Advanced Rhodium Catalysis for Commercial Scale Isothiocyanate Production
Advanced Rhodium Catalysis for Commercial Scale Isothiocyanate Production
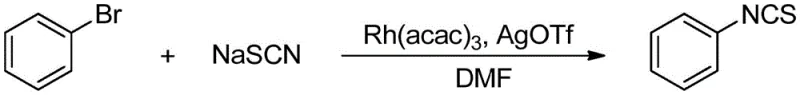
The chemical industry is constantly seeking safer and more efficient pathways to access high-value heterocyclic precursors, and the technology disclosed in patent CN114716356A represents a significant leap forward in this domain. This patent introduces a robust rhodium-catalyzed methodology for synthesizing isothiocyanate compounds, utilizing readily available halogenated hydrocarbons and stable inorganic thiocyanate salts as starting materials. Unlike legacy processes that depend on hazardous reagents, this novel approach employs Rh(acac)3 as a catalyst in conjunction with silver salts within a DMF solvent system to achieve direct coupling under mild thermal conditions. The strategic shift towards using abundant feedstocks not only aligns with green chemistry principles but also addresses critical supply chain vulnerabilities associated with specialized reagents. For R&D directors and procurement strategists, this innovation offers a compelling alternative that balances high synthetic efficiency with operational safety and cost-effectiveness. By leveraging transition metal catalysis, the method achieves impressive yields across a diverse range of substrates, establishing a new benchmark for the manufacturing of these vital pharmaceutical and agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of isothiocyanates has been plagued by significant safety and environmental challenges inherent to traditional synthetic routes. Conventional methodologies typically rely on the reaction of primary amines with highly toxic reagents such as thiophosgene, carbon disulfide, or phenyl thiochloroformate, which necessitate rigorous safety protocols and expensive containment infrastructure. These processes often operate under harsh conditions requiring elevated temperatures and extended reaction times, leading to increased energy consumption and potential degradation of sensitive functional groups on the molecular scaffold. Furthermore, the handling of volatile and poisonous gases like thiophosgene poses severe occupational health risks, complicating regulatory compliance and increasing insurance liabilities for manufacturing facilities. Alternative approaches, such as those utilizing alkyne halides, suffer from the extreme difficulty in synthesizing the starting materials themselves, resulting in low overall yields and narrow substrate applicability. Consequently, the reliance on these outdated techniques creates a bottleneck in the supply chain, limiting the ability to scale production efficiently while maintaining strict purity specifications required by the pharmaceutical sector.
The Novel Approach
In stark contrast to these legacy issues, the rhodium-catalyzed protocol described in the patent offers a streamlined and environmentally benign solution that directly couples halogenated hydrocarbons with thiocyanate salts. This innovative strategy eliminates the need for toxic thiophosgene derivatives, replacing them with stable, non-volatile inorganic salts like sodium thiocyanate or potassium thiocyanate that are easy to handle and store. The reaction proceeds smoothly at moderate temperatures between 50°C and 80°C, significantly reducing the thermal stress on the reaction mixture and minimizing the formation of thermal byproducts. By utilizing common aryl halides including chlorides, bromides, and iodides as electrophiles, the method unlocks access to a vast library of commercially available starting materials, thereby enhancing supply chain resilience. The integration of a rhodium catalyst system ensures high conversion rates and excellent selectivity, delivering target isothiocyanates with high purity suitable for downstream applications without extensive purification burdens. This paradigm shift not only simplifies the operational workflow but also drastically reduces the environmental footprint of the synthesis, making it an ideal candidate for modern sustainable manufacturing practices.
Mechanistic Insights into Rh(acac)3-Catalyzed C-S Bond Formation
The core of this technological advancement lies in the sophisticated interplay between the rhodium catalyst and the silver salt additive, which facilitates the challenging formation of carbon-sulfur bonds under mild conditions. The catalytic cycle likely initiates with the activation of the rhodium precursor Rh(acac)3, which interacts with the silver salt to generate a highly reactive cationic rhodium species capable of oxidative addition into the carbon-halogen bond. This step is crucial as it overcomes the kinetic barrier associated with unactivated aryl chlorides and bromides, which are traditionally less reactive in nucleophilic substitution reactions. Once the organorhodium intermediate is formed, the thiocyanate anion coordinates to the metal center, positioning the sulfur atom for subsequent migratory insertion or reductive elimination. The presence of the silver salt serves a dual purpose: it acts as a halide scavenger to drive the equilibrium forward and potentially stabilizes the active catalytic species against deactivation. This mechanistic pathway allows for the tolerance of various electronic environments on the aromatic ring, enabling the successful transformation of both electron-rich and electron-deficient substrates without compromising yield. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters for specific substrates, ensuring consistent quality and reproducibility during scale-up operations.
