Advanced Manufacturing of 2-Chloro-4-amino-6,7-dimethoxy Quinazoline for Global Pharma Supply Chains
Advanced Manufacturing of 2-Chloro-4-amino-6,7-dimethoxy Quinazoline for Global Pharma Supply Chains
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable pathways for the production of critical heterocyclic intermediates. Patent CN101353328B introduces a transformative methodology for the preparation of 2-chloro-4-amino-6,7-dimethoxy quinazoline, a pivotal building block for alpha-adrenergic antagonists such as Prazosin and Doxazosin. This technical insight report analyzes the proprietary synthesis route disclosed in the patent, highlighting its strategic advantages over legacy manufacturing processes. By replacing hazardous oxidants like potassium permanganate with hydrogen peroxide and eliminating the need for high-pressure hydrogenation equipment, this process offers a compelling value proposition for R&D directors focused on purity profiles and supply chain leaders concerned with operational continuity. The following analysis dissects the chemical innovations that enable higher yields exceeding 48%, compared to the historical average of roughly 30%, while simultaneously addressing the critical imperatives of cost reduction and environmental compliance in modern fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of quinazoline derivatives has been plagued by significant technical and economic bottlenecks that hinder efficient commercial scale-up. Traditional routes often commence with Vanillin, necessitating a methylation step that employs dimethyl sulfate, a reagent known for its extreme toxicity and severe environmental impact, posing substantial risks to operator health and requiring complex waste neutralization protocols. Furthermore, the oxidation of aldehyde intermediates in these legacy processes typically relies on potassium permanganate, which generates massive quantities of manganese dioxide sludge, creating a difficult-to-manage solid waste stream that inflates disposal costs. Another critical limitation is the reliance on catalytic hydrogenation for the reduction of nitro groups, which mandates the use of expensive heavy metal catalysts and high-pressure autoclaves, thereby increasing both capital expenditure and safety risks. Additionally, the chlorination steps in conventional methods frequently utilize noxious solvents such as N,N-dimethylaniline or high-boiling polar aprotic solvents like DMF, which are difficult to remove completely and can lead to persistent impurities in the final API intermediate, complicating downstream purification.
The Novel Approach
The innovative pathway detailed in patent CN101353328B fundamentally re-engineers the synthetic sequence to bypass these historical inefficiencies through a series of strategic chemical substitutions. By initiating the synthesis with commercially available 3,4-dimethoxy benzaldehyde, the process entirely eliminates the hazardous methylation step, immediately reducing the toxicological burden on the production facility. The oxidation phase is revolutionized by the use of hydrogen peroxide in an alkaline medium, a green oxidant that produces water as the only byproduct, effectively eradicating the heavy metal sludge associated with permanganate oxidation. The reduction strategy shifts from high-pressure catalytic hydrogenation to a chemoselective reduction using iron powder and hydrochloric acid under atmospheric conditions, which not only removes the need for specialized pressure vessels but also utilizes inexpensive, readily available reagents. Moreover, the chlorination and cyclization stages are optimized to proceed without the aid of toxic organic solvents like DMF or N,N-dimethylaniline, relying instead on direct reaction conditions that simplify work-up procedures and significantly enhance the purity profile of the resulting 2-chloro-4-amino-6,7-dimethoxy quinazoline.
Mechanistic Insights into Green Oxidation and Cyclization
The core of this technological advancement lies in the meticulous control of reaction conditions to maximize selectivity while minimizing side reactions. The initial oxidation of 3,4-dimethoxy benzaldehyde to 3,4-dimethoxybenzoic acid utilizes a hydrogen peroxide and sodium hydroxide system at temperatures ranging from 20°C to 60°C. This Dakin-like oxidation mechanism ensures the complete conversion of the aldehyde functionality without over-oxidizing the electron-rich aromatic ring, a common issue with harsher oxidants. Following nitration with concentrated nitric acid in chloroform, the subsequent reduction step employs iron powder in the presence of hydrochloric acid and sodium chloride. This heterogeneous reduction proceeds via electron transfer from the metal surface to the nitro group, generating the corresponding aniline derivative with high fidelity. The absence of noble metal catalysts prevents potential contamination of the product stream with trace metals, a critical quality attribute for pharmaceutical intermediates intended for human consumption.
