Advanced Oxibendazole Manufacturing: Cost-Effective m-Dichlorobenzene Route for Global Supply Chains
The pharmaceutical and veterinary industries are constantly seeking more efficient, cost-effective, and environmentally sustainable pathways for active pharmaceutical ingredient (API) production. A significant breakthrough in this domain is detailed in patent CN103601685A, which introduces a novel synthesis route for Oxibendazole, a broad-spectrum benzimidazole anthelmintic widely used in veterinary medicine. Unlike traditional methods that rely on expensive starting materials and generate hazardous waste, this new protocol utilizes m-dichlorobenzene as the foundational feedstock. This strategic shift not only lowers the raw material cost baseline but also streamlines the downstream purification processes. The patent outlines a comprehensive five-step sequence involving nitration, ammonification, condensation, reduction, and cyclization, each optimized for high yield and purity. For procurement managers and supply chain directors, this represents a pivotal opportunity to secure a reliable veterinary drug intermediate supplier capable of delivering high-quality intermediates with a significantly reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Oxibendazole has relied heavily on acetaminophen (paracetamol) as the starting material, a route that presents substantial economic and ecological drawbacks. As illustrated in the conventional pathway below, the process typically involves refluxing acetaminophen with n-propyl bromide in ethanol under potassium hydroxide catalysis. This reliance on n-propyl bromide is particularly problematic because it generates significant amounts of brominated wastewater, which is difficult and costly to treat due to its high toxicity and persistence in the environment. Furthermore, subsequent steps often employ harsh reducing agents such as tin(II) chloride or sodium sulfide. The use of tin chloride leads to heavy metal contamination requiring complex removal protocols, while sodium sulfide produces foul-smelling and toxic sulfur-containing byproducts. These factors collectively inflate the operational expenditure (OPEX) and complicate regulatory compliance for manufacturers.
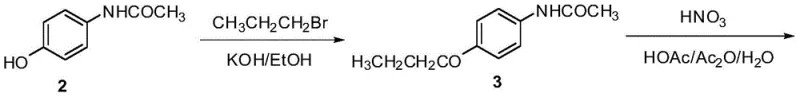
The Novel Approach
In stark contrast, the innovative methodology disclosed in the patent fundamentally restructures the synthetic logic by commencing with m-dichlorobenzene, a commodity chemical that is vastly more affordable than acetaminophen. The new route, depicted below, bypasses the need for alkyl halides entirely by employing n-propanol directly in the condensation step, thereby eliminating the source of brominated effluents. Additionally, the reduction phase is modernized through the adoption of clean technologies such as catalytic hydrogenation or hydrazine hydrate reduction, which avoid heavy metal sludge and sulfur pollution. The final cyclization utilizes O-methyl-isourea methyl-formiate or cyanamide methyl-formiate instead of sulfur-bearing agents, ensuring the final product is free from difficult-to-remove sulfur impurities. This holistic redesign results in a process that is not only economically superior but also aligns perfectly with modern green chemistry principles, offering a compelling value proposition for cost reduction in pharmaceutical manufacturing.
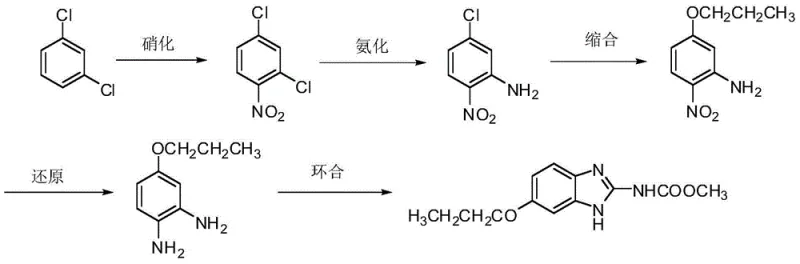
Mechanistic Insights into the Five-Step Synthetic Sequence
The success of this new route lies in the precise control of reaction mechanisms across five distinct transformations. The process initiates with the electrophilic aromatic nitration of m-dichlorobenzene using a mixture of concentrated sulfuric and nitric acids at controlled low temperatures (0-20°C). This step selectively introduces a nitro group to the 4-position relative to one chlorine atom, yielding 2,4-dichloronitrobenzene with high regioselectivity. Following isolation, the intermediate undergoes nucleophilic aromatic substitution (ammonification) under high pressure (0.8-10.0 MPa) and elevated temperatures (80-250°C) with liquid ammonia or ammonium hydroxide. This critical step displaces the chlorine atom ortho to the nitro group, facilitated by the electron-withdrawing nature of the nitro group, to form 2-nitro-5-chloroaniline. The subsequent condensation step involves the formation of an alkoxide from n-propanol and a base like sodium hydroxide, which then attacks the remaining chloro-substituted carbon to form the ether linkage, creating 2-nitro-5-propoxy-aniline.
The fourth stage involves the reduction of the nitro group to an amine, a transformation achieved either through catalytic hydrogenation using Pd/C or Raney Nickel, or via hydrazine hydrate reduction catalyzed by ferric chloride. Both methods are highly efficient, converting the nitro functionality into an amino group without affecting the ether linkage, yielding 4-propoxy-1,2-phenylenediamine. The final cyclization is a condensation reaction between the diamine and a cyclizing agent such as O-methyl-isourea methyl-formiate. This reaction constructs the benzimidazole ring system through the formation of two new carbon-nitrogen bonds, closing the heterocyclic ring to produce the final Oxibendazole molecule. The mechanistic elegance of this route ensures that impurities are minimized at every stage, resulting in a final product with exceptional purity profiles suitable for sensitive veterinary applications.