Impurity control is another critical aspect where this catalytic system excels, primarily due to the mild reaction conditions and the high specificity of the transition metal catalyst. Traditional high-temperature methods often promote side reactions such as polymerization or decomposition of the isothiocyanate functionality, leading to complex impurity profiles that are difficult to separate. In this rhodium-mediated process, the temperature is strictly maintained between 50°C and 80°C, which is sufficiently low to prevent thermal degradation yet high enough to ensure complete conversion within a reasonable timeframe of 5 to 10 hours. The use of DMF as a polar aprotic solvent further enhances the solubility of the inorganic thiocyanate salts, ensuring homogeneous reaction conditions that minimize localized hot spots and uneven reactivity. Moreover, the catalyst loading is optimized to be quite low, typically in the range of 0.02 to 0.05 molar equivalents, which reduces the risk of metal contamination in the final product. This precise control over reaction variables results in a cleaner crude reaction mixture, simplifying the downstream purification process and ensuring that the final isothiocyanate compounds meet the stringent purity requirements demanded by regulatory bodies for pharmaceutical intermediates.
How to Synthesize Isothiocyanate Compounds Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to a standardized protocol that maximizes yield while ensuring safety and reproducibility. The process begins with the careful weighing and charging of the halogenated hydrocarbon substrate and the inorganic thiocyanate salt into a dry reaction vessel equipped with magnetic stirring and temperature control. Following the addition of the rhodium catalyst and the silver salt additive, anhydrous DMF is introduced as the solvent to create a homogeneous reaction mixture that facilitates efficient mass transfer. The detailed standardized synthesis steps see the guide below for exact molar ratios and specific workup procedures tailored to different substrate classes. Adhering to these precise instructions is essential for achieving the high isolated yields reported in the patent data, which consistently exceed 85% across a wide variety of structural motifs. Proper execution of this protocol ensures that the benefits of this green chemistry approach are fully realized in practical applications.
- Charge a reaction vessel with the selected halogenated hydrocarbon substrate and an inorganic thiocyanate salt such as NaSCN or KSCN.
- Add the rhodium catalyst Rh(acac)3 and a silver salt additive like AgOTf or AgNO3 to the mixture under inert atmosphere conditions.
- Dissolve the reagents in DMF solvent and heat the mixture to 50-80°C for 5 to 10 hours, followed by concentration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this rhodium-catalyzed technology offers profound advantages that extend far beyond simple chemical efficiency, directly impacting the bottom line and operational stability of manufacturing organizations. The substitution of hazardous reagents like thiophosgene with benign inorganic salts fundamentally alters the risk profile of the production facility, potentially lowering insurance premiums and reducing the capital expenditure required for specialized safety equipment. Furthermore, the reliance on commodity chemicals such as aryl halides and simple thiocyanates ensures a stable and diversified supply base, mitigating the risks associated with single-source suppliers of exotic reagents. This robustness in raw material sourcing translates directly into enhanced supply chain reliability, allowing procurement managers to negotiate better terms and secure long-term contracts with confidence. The simplified workflow also意味着 reduced labor costs and shorter batch cycles, contributing to overall operational excellence and competitiveness in the global market for fine chemical intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive and dangerous reagents, which often carry high procurement and disposal costs due to their hazardous nature. By utilizing cheap and readily available halogenated hydrocarbons alongside stable inorganic salts, the raw material cost structure is significantly optimized, leading to substantial savings per kilogram of produced isothiocyanate. Additionally, the mild reaction conditions reduce energy consumption compared to traditional high-temperature processes, further lowering the utility costs associated with heating and cooling large-scale reactors. The high yields achieved minimize waste generation and maximize the throughput of the existing infrastructure, effectively increasing the capacity of the plant without the need for major capital investment. These cumulative factors result in a markedly lower cost of goods sold, providing a competitive edge in price-sensitive markets while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by the use of universally available starting materials that are produced by multiple vendors globally, reducing dependency on niche chemical suppliers. The stability of the inorganic thiocyanate salts allows for bulk purchasing and long-term storage without significant degradation, enabling manufacturers to build strategic inventory buffers against market fluctuations. Moreover, the robustness of the catalytic system means that minor variations in raw material quality can be tolerated without catastrophic failure of the reaction, ensuring consistent production output even when supply chains are stressed. This reliability is crucial for meeting the just-in-time delivery requirements of downstream pharmaceutical clients, fostering stronger partnerships and trust between suppliers and buyers. Ultimately, a more predictable and stable supply chain reduces the administrative burden of expediting orders and managing shortages, allowing teams to focus on strategic growth initiatives.