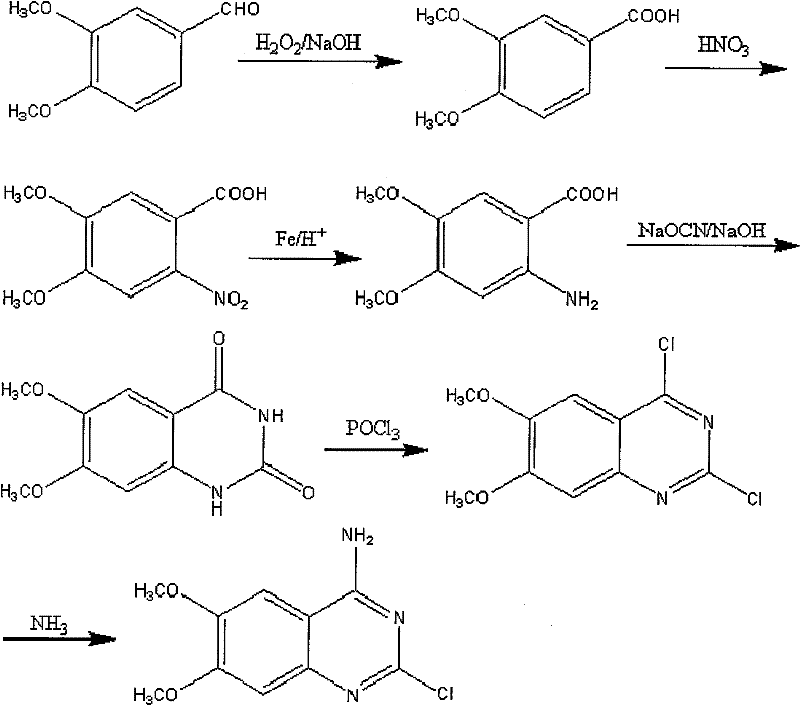
The cyclization of the amino acid intermediate to form the quinazoline ring system is achieved using sodium cyanate under controlled pH conditions, followed by dehydration to form the dione structure. This is subsequently converted to the dichloro derivative using phosphorus oxychloride at elevated temperatures between 80°C and 120°C. The mechanistic advantage here is the direct activation of the carbonyl groups by POCl3 without the need for solvating agents that could interfere with the reaction kinetics or introduce difficult-to-remove impurities. Finally, the selective nucleophilic substitution of the 4-position chlorine with ammonia yields the target 2-chloro-4-amino product. The regioselectivity is governed by the electronic differences between the 2 and 4 positions of the quinazoline ring, ensuring that the 2-chloro group remains intact for further functionalization in downstream API synthesis. This precise control over the reaction trajectory is what allows the total yield to surpass 48%, a substantial improvement over the literature benchmarks of 26% to 30%.
How to Synthesize 2-Chloro-4-amino-6,7-dimethoxy Quinazoline Efficiently
Implementing this synthesis route requires strict adherence to the stoichiometric ratios and temperature profiles defined in the patent to ensure reproducibility and safety. The process begins with the dissolution of the aldehyde starting material in a basic solution, followed by the controlled addition of hydrogen peroxide to manage the exothermic nature of the oxidation. Subsequent steps involve careful pH adjustments during the reduction and cyclization phases to precipitate intermediates effectively, minimizing product loss in the mother liquor. The chlorination step demands rigorous temperature control to prevent the formation of poly-chlorinated byproducts, while the final ammoniation requires sufficient reaction time to ensure complete conversion of the 4-chloro group. For a comprehensive understanding of the specific operational parameters, including exact molar ratios and stirring speeds required for GMP-compliant manufacturing, please refer to the standardized technical protocol outlined below.
- Oxidize 3,4-dimethoxy benzaldehyde to 3,4-dimethoxybenzoic acid using hydrogen peroxide and sodium hydroxide at 20°C to 60°C.
- Perform nitration with nitric acid in chloroform, followed by reduction of the nitro group using iron powder and hydrochloric acid.
