Revolutionizing Alcohol Oxidation: Scalable Synthesis of High-Purity TEMPO Derivatives for Industrial Applications
Revolutionizing Alcohol Oxidation: Scalable Synthesis of High-Purity TEMPO Derivatives for Industrial Applications
The landscape of organic synthesis is constantly evolving, driven by the urgent need for greener, more efficient catalytic systems that can withstand the rigors of industrial scale-up. A pivotal advancement in this domain is detailed in patent CN112851571A, which discloses a sophisticated preparation method for 2-(2,2,6,6-tetramethylpiperidine nitroxide radical-4-subunit) acetic acid derivatives. This technology represents a significant leap forward from traditional oxidation methods, addressing critical pain points such as catalyst recovery, environmental impact, and operational stability. By modifying the core TEMPO structure with specific carboxylic acid functionalities, the inventors have created a class of catalysts that exhibit exceptional solubility in aqueous phases while maintaining high oxidative activity. For R&D directors and process chemists seeking reliable pharma intermediates supplier partnerships, understanding the nuances of this patented route is essential for developing next-generation oxidation processes that are both economically and environmentally sustainable.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional TEMPO-mediated oxidations, while widely regarded as a gold standard for converting alcohols to aldehydes or ketones, are plagued by inherent physicochemical limitations that hinder their broader application in large-scale manufacturing. The primary issue lies in the steric hindrance and lipophilicity of the standard TEMPO molecule, which results in poor solubility in water phases. This limitation necessitates the use of higher catalyst loadings, typically ranging from 1% to 10% relative to the substrate, to achieve acceptable reaction rates, thereby inflating raw material costs. Furthermore, the separation of TEMPO from the reaction mixture post-synthesis is notoriously difficult, often requiring multiple washing steps that generate substantial volumes of nitrogen-containing wastewater, posing significant environmental disposal challenges. Additionally, standard TEMPO is characterized by an unpleasant odor and thermal instability, mandating refrigerated storage conditions that complicate logistics and increase supply chain overheads for procurement managers.
The Novel Approach
In stark contrast, the methodology outlined in CN112851571A introduces a structurally modified TEMPO derivative that effectively circumvents these historical bottlenecks through rational molecular design. The novel approach involves the introduction of a carboxylic acid or ester moiety at the 4-position of the piperidine ring, fundamentally altering the solubility profile of the catalyst without compromising its radical stability. This structural modification allows the catalyst to function efficiently in biphasic systems or purely aqueous environments, drastically reducing the required catalyst loading to as low as 0.01% to 1% by weight. The presence of the acidic group also facilitates a streamlined work-up procedure; post-reaction, the catalyst can be easily separated from the organic product by a simple wash with weak alkaline water, eliminating the need for complex chromatographic purification. This not only enhances product quality but also aligns perfectly with the goals of cost reduction in fine chemical manufacturing by minimizing waste and solvent consumption.
Mechanistic Insights into Horner-Wadsworth-Emmons Olefination and Hydrolysis
The synthetic strategy employed to generate these advanced catalysts is a masterclass in efficient organic transformation, relying on a robust three-step sequence that begins with the oxidation of a commercially available ketone. The process initiates with the oxidation of 2,2,6,6-tetramethyl-4-piperidone using a sodium tungstate-hydrogen peroxide system, a green oxidant combination that cleanly generates the key 4-carbonyl-TEMPO intermediate with high fidelity. This intermediate then undergoes a Horner-Wadsworth-Emmons (HWE) olefination reaction with various phosphonate esters, such as triethyl phosphonoacetate, in the presence of strong bases like sodium hydride. This carbon-carbon bond-forming step is critical as it installs the requisite side chain that will eventually become the solubilizing carboxylic acid group. The reaction conditions are mild, typically proceeding between 0°C and 50°C, which preserves the integrity of the sensitive nitroxide radical throughout the synthesis.
Following the olefination, the final step involves the hydrolysis of the resulting ester to yield the free acid derivative, a transformation that is crucial for unlocking the catalyst's water-soluble properties. The hydrolysis is conducted under basic conditions using inorganic bases such as sodium hydroxide or potassium carbonate, followed by careful pH adjustment to precipitate the pure product. This purification strategy is particularly elegant because it avoids the use of hazardous organic solvents for crystallization, instead utilizing mixtures of petroleum ether and ethyl acetate to induce precipitation of the off-white solid. The result is a highly pure compound, with patent examples demonstrating purities exceeding 98%, which is vital for ensuring consistent catalytic performance in downstream applications. The mechanistic robustness of this route ensures that impurities are minimized, providing R&D teams with a reliable source of high-purity OLED material or pharmaceutical intermediates.
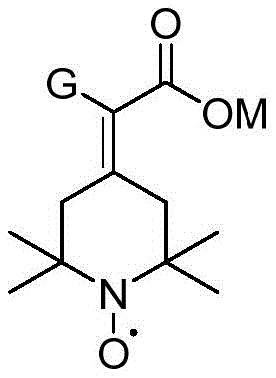
How to Synthesize 2-(2,2,6,6-tetramethylpiperidine nitroxide radical-4-subunit) Acetic Acid Derivative Efficiently
Executing the synthesis of these specialized catalysts requires precise control over reaction parameters to maximize yield and purity, particularly during the sensitive oxidation and hydrolysis stages. The patent provides a comprehensive blueprint that guides chemists through the stoichiometry and thermal profiles necessary for success, emphasizing the importance of reagent quality and temperature management. By adhering to the specified molar ratios—such as using 1.5 to 6 equivalents of hydrogen peroxide relative to the piperidone substrate—operators can ensure complete conversion while minimizing side reactions. The following guide summarizes the critical operational phases derived directly from the intellectual property, serving as a foundational reference for process development teams aiming to replicate or scale this technology.
