Advanced Synthesis of Chiral Anthracene Derivatives for High-Performance Circularly Polarized Luminescence
Introduction to Next-Generation Chiral Luminescent Materials
The rapid evolution of the organic light-emitting diode (OLED) industry has created an urgent demand for advanced materials capable of emitting circularly polarized light (CPL), which eliminates the need for external polarizers and significantly enhances device efficiency. Patent CN113105472A introduces a groundbreaking class of chiral fluorescent molecules based on an anthracene core, specifically designed to address these technological needs. These derivatives, characterized by their rigid polycyclic aromatic hydrocarbon skeleton and strategically positioned chiral groups, offer superior spatial geometric structures that facilitate photoinduced electroluminescence. As a leading entity in the fine chemical sector, we recognize the immense potential of these compounds to revolutionize the reliable OLED material supplier landscape by providing access to high-performance emitters with tunable emission wavelengths.
The core innovation lies in the structural versatility of the anthracene derivative, where substituents at the 9 and 10 positions can be independently selected from phenyl, substituted phenyl, dianilino, or acridino groups, while chiral moieties are introduced at the 2, 3, 6, and 7 positions. This specific architectural design ensures that the terminal substituents do not adversely affect the geometric and electronic properties of the derivative, thereby maintaining high fluorescence quantum yields. The following general structure illustrates the fundamental scaffold of these high-value intermediates:
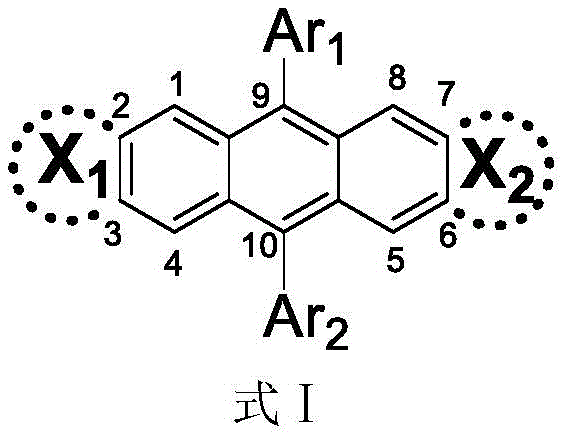
Furthermore, the patent highlights that these materials exhibit excellent stability and ease of derivatization, making them ideal candidates for large-scale commercial scale-up of complex electronic chemicals. By leveraging the intrinsic properties of the anthracene backbone, manufacturers can achieve precise control over the emission color and polarization, which is critical for applications in 3D displays and advanced optical storage systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral organic luminescent materials has been plagued by significant challenges, including multi-step procedures that suffer from low overall yields and the frequent use of expensive, scarce chiral catalysts. Conventional routes often require harsh reaction conditions that can lead to racemization, thereby compromising the optical purity of the final product and diminishing its circularly polarized luminescence (CPL) performance. Additionally, the purification of intermediates in standard anthracene functionalization processes is frequently complicated by the formation of regioisomers, necessitating energy-intensive chromatographic separations that are impractical for industrial production. These inefficiencies result in prohibitive costs and extended lead times, creating a bottleneck for the cost reduction in electronic chemical manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN113105472A presents a streamlined, efficient synthetic pathway that utilizes inexpensive and commercially available starting materials such as 4,5-difluorophthalic anhydride and 1,2-difluorobenzene. The novel approach employs a robust sequence of Friedel-Crafts acylation followed by acid-catalyzed cyclization to construct the anthraquinone core, ensuring high conversion rates and minimal byproduct formation. Crucially, the introduction of chirality is achieved through nucleophilic substitution with stable chiral diols or diamines, a process that preserves the stereochemical integrity of the molecule throughout the synthesis. This strategic design not only simplifies the operational complexity but also drastically improves the scalability of the process, allowing for the production of high-purity intermediates suitable for mass market adoption.
Mechanistic Insights into the Multi-Step Synthesis Strategy
The synthetic route detailed in the patent is a masterclass in logical retrosynthetic analysis, breaking down the complex target molecule into manageable transformations. The process initiates with a Friedel-Crafts acylation where aluminum trichloride acts as a Lewis acid to facilitate the reaction between the anhydride and difluorobenzene, forming a benzoyl benzoic acid derivative. This is followed by a cyclization step using hot concentrated sulfuric acid, which effectively closes the central ring to form the anthraquinone skeleton. The subsequent etherification or amination step is particularly noteworthy, as it attaches the chiral units (such as octahydrobinaphthol) to the electron-deficient anthraquinone core via nucleophilic aromatic substitution, a reaction driven by the activating effect of the carbonyl groups.