How to Synthesize Oxibendazole Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly during the high-pressure ammonification and the final cyclization steps. The patent provides detailed embodiments demonstrating that yields can consistently exceed 90% in the final steps when optimal stoichiometry and temperature controls are maintained. For R&D teams looking to replicate or scale this process, the key lies in selecting the appropriate reduction method—catalytic hydrogenation is preferred for large-scale continuous flow due to its cleanliness, while hydrazine reduction offers a viable batch alternative. The standardized synthesis steps outlined in the patent serve as a robust foundation for process development, ensuring that the transition from laboratory bench to pilot plant is seamless and reproducible.
- Nitration of m-dichlorobenzene using sulfuric and nitric acid at 0-20°C to form 2,4-dichloronitrobenzene.
- High-pressure ammonification with liquid ammonia or ammonium hydroxide to yield 2-nitro-5-chloroaniline.
- Condensation with n-propanol and alkali metal salts to form the propoxy ether linkage.
- Reduction of the nitro group via catalytic hydrogenation or hydrazine hydrate to form the diamine intermediate.
- Cyclization using O-methyl-isourea methyl-formiate to close the benzimidazole ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this m-dichlorobenzene-based route offers transformative benefits that extend far beyond simple chemistry. The most immediate impact is seen in the raw material cost structure; replacing acetaminophen with m-dichlorobenzene represents a fundamental shift to a lower-cost commodity feedstock, which stabilizes pricing against fluctuations in the paracetamol market. Moreover, the elimination of n-propyl bromide removes a volatile and regulated hazardous material from the supply chain, simplifying logistics and storage requirements. The absence of heavy metals and sulfur byproducts significantly reduces the burden on waste treatment facilities, leading to lower disposal costs and faster regulatory approvals for manufacturing sites. This streamlined workflow enhances overall supply chain resilience, making it easier to maintain consistent production schedules without the interruptions often caused by environmental compliance audits or hazardous waste bottlenecks.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the substitution of high-value starting materials with low-cost alternatives and the simplification of downstream processing. By avoiding the use of expensive alkylating agents like n-propyl bromide and eliminating the need for complex heavy metal scavenging resins or sulfur removal steps, the overall variable cost per kilogram of product is substantially decreased. The high yields reported in the patent embodiments further contribute to cost efficiency by maximizing the output from each batch of raw materials. Additionally, the use of recyclable solvents and catalysts in the hydrogenation step adds another layer of long-term savings, ensuring that the manufacturing process remains economically competitive even at large commercial scales.
- Enhanced Supply Chain Reliability: Supply chain continuity is critically improved by relying on m-dichlorobenzene, a bulk chemical with a stable and abundant global supply, rather than specialized intermediates that may face availability constraints. The robustness of the reaction conditions, which tolerate a range of temperatures and pressures without compromising yield, ensures that production can continue reliably even under varying operational circumstances. Furthermore, the reduction in hazardous waste generation minimizes the risk of production stoppages due to waste tank capacity limits or transportation delays for hazardous waste disposal. This reliability is essential for maintaining the steady flow of veterinary medicines to the market, preventing stockouts that could impact animal health sectors globally.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is uniquely positioned for rapid industrial expansion. The replacement of batch-intensive, pollution-heavy steps with cleaner catalytic processes facilitates easier scale-up from pilot plants to multi-ton reactors. The absence of brominated and sulfur-containing effluents simplifies the environmental impact assessment (EIA) process for new manufacturing facilities, accelerating the time-to-market for new production lines. This alignment with green chemistry standards not only future-proofs the manufacturing asset against tightening environmental regulations but also enhances the brand reputation of the supplier as a responsible and sustainable partner in the global pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Oxibendazole synthesis route. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on feasibility, safety, and quality control. Understanding these details is crucial for technical teams evaluating the potential for technology transfer or licensing agreements.
Q: What are the primary cost advantages of the m-dichlorobenzene route over the acetaminophen route?
A: The m-dichlorobenzene route replaces expensive acetaminophen with a significantly cheaper commodity chemical. Furthermore, it eliminates the need for costly n-propyl bromide and expensive heavy metal reducing agents like tin chloride, drastically lowering raw material and waste treatment costs.
Q: How does this new method address environmental compliance issues?
A: This method avoids the generation of brominated wastewater associated with n-propyl bromide and eliminates sulfur-containing byproducts and heavy metal sludge from traditional reduction methods, resulting in a much cleaner production profile suitable for strict environmental regulations.
Q: Is the catalytic hydrogenation step scalable for industrial production?
A: Yes, the patent details robust conditions for catalytic hydrogenation using Pd/C or Raney Ni at moderate pressures (0.1-5 MPa) and temperatures, which are standard parameters for large-scale industrial reactors, ensuring high scalability and safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxibendazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting advanced synthetic routes like the one described in CN103601685A to maintain competitiveness in the global veterinary pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is managed with precision and expertise. We are committed to delivering high-purity Oxibendazole intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement clean, cost-effective chemistries allows us to offer products that not only meet performance criteria but also align with the sustainability goals of our international partners.
We invite procurement leaders and R&D directors to engage with us to explore how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic impact of switching to this m-dichlorobenzene-based process. We encourage you to contact our technical procurement team today to obtain specific COA data for our current batches and to discuss route feasibility assessments tailored to your production volumes. Let us collaborate to build a more efficient, sustainable, and reliable supply chain for essential veterinary medicines.