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of standard equipment and solvents that are already prevalent in the fine chemical industry, avoiding the need for custom-engineered reactors. The green nature of the synthesis, characterized by the absence of toxic gases and the use of recyclable catalysts, aligns perfectly with increasingly stringent environmental regulations and corporate sustainability goals. Waste streams generated from this process are easier to treat and dispose of compared to those containing heavy metals or persistent organic pollutants, simplifying compliance with local and international environmental standards. The ability to run the reaction at atmospheric pressure and moderate temperatures also enhances operational safety during scale-up, reducing the likelihood of runaway reactions or equipment failures. These factors collectively make the technology highly attractive for large-scale manufacturing, ensuring long-term viability and social license to operate in a regulated environment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-catalyzed synthesis method, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational parameters and benefits for stakeholders evaluating the technology for potential adoption or licensing. Understanding these details is essential for making informed decisions about integrating this process into existing manufacturing portfolios. The answers reflect the specific advantages and constraints identified in the intellectual property documentation.
Q: What are the primary safety advantages of this Rh-catalyzed method over traditional isothiocyanate synthesis?
A: Traditional methods often rely on highly toxic reagents like thiophosgene or carbon disulfide, which pose significant health risks and require specialized containment equipment. The patented Rh-catalyzed approach utilizes stable, inexpensive inorganic thiocyanate salts and common halogenated hydrocarbons, drastically reducing operational hazards and simplifying waste management protocols.
Q: Does this catalytic system support substrates with sensitive functional groups?
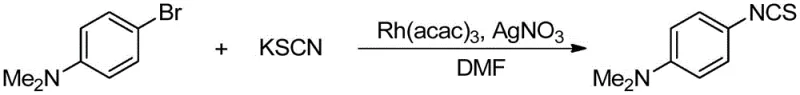
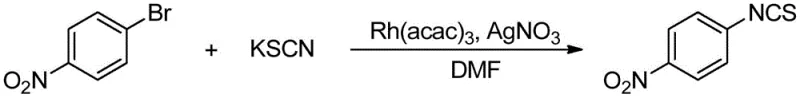
A: Yes, the method demonstrates excellent substrate universality, successfully tolerating electron-donating groups like dimethylamino and electron-withdrawing groups like nitro and fluoro substituents. The mild reaction temperatures ranging from 50°C to 80°C prevent the decomposition of thermally sensitive moieties, ensuring high purity profiles for complex pharmaceutical intermediates.
Q: How does the choice of silver salt impact the reaction efficiency?
A: The process utilizes silver salts such as AgOTf or AgNO3 as essential additives to facilitate the halide abstraction and activate the catalytic cycle. Optimization data indicates that different silver salts can be tuned depending on the specific halogenated substrate used, allowing for flexibility in reagent sourcing and cost management during scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isothiocyanate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN114716356A and are fully equipped to bring such innovations to commercial reality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are designed to handle complex organometallic chemistry with precision, supported by rigorous QC labs that enforce stringent purity specifications for every batch released. We understand that consistency and quality are paramount in the pharmaceutical supply chain, and our dedicated technical team works tirelessly to optimize every parameter of the synthesis to meet your exact requirements. By partnering with us, you gain access to a wealth of chemical expertise and manufacturing capacity that can accelerate your product development timelines significantly.
We invite you to engage with our technical procurement team to discuss how this rhodium-catalyzed route can be tailored to your specific project needs and cost targets. Request a Customized Cost-Saving Analysis today to quantify the potential economic benefits of switching to this greener, more efficient synthesis method for your isothiocyanate intermediates. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate the viability of this technology for your portfolio. Let us help you navigate the complexities of modern chemical manufacturing and secure a reliable, high-quality supply of critical intermediates for your business growth.