Following the installation of the chiral periphery, the carbonyl groups at the 9 and 10 positions are reduced to methylene groups using hydroiodic acid, a transformation that restores the aromaticity of the central ring while maintaining the chiral environment. The final stages involve regioselective bromination at the 9 and 10 positions, followed by a palladium-catalyzed cross-coupling reaction (either Suzuki or Buchwald-Hartwig) to attach the bulky aryl amine or carbazole groups. This final coupling step is critical for tuning the electronic properties and enhancing the solid-state fluorescence of the material. The comprehensive reaction sequence is depicted below, showcasing the progression from simple precursors to the sophisticated chiral emitter:
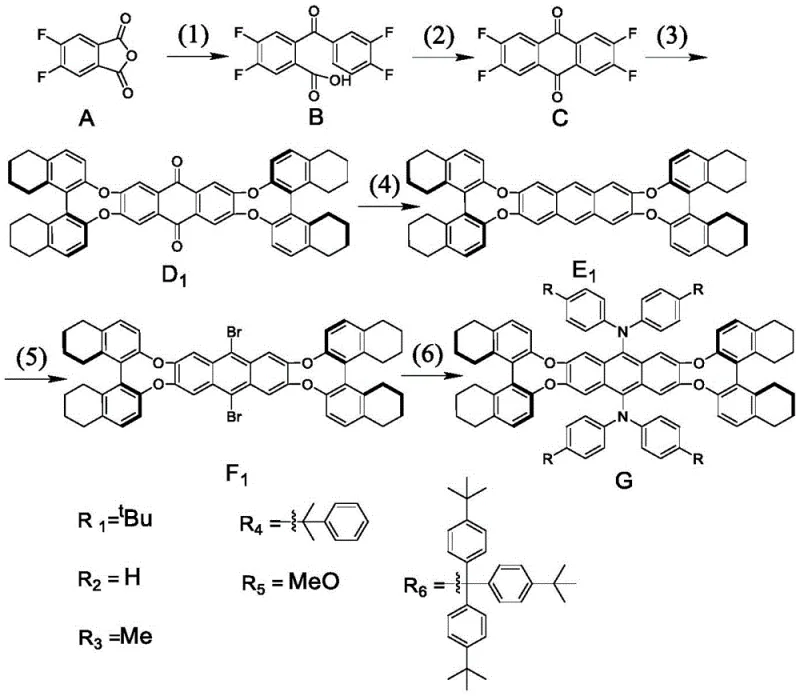
From a quality control perspective, this mechanism offers distinct advantages in impurity management. The use of specific reagents like liquid bromine allows for precise control over the degree of halogenation, minimizing the risk of over-bromination which could complicate downstream purification. Moreover, the final palladium-catalyzed step is conducted under inert atmosphere with rigorous exclusion of oxygen and moisture, ensuring that the sensitive amine functionalities remain intact and that the final product meets the stringent purity specifications required for electronic grade materials.
How to Synthesize Chiral Anthracene Derivatives Efficiently
Implementing this synthesis in a production environment requires careful attention to reaction parameters such as temperature control and stoichiometry to maximize yield and safety. The patent provides explicit guidance on molar ratios, such as using a 1:5 ratio of anhydride to difluorobenzene to drive the initial acylation to completion, and maintaining specific temperatures (e.g., 100°C for cyclization, 120°C for reduction) to ensure optimal kinetics. For R&D teams looking to replicate or optimize this process, the detailed experimental procedures offer a robust foundation for method development.
- Perform Friedel-Crafts acylation of 4,5-difluorophthalic anhydride with 1,2-difluorobenzene using AlCl3 to form the intermediate ketone.
- Cyclize the intermediate using concentrated sulfuric acid at elevated temperatures to form the anthraquinone core.
- React the anthraquinone with a chiral diol or diamine (e.g., octahydrobinaphthol) in DMF to introduce chirality at the 2,3,6,7 positions.
- Reduce the quinone carbonyls to methylene groups using hydroiodic acid and acetic acid.
- Brominate the 9,10 positions using liquid bromine in chloroform.
- Conclude with a palladium-catalyzed cross-coupling reaction with aryl amines or boronic acids to attach the final emissive groups.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible benefits that extend beyond mere technical performance. The reliance on commodity chemicals like fluorobenzenes and sulfuric acid significantly de-risks the supply chain, as these materials are produced globally in massive quantities, ensuring consistent availability and price stability. Unlike processes dependent on rare earth metals or exotic ligands, this route utilizes standard transition metal catalysts like palladium acetate in the final step, which can be recovered and recycled, further contributing to cost reduction in electronic chemical manufacturing.
- Cost Reduction in Manufacturing: The elimination of complex chiral catalysts in favor of chiral pool starting materials (like binaphthol derivatives) removes a major cost driver from the bill of materials. The high yields reported in the patent examples, such as the 93.8% yield in the final coupling step, indicate a highly efficient process that minimizes waste and maximizes throughput. By avoiding low-yielding steps and extensive purification protocols, manufacturers can achieve substantial cost savings per kilogram of finished product, making high-end CPL materials economically viable for broader applications.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for flexible production scheduling. Since the chiral information is introduced early in the sequence via stable intermediates, the risk of batch-to-batch variability due to racemization is virtually eliminated. This consistency is crucial for maintaining long-term supply contracts with display manufacturers who require absolute reproducibility. Furthermore, the use of common solvents like toluene, DMF, and chloroform simplifies solvent recovery and waste management, reducing the logistical burden associated with hazardous material handling.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-kilogram pilot runs without requiring specialized high-pressure equipment. The workup procedures primarily involve standard extraction and recrystallization techniques, which are easily adapted to large-scale reactors. From an environmental standpoint, the atom economy of the key bond-forming steps is favorable, and the ability to recycle solvents and catalysts aligns with modern green chemistry principles, facilitating easier regulatory approval and reducing the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these chiral anthracene derivatives. These insights are derived directly from the patent specifications and are intended to clarify the feasibility of integrating this technology into existing supply chains.
Q: What is the primary advantage of this chiral anthracene synthesis?
A: The primary advantage is the use of cheap, readily available raw materials and a simple, high-yield synthetic route that preserves the chirality of the starting materials without racemization, resulting in stable CPL materials.
Q: Can these derivatives be used for commercial OLED displays?
A: Yes, the patent explicitly states these derivatives possess fluorescence and circularly polarized light emission properties, making them highly suitable for next-generation chiral optoelectronic materials and OLED applications.
Q: How is the chiral configuration controlled during synthesis?
A: The chiral configuration (R-type or S-type) is directly inherited from the chiral