- Cyclize the amino acid intermediate with sodium cyanate, followed by chlorination with phosphorus oxychloride and final ammoniation.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented synthesis route offers profound strategic benefits that extend beyond simple yield improvements. The elimination of expensive and regulated reagents such as potassium permanganate and dimethyl sulfate directly translates to a reduction in raw material costs and a simplification of the purchasing process, as these substances often require special handling permits and storage facilities. Furthermore, the removal of high-pressure hydrogenation steps significantly lowers the barrier to entry for contract manufacturing organizations, as it negates the need for specialized autoclave infrastructure, thereby expanding the pool of qualified suppliers capable of producing this intermediate at scale. The simplified solvent profile, which avoids high-boiling toxic solvents like DMF in the refining process, drastically reduces the energy consumption associated with solvent recovery and distillation, leading to substantial operational cost savings. Additionally, the generation of less hazardous waste streams aligns with increasingly stringent global environmental regulations, mitigating the risk of production shutdowns due to compliance issues and ensuring a more reliable supply continuity for downstream pharmaceutical customers.
- Cost Reduction in Manufacturing: The substitution of costly oxidants and catalysts with commodity chemicals like hydrogen peroxide and iron powder creates a leaner cost structure for the manufacturing process. By avoiding the use of precious metal catalysts, the process eliminates the financial burden associated with catalyst recovery and the potential loss of valuable metals in the waste stream. The simplified work-up procedures, which do not require extensive chromatography or complex solvent exchanges, further reduce labor and utility costs per kilogram of product. Consequently, the overall cost of goods sold (COGS) is significantly optimized, allowing for more competitive pricing in the global market for antihypertensive intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as 3,4-dimethoxy benzaldehyde ensures that the supply chain is resilient against raw material shortages that might affect more exotic precursors. The robustness of the iron powder reduction method means that production is not dependent on the availability of specialized hydrogenation services or the logistics of transporting compressed hydrogen gas. This decentralization of production capabilities allows for greater flexibility in sourcing and reduces the lead time for order fulfillment. Moreover, the stability of the intermediates formed during this process facilitates easier storage and transportation, minimizing the risk of degradation during transit and ensuring that the material arrives at the customer's facility with the specified purity intact.
- Scalability and Environmental Compliance: The inherent safety of the reaction conditions, particularly the avoidance of high-pressure and high-temperature extremes in the reduction step, makes this process highly amenable to scale-up from pilot plant to multi-ton commercial production. The reduction in "three wastes" (wastewater, waste gas, and solid waste) pollution simplifies the environmental impact assessment and permitting process for new manufacturing lines. By utilizing water and benign salts for quenching and precipitation, the process minimizes the load on wastewater treatment facilities, reducing the environmental footprint of the operation. This alignment with green chemistry principles not only protects the health of operators but also future-proofs the manufacturing asset against tightening environmental legislation, ensuring long-term viability and sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical implications of adopting this route. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing supply chains or for R&D groups looking to optimize their own synthetic pathways for quinazoline derivatives.
Q: How does this synthesis route improve upon traditional methods using Vanillin?
A: Traditional routes starting from Vanillin often require toxic methylating agents like dimethyl sulfate and expensive oxidants like potassium permanganate. This patented method starts directly from 3,4-dimethoxy benzaldehyde, utilizing hydrogen peroxide for oxidation, which significantly reduces hazardous waste and eliminates the need for methylation steps.
Q: What are the safety advantages regarding solvents in the chlorination step?
A: Conventional chlorination processes frequently employ highly toxic solvents such as N,N-dimethylaniline or high-boiling solvents like DMF. This optimized process utilizes phosphorus oxychloride directly without these noxious organic solvents, drastically improving operator safety and simplifying solvent recovery and waste treatment.
Q: Does this method require high-pressure equipment for reduction?
A: No. Unlike catalytic hydrogenation methods that necessitate expensive autoclaves and heavy metal catalysts, this protocol employs iron powder and hydrochloric acid for the reduction of the nitro group. This atmospheric pressure process lowers capital expenditure and simplifies reactor requirements for scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-4-amino-6,7-dimethoxy Quinazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green synthesis route are fully realized in large-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of 2-chloro-4-amino-6,7-dimethoxy quinazoline meets the exacting standards required for pharmaceutical applications. Our commitment to process safety and environmental stewardship mirrors the principles of the patented method, making us an ideal partner for companies seeking sustainable and reliable sources of critical heterocyclic intermediates.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages tailored to your volume needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on empirical evidence and our proven track record in delivering high-purity pharmaceutical intermediates to the global market.
