids, necessitating tedious protection and deprotection steps that added cost and waste to the process. In contrast, the ruthenium and osmium carbene compounds disclosed herein maintain their activity in the presence of these groups, allowing for direct functionalization. The patent data indicates that the alkylidene unit is perpendicular to the P-Ru-P face, minimizing electronic interference from the ligands and allowing the metal center to interact selectively with the olefinic bond. This precise geometric arrangement ensures that side reactions are minimized, leading to cleaner impurity profiles and simplifying downstream purification processes, which is a major concern for regulatory compliance in API manufacturing.
How to Synthesize High Activity Ruthenium Carbene Complexes Efficiently
The synthesis of these high-performance catalysts is designed for scalability and operational simplicity, making it accessible for both laboratory research and industrial production. The process typically begins with the reaction of a ruthenium precursor, such as RuCl2(PPh3)3, with a diazo compound at low temperatures to generate an intermediate alkylidene species. This intermediate is then subjected to a ligand exchange reaction with a neutral electron donor, preferably a bulky phosphine like tricyclohexylphosphine, to yield the final active catalyst. The detailed standardized synthesis steps see the guide below, which outlines the specific stoichiometry, temperature controls, and workup procedures required to achieve the high purity and activity levels described in the patent. This streamlined approach eliminates the need for specialized equipment beyond standard Schlenk techniques, facilitating easy technology transfer from R&D to commercial manufacturing sites.
- React RuCl2(PPh3)3 with appropriate diazo compounds (e.g., phenyl diazomethane) at low temperatures (-78°C to -50°C) to form the initial triphenylphosphine-stabilized alkylidene intermediate.
- Perform a ligand exchange reaction by treating the intermediate with excess tricyclohexylphosphine (PCy3) at room temperature in dichloromethane to replace PPh3 ligands.
- Isolate the final purple microcrystalline solid product via filtration and washing with methanol or acetone, yielding air-stable catalysts suitable for ROMP and RCM reactions.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers transformative benefits that extend far beyond simple reaction kinetics. The primary advantage lies in the drastic simplification of the catalyst synthesis itself; by moving from a multi-step, low-yield process to a high-yield, one-pot methodology, manufacturers can significantly reduce the cost of goods sold (COGS) associated with catalyst production. This efficiency gain is compounded by the catalyst's high turnover number and rapid initiation rate, which allow for lower catalyst loadings to achieve the same conversion levels as older technologies. Reduced catalyst loading not only lowers raw material costs but also minimizes the burden on downstream purification systems, as there is less residual metal to remove from the final product. This leads to substantial cost savings in manufacturing by reducing the consumption of scavengers and chromatography media, which are often significant expense drivers in fine chemical production.
- Cost Reduction in Manufacturing: The implementation of these high-activity ruthenium complexes enables a leaner manufacturing process by eliminating the need for expensive protecting group strategies. Because the catalysts are tolerant of diverse functional groups, synthetic routes can be shortened, reducing the total number of unit operations and the associated labor and utility costs. Furthermore, the high stability of the catalysts in air and solution reduces waste generated from catalyst decomposition, ensuring that every gram of purchased material contributes effectively to the reaction. This qualitative improvement in process efficiency translates directly to a more competitive pricing structure for the final pharmaceutical intermediates, allowing companies to maintain healthy margins even in volatile market conditions.
- Enhanced Supply Chain Reliability: The robustness of these catalysts ensures consistent batch-to-batch performance, which is critical for maintaining uninterrupted supply chains. Unlike sensitive catalysts that may degrade during storage or transport, these complexes are stable solids that can be stockpiled without significant loss of activity, providing a buffer against supply disruptions. The use of readily available starting materials, such as commercially sourced phosphines and diazo compounds, further secures the supply chain by reducing dependence on exotic or single-source reagents. This reliability allows procurement teams to negotiate better terms with suppliers and plan production schedules with greater confidence, knowing that the catalytic performance will remain consistent regardless of minor variations in raw material sourcing.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this technology aligns perfectly with green chemistry principles. The high atom economy of metathesis reactions, combined with the reduced need for solvent-intensive purification steps, results in a significantly lower environmental footprint. The ability to run reactions in less hazardous solvents or even under solvent-free conditions, as suggested by the patent's versatility, further enhances the sustainability profile of the manufacturing process. Scalability is assured by the straightforward nature of the synthesis and the thermal stability of the catalysts, which can withstand the exotherms and mixing challenges inherent in large-scale reactors. This makes the commercial scale-up of complex polymer additives and pharmaceutical intermediates feasible without requiring prohibitive capital investment in specialized containment or cooling infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these ruthenium carbene catalysts in industrial settings. These answers are derived directly from the experimental data and embodiments provided in the patent literature, ensuring accuracy and relevance for decision-makers evaluating this technology for adoption. Understanding these nuances is essential for integrating this catalytic system into existing workflows and maximizing its potential value.
Q: How does the initiation rate of these new ruthenium complexes compare to traditional vinyl alkylidene catalysts?
A: The disclosed ruthenium carbene complexes exhibit an initiation rate approximately 1000 times faster than corresponding vinyl alkylidene complexes, significantly reducing reaction times for ring-opening metathesis polymerization (ROMP) and ring-closing metathesis (RCM).
Q: Are these catalysts stable in the presence of sensitive functional groups?
A: Yes, the complexes are exceptionally stable in the presence of various functional groups including alcohols, amines, esters, and ketones, allowing for the direct synthesis of functionalized intermediates without extensive protecting group strategies.
Q: What represents the primary commercial advantage of this synthesis method?
A: The primary advantage is the simplified one-pot synthesis route which eliminates multi-step purification processes, thereby drastically reducing production costs and improving overall yield consistency for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ruthenium Carbene Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that advanced catalysis plays in driving innovation within the pharmaceutical and fine chemical industries. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1990493A are fully realized in practical, large-volume manufacturing. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of analyzing trace metal residues and organic impurities to meet the most demanding global regulatory standards. We understand that the transition from bench-scale discovery to commercial supply requires not just chemical expertise but also a deep commitment to quality assurance and process safety, values that are embedded in our operational DNA.
We invite you to collaborate with our technical procurement team to explore how these high-activity ruthenium carbene complexes can optimize your specific synthetic routes. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into how switching to this catalytic system might impact your overall production economics and timeline. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us help you leverage this cutting-edge technology to enhance your product portfolio, reduce manufacturing costs, and secure a resilient supply chain for your critical pharmaceutical intermediates.
