Revolutionizing Trimellitic Acid Production: A Deep Dive into Iron-Promoted Liquid Phase Oxidation for Industrial Scale-Up
Revolutionizing Trimellitic Acid Production: A Deep Dive into Iron-Promoted Liquid Phase Oxidation for Industrial Scale-Up
The global demand for high-purity aromatic polycarboxylic acids continues to surge, driven by their critical applications in advanced polymer additives and pharmaceutical intermediates. In this landscape, the technological breakthrough detailed in patent CN101980997A represents a paradigm shift in the manufacturing of trimellitic acid (TMA). Traditionally, the industry has relied on the oxidation of pseudocumene or 2,4-dimethylbenzaldehyde, processes often plagued by solvent recovery costs and inconsistent impurity profiles. However, this novel methodology introduces a continuous liquid-phase oxidation system utilizing 3,4-dimethylbenzaldehyde (3,4-DBAL) as the primary feedstock. By ingeniously incorporating a trace amount of iron into a manganese-bromine catalyst system within an aqueous solvent, the process achieves unprecedented selectivity. For procurement leaders and technical directors seeking a reliable trimellitic acid supplier, understanding the mechanistic advantages of this iron-promoted pathway is essential for securing long-term supply chain stability and cost efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of trimellitic acid has predominantly utilized 2,4-dimethylbenzaldehyde or pseudocumene as starting materials, typically employing cobalt-manganese-bromine catalyst systems in acetic acid solvents. While established, these legacy processes suffer from inherent thermodynamic and kinetic limitations that impact overall process economics. A significant drawback is the substantial formation of organobromine byproducts, such as trimellitic acid bromide, which not only lowers the final yield but also complicates downstream purification. Furthermore, the consumption of bromine in these side reactions necessitates frequent and costly replenishment of the catalyst system. From an environmental compliance perspective, the discharge of acetic acid solvent and brominated organic waste places a heavy burden on wastewater treatment infrastructure. For supply chain managers, these factors translate into volatile production costs and potential regulatory bottlenecks that threaten the continuity of high-purity trimellitic acid supplies.
The Novel Approach
The innovative process described in the patent data fundamentally alters the reaction landscape by switching the substrate to 3,4-dimethylbenzaldehyde and, crucially, introducing iron as a co-catalyst. Initial investigations revealed that applying standard manganese-bromine catalysis to the 3,4-isomer resulted in poor conversion rates, failing to reach commercially viable yields. However, the discovery that adding minute quantities of iron (specifically between 0.0001 to 0.0015 parts by mass relative to the solvent) dramatically reverses this trend is a game-changer. This modified catalyst system operates effectively in an aqueous environment at temperatures between 200°C and 250°C, eliminating the need for flammable organic solvents. The result is a robust continuous flow process that not only boosts yields to over 90% but also drastically suppresses the formation of stubborn impurities. This approach offers a clear pathway for cost reduction in pharma intermediate manufacturing by simplifying the purification train and extending catalyst life.
Mechanistic Insights into Fe-Promoted Liquid Phase Oxidation
To fully appreciate the technical superiority of this method, one must examine the catalytic cycle at a molecular level. The oxidation of dimethylbenzaldehydes proceeds through a free-radical mechanism where the metal catalysts facilitate the decomposition of hydroperoxides. In the absence of iron, the 3,4-dimethylbenzaldehyde substrate appears to deactivate the standard manganese-bromine redox cycle, leading to the accumulation of partial oxidation products like carboxyphthalides. The introduction of iron ions acts as a powerful promoter, likely by accelerating the initiation step of the radical chain reaction or by stabilizing specific transition states unique to the 3,4-substitution pattern. This synergistic effect ensures that the oxidation proceeds rapidly to the tricarboxylic acid stage rather than stalling at intermediate stages. Consequently, the process demonstrates exceptional tolerance to variations in oxygen pressure and temperature, making it highly suitable for large-scale commercial scale-up of complex pharmaceutical intermediates.
