Advanced Resolution of Axially Chiral Oxindole-Styrenes for High-Value Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for accessing high-value chiral scaffolds, particularly those exhibiting axial chirality which are pivotal for advanced drug discovery. Patent CN111848322B introduces a groundbreaking approach to the synthesis and resolution of axially chiral oxindole-substituted styrene compounds, addressing a significant gap in current organic synthesis capabilities. This technology leverages a highly efficient organocatalytic resolution strategy that transforms readily available racemic precursors into optically pure intermediates with exceptional stereocontrol. By utilizing a specialized chiral phosphoric acid catalyst, the process operates under remarkably mild conditions, avoiding the harsh reagents often associated with traditional metal-catalyzed couplings. This innovation not only expands the chemical space available for medicinal chemists but also provides a scalable pathway for producing complex chiral building blocks essential for next-generation therapeutics and functional materials.
Furthermore, the utility of these resolved compounds extends beyond their role as static intermediates; they serve as potent precursors for novel chiral catalysts. The patent details a seamless derivatization pathway where the resolved styrene is converted into a chiral tertiary amine catalyst, which subsequently demonstrates superior performance in asymmetric [4+2] cyclization reactions. This dual utility—acting both as a high-purity intermediate and a catalyst precursor—significantly enhances the value proposition for supply chain managers and R&D directors alike. The ability to access such structurally diverse and complex molecules with high atom economy positions this technology as a critical asset for companies aiming to streamline their pipeline of chiral active pharmaceutical ingredients (APIs) and fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axially chiral styrene skeletons has been fraught with significant synthetic challenges, primarily relying on nucleophilic additions to alkynes or transition-metal-catalyzed coupling reactions. Literature precedents, such as those involving palladium-catalyzed allylation or titanium-mediated additions, often suffer from limited substrate scope and stringent reaction requirements. These conventional methods frequently necessitate the use of expensive transition metals, which introduces complex downstream purification burdens to meet strict residual metal specifications required in pharmaceutical manufacturing. Moreover, the reliance on stoichiometric chiral auxiliaries or harsh reaction conditions can lead to poor atom economy and generate substantial chemical waste. The scarcity of efficient methods specifically tailored for oxindole-substituted variants has further constrained the exploration of this chemical space, leaving a void in the availability of reliable pharmaceutical intermediate supplier options for these specific scaffolds.
The Novel Approach
In stark contrast, the methodology disclosed in CN111848322B utilizes a dynamic kinetic resolution or direct enantioselective functionalization strategy driven by organocatalysis. By employing a chiral phosphoric acid derived from a BINOL skeleton, the reaction proceeds through a highly organized transition state that effectively differentiates between enantiomers. This approach eliminates the need for transition metals entirely, thereby simplifying the purification process and reducing the environmental footprint of the synthesis. The reaction conditions are exceptionally mild, typically operating at temperatures between 0°C and 10°C in dichloromethane, which facilitates easier thermal management during scale-up. This novel route accommodates a wide array of substituents on the indole and phenyl rings, demonstrating remarkable versatility and robustness. Consequently, this method offers a viable solution for cost reduction in fine chemical manufacturing by minimizing raw material costs associated with precious metals and reducing waste disposal expenses.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Resolution
The core of this technological breakthrough lies in the precise mechanistic interaction between the chiral phosphoric acid catalyst and the substrate. The catalyst, typically a BINOL-derived phosphoric acid substituted with bulky 2-naphthyl groups, acts as a bifunctional activator. It simultaneously activates the electrophilic and nucleophilic components of the reaction mixture through a network of hydrogen bonding interactions. The acidic proton of the phosphate group engages with the basic sites on the substrate, while the phosphoryl oxygen can stabilize developing positive charges or interact with other hydrogen bond donors. This dual activation lowers the energy barrier for the desired transformation while the bulky aryl groups on the catalyst backbone create a well-defined chiral pocket. This steric environment forces the substrate to adopt a specific orientation, ensuring that the reaction occurs exclusively on one face of the prochiral center or effectively resolves the racemic mixture.
This rigorous control over the stereochemical outcome is evidenced by the high enantiomeric excess (ee) values reported, often exceeding 90% and reaching up to 98% for optimized substrates. The mechanism avoids the formation of racemic byproducts that are common in non-catalyzed thermal reactions. Furthermore, the inclusion of 3A molecular sieves in the reaction mixture plays a critical role in scavenging trace water, which could otherwise deactivate the sensitive phosphoric acid catalyst or hydrolyze reactive intermediates. This attention to detail in the reaction design ensures consistent reproducibility and high purity profiles. For R&D directors, understanding this mechanism underscores the reliability of the process for generating high-purity axially chiral compounds, which is paramount for maintaining the integrity of subsequent synthetic steps in a drug discovery campaign.