- Oxidize commercial 2,2,6,6-tetramethyl-4-piperidone using sodium tungstate and hydrogen peroxide to form 4-carbonyl-TEMPO.
- React the 4-carbonyl intermediate with a phosphonylation reagent (e.g., triethyl phosphonoacetate) in the presence of a base like NaH to form the olefinic ester.
- Hydrolyze the ester intermediate using an inorganic base (e.g., NaOH) followed by acidification and recrystallization to isolate the pure acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel TEMPO derivative synthesis offers profound advantages that resonate deeply with procurement managers and supply chain heads focused on efficiency and risk mitigation. The primary value driver is the drastic simplification of the supply chain for raw materials; the process relies on 2,2,6,6-tetramethyl-4-piperidone, a commodity chemical that is cheap and easy to obtain in bulk quantities globally. This contrasts sharply with the sourcing of pre-functionalized, expensive catalysts that often suffer from long lead times and supply volatility. Furthermore, the elimination of transition metal catalysts, which are frequently required in alternative oxidation protocols, removes the need for costly and time-consuming heavy metal scavenging steps, thereby streamlining the production workflow and reducing overall processing time.
- Cost Reduction in Manufacturing: The economic implications of this technology are substantial, primarily driven by the enhanced catalytic efficiency and simplified downstream processing. Because the derivative exhibits superior activity, the dosage required is minimal, leading to direct savings on catalyst expenditure per batch. Moreover, the ability to separate the catalyst via simple aqueous extraction rather than complex distillation or chromatography significantly lowers energy consumption and solvent usage. These operational efficiencies translate into a lower cost of goods sold (COGS), allowing manufacturers to maintain competitive pricing margins while adhering to strict budgetary constraints without compromising on quality.
- Enhanced Supply Chain Reliability: Stability is a critical factor in supply chain management, and this new derivative excels where traditional TEMPO fails. The patent explicitly notes that the compound is stable, odorless, and can be stored at room temperature, eliminating the need for energy-intensive cold chain logistics. This characteristic ensures that inventory can be held safely for extended periods without degradation, providing a buffer against market fluctuations and ensuring continuous availability for production lines. For supply chain heads, this means reduced risk of spoilage and greater flexibility in inventory planning, securing the continuity of operations for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions intensify, the environmental profile of this synthesis offers a distinct competitive advantage. The process generates minimal waste, primarily salt and water, and avoids the release of toxic volatile organic compounds associated with other oxidation methods. The ease of catalyst recovery means less nitrogenous waste enters the wastewater stream, simplifying effluent treatment and ensuring compliance with stringent environmental regulations. This green chemistry approach not only future-proofs the manufacturing process against tightening laws but also enhances the corporate sustainability profile, a key metric for modern stakeholders.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial potential of this innovation, we have compiled a set of frequently asked questions based on the specific data points and claims within the patent documentation. These inquiries address common concerns regarding scalability, purity specifications, and comparative performance against legacy systems. Understanding these details is crucial for technical decision-makers who are evaluating the feasibility of integrating this new catalyst system into their existing manufacturing frameworks for complex polymer additives or agrochemical intermediates.
Q: How does this new TEMPO derivative improve upon traditional TEMPO catalysts?
A: Unlike traditional TEMPO, which suffers from poor water solubility and unpleasant odor requiring refrigerated storage, this derivative features a carboxylic acid side chain that significantly enhances aqueous solubility. This allows for easier separation via simple alkaline washing, reduces nitrogen-containing wastewater, and enables stable room-temperature storage.
Q: What are the typical yields for the synthesis of these derivatives?
A: According to patent data, the overall yields vary by specific substituent but are commercially viable. For instance, the propionic acid derivative (Compound B) achieves a total yield of 73% with 98.1% purity, while the phenylacetic acid derivative reaches 76.7% yield, demonstrating robust process efficiency.
Q: Can these derivatives be used for large-scale industrial oxidation?
A: Yes, the process utilizes cheap and readily available raw materials like 2,2,6,6-tetramethyl-4-piperidone and avoids expensive transition metals. The catalytic activity is high even at low loadings (0.01-1%), making it highly suitable for cost reduction in fine chemical manufacturing and scalable production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2,2,6,6-tetramethylpiperidine nitroxide radical-4-subunit) Acetic Acid Derivative Supplier
The technological breakthroughs encapsulated in CN112851571A represent more than just a laboratory curiosity; they are a blueprint for the future of sustainable industrial oxidation. At NINGBO INNO PHARMCHEM, we recognize the transformative potential of these water-soluble TEMPO derivatives and are fully equipped to bring this chemistry from the bench to the plant floor. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purities reported in the patent are faithfully reproduced at an industrial level. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch meets the exacting standards required by the global pharmaceutical and fine chemical industries.
We invite forward-thinking partners to collaborate with us to unlock the full value of this proprietary technology. Whether you are looking to optimize an existing oxidation line or develop a new green synthesis route, our technical procurement team is ready to assist. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can drive efficiency and profitability in your operations.