Impurity control is perhaps the most compelling aspect of this mechanistic design. In conventional processes using the 2,4-isomer, the electrophilic bromination of the aromatic ring is a persistent side reaction, leading to brominated TMA derivatives that are difficult to separate. The data indicates that the 3,4-isomer, under these specific iron-promoted conditions, exhibits a significantly lower propensity for ring bromination. 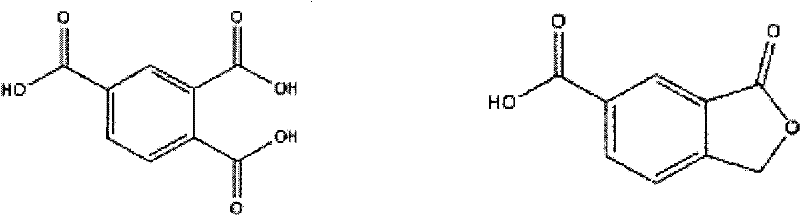 As illustrated in the structural comparison, the primary impurity concern shifts towards carboxyphthalides (lactone forms), but even these are minimized through the optimized two-stage reactor configuration. The high retention rate of bromide ions (approximately 80% compared to 50% in older methods) confirms that the bromine remains active as a catalyst rather than being consumed as a contaminant. This mechanistic fidelity ensures that the final product meets stringent purity specifications required for sensitive applications like polyimide films and plasticizers.
As illustrated in the structural comparison, the primary impurity concern shifts towards carboxyphthalides (lactone forms), but even these are minimized through the optimized two-stage reactor configuration. The high retention rate of bromide ions (approximately 80% compared to 50% in older methods) confirms that the bromine remains active as a catalyst rather than being consumed as a contaminant. This mechanistic fidelity ensures that the final product meets stringent purity specifications required for sensitive applications like polyimide films and plasticizers.
How to Synthesize Trimellitic Acid Efficiently
Implementing this synthesis route requires precise control over reactor parameters and catalyst stoichiometry to maximize the benefits of the iron promotion effect. The process is designed as a continuous two-stage operation, where the first reactor handles the bulk of the aldehyde oxidation, and the second reactor ensures the completion of the reaction to the tri-acid state. Operators must carefully prepare the catalyst solution, ensuring that the iron concentration remains within the narrow window of 2 to 10 ppm relative to the water solvent; deviations outside this range can lead to diminished returns or increased sludge formation. The following guide outlines the standardized operational procedure derived from the patent examples, providing a roadmap for R&D teams looking to replicate this high-efficiency pathway in pilot or production settings. Detailed standard operating procedures for this synthesis are provided below.
- Prepare the catalyst solution by mixing water, hydrobromic acid, manganese bromide, and a precise trace amount of ferric bromide (0.0001-0.0015 parts by mass relative to solvent).
- Feed 3,4-dimethylbenzaldehyde and the catalyst solution into the first stage of a two-stage continuous zirconium reactor system maintained at 200-250°C.
- Introduce compressed air to maintain oxygen concentration, transfer the reaction mixture to the second reactor with additional bromine supply, and extract the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this iron-promoted aqueous oxidation process offers tangible strategic advantages beyond mere technical novelty. The shift from organic solvents to water fundamentally changes the safety and environmental profile of the manufacturing site, reducing insurance premiums and regulatory compliance costs associated with volatile organic compound (VOC) emissions. Moreover, the dramatic improvement in catalyst efficiency directly impacts the bottom line. Because the bromide ion retention rate is significantly higher, the frequency of purchasing expensive bromine supplements is drastically reduced. This creates a more predictable cost structure, shielding the organization from market volatility in raw material pricing. Additionally, the simplified impurity profile means that downstream purification units, such as centrifuges and dryers, can operate at higher throughput rates with less energy consumption. These factors combine to create a resilient supply chain capable of meeting the rigorous demands of global API manufacturers.