How to Synthesize Axially Chiral Oxindole-Styrene Efficiently
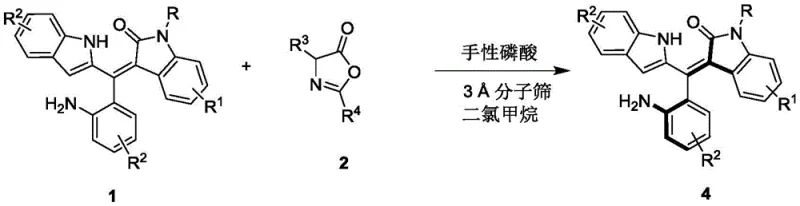
The synthesis protocol outlined in the patent provides a clear and actionable roadmap for laboratory and pilot-scale production. The process begins with the preparation of the reaction vessel under anhydrous conditions, emphasizing the importance of moisture control for optimal catalyst performance. The reactants, comprising the racemic oxindole-styrene precursor and the specific coupling partner, are dissolved in dry dichloromethane. The addition of activated 3A molecular sieves is a critical step to maintain the anhydrous environment throughout the reaction duration. Following this, the chiral phosphoric acid catalyst is introduced at a loading of approximately 10 mol%, and the mixture is stirred at a controlled low temperature, preferably 0°C. Reaction progress is monitored via thin-layer chromatography (TLC) to ensure complete conversion before workup.
- Prepare the reaction mixture by combining racemic Formula 1 compound and Formula 2 compound in dichloromethane solvent with 3A molecular sieves.
- Add 10 mol% of chiral phosphoric acid catalyst (Formula 3) and stir the reaction at 0°C to 10°C until TLC indicates completion.
- Filter off the molecular sieves, concentrate the filtrate, and purify the residue via silica gel column chromatography to isolate the axially chiral Formula 4 compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers compelling advantages that directly address the pain points of procurement and supply chain management in the specialty chemical sector. The shift from metal-catalyzed processes to organocatalysis fundamentally alters the cost structure and risk profile of the manufacturing operation. By removing the dependency on volatile precious metal markets and eliminating the need for specialized metal-scavenging resins during purification, the overall cost of goods sold (COGS) is significantly optimized. Additionally, the mild reaction conditions reduce energy consumption and equipment wear, contributing to a more sustainable and economically viable production model. The robustness of the catalyst system also implies a longer shelf-life for reagents and greater flexibility in scheduling production runs, enhancing overall supply chain resilience.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts such as palladium or titanium removes a major cost driver associated with both raw material acquisition and waste treatment. Traditional metal-catalyzed routes often require expensive ligands and rigorous purification steps to meet regulatory limits on heavy metal residues, which adds substantial time and expense to the manufacturing process. In contrast, the organocatalytic nature of this resolution allows for simpler workup procedures, often requiring only filtration and standard chromatography. This simplification translates to reduced labor hours, lower solvent consumption, and decreased waste disposal fees, resulting in substantial cost savings without compromising on the quality or purity of the final product.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are structurally simple and commercially accessible, reducing the risk of supply bottlenecks that often plague complex chiral syntheses. Unlike processes that rely on bespoke ligands or unstable organometallic reagents, the reagents used here are stable and can be sourced from multiple vendors, ensuring continuity of supply. The mild reaction conditions also mean that the process is less sensitive to minor fluctuations in utility supplies, such as cooling water temperature, making it more robust for large-scale operations. This reliability is crucial for maintaining consistent delivery schedules to downstream customers, thereby strengthening long-term partnerships and reducing the lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction parameters that are easily transferable from gram-scale laboratory experiments to multi-kilogram pilot plants. The use of dichloromethane, while requiring careful handling, is a well-understood solvent in industrial settings with established recovery and recycling protocols. Furthermore, the high atom economy and lack of toxic metal waste align perfectly with modern green chemistry principles and increasingly stringent environmental regulations. This compliance reduces the regulatory burden on manufacturing sites and minimizes the risk of production shutdowns due to environmental non-compliance, ensuring a stable and sustainable supply of complex intermediates for the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These insights are derived directly from the experimental data and scope defined within the patent documentation, providing clarity on the practical aspects of adopting this synthetic route. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into their existing workflows or for procurement specialists assessing the quality standards of the resulting materials.
Q: What is the enantioselectivity achieved in this resolution process?
A: The process achieves high enantioselectivity, with ee values reaching up to 98% depending on the substrate substituents, as demonstrated in the patent examples.
Q: Can the resolved product be further derivatized?
A: Yes, the resolved axially chiral styrene (Formula 4) can be derivatized into chiral tertiary amine catalysts (Formula 5) which show excellent activity in asymmetric [4+2] cyclization reactions.
Q: What are the optimal reaction conditions for this resolution?
A: The optimal conditions involve using dichloromethane as the solvent, 3A molecular sieves as an additive, and a reaction temperature of 0°C with a chiral phosphoric acid catalyst loading of 10 mol%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Oxindole-Styrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality chiral intermediates for the development of innovative pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demands of both clinical trial material supply and full-scale commercial manufacturing. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify enantiomeric excess and chemical purity. Our expertise in chiral resolution and organocatalysis allows us to optimize these patented routes for maximum efficiency and yield, providing our partners with a competitive edge in the marketplace.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis that details how implementing this resolution method can optimize your specific supply chain. We encourage you to contact our technical procurement team to request specific COA data for our catalog items or to discuss route feasibility assessments for your custom synthesis projects. By partnering with us, you gain access to a reliable source of high-value chiral building blocks that will accelerate your drug discovery timelines and enhance the quality of your final therapeutic products.