- Cost Reduction in Manufacturing: The economic argument for adopting this technology is anchored in the substantial reduction of catalyst consumption and waste disposal fees. By retaining a high proportion of the bromine catalyst within the aqueous loop, the process eliminates the need for continuous, high-volume makeup of bromine salts, which are among the more expensive components of the catalyst cocktail. Furthermore, the use of water as a solvent removes the capital and operational expenditures associated with acetic acid recovery columns and solvent incineration. The reduction in organobromine byproducts also translates to lower costs in wastewater treatment, as the effluent load is significantly lighter. Collectively, these efficiencies drive down the unit cost of production, allowing for more competitive pricing strategies in the global market for specialty chemicals.
- Enhanced Supply Chain Reliability: Supply continuity is often jeopardized by complex purification bottlenecks or reliance on scarce reagents. This process mitigates those risks by utilizing readily available raw materials like 3,4-dimethylbenzaldehyde and common inorganic salts. The robustness of the iron-promoted catalyst system ensures consistent campaign lengths without frequent shutdowns for catalyst regeneration or reactor cleaning. This reliability is critical for long-term contracts with major pharmaceutical clients who require guaranteed delivery schedules. By minimizing the formation of hard-to-remove impurities, the manufacturing timeline is shortened, effectively reducing lead time for high-purity trimellitic acid batches. This agility allows suppliers to respond more quickly to fluctuations in market demand without compromising on quality standards.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen thermal and mixing challenges, but the continuous nature of this liquid-phase oxidation is inherently scalable. The use of water as a solvent provides excellent heat capacity, facilitating temperature control in large reactors and reducing the risk of thermal runaways. From an environmental standpoint, the process aligns perfectly with modern green chemistry principles by avoiding chlorinated or acidic organic solvents. The significant reduction in hazardous organic bromide waste simplifies the permitting process for new production lines and ensures compliance with increasingly strict environmental regulations. This future-proofs the manufacturing asset, ensuring that the facility remains operational and compliant for decades, thereby securing the long-term investment value for stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced oxidation technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing clarity on the operational nuances of the iron-promoted system. Understanding these details is vital for engineering teams evaluating the feasibility of retrofitting existing oxidation units or designing new grassroots facilities. The answers below reflect the consensus on performance metrics and impurity management strategies observed during the development of this process.
Q: Why is the addition of trace iron critical when using 3,4-dimethylbenzaldehyde as a raw material?
A: Unlike the conventional 2,4-isomer, the 3,4-dimethylbenzaldehyde substrate exhibits different reactivity. Without the addition of trace iron (2-10 ppm), the oxidation yield drops significantly below 70%. The iron promoter activates the catalytic cycle specifically for this isomer, restoring yields to over 90%.
Q: How does this process reduce environmental waste compared to traditional acetic acid methods?
A: This process utilizes water as the solvent instead of acetic acid, eliminating the need for solvent recovery distillation columns. Furthermore, the high retention rate of bromide ions (approx. 80%) means less catalyst is lost to organic bromide byproducts, significantly reducing the burden on wastewater treatment facilities.
Q: What is the primary impurity profile of the trimellitic acid produced via this method?
A: The process minimizes the formation of carboxyphthalides and phthalic acid derivatives. Specifically, the generation of trimellitic acid bromide is reduced to approximately one-ninth of the levels seen in conventional 2,4-isomer processes, resulting in a much cleaner crude product suitable for direct crystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trimellitic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of cutting-edge synthesis technologies like the iron-promoted oxidation of 3,4-dimethylbenzaldehyde is key to maintaining a competitive edge in the fine chemical sector. Our team of process engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of specialized intermediates or large-volume supply of commodity acids, our infrastructure is designed to support your growth with flexibility and precision.
We invite you to engage with our technical procurement team to discuss how this innovative manufacturing route can benefit your specific application. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic impact of switching to this high-efficiency process. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project requirements. Let us collaborate to optimize your supply chain and secure a sustainable source of high-quality chemical intermediates for your future projects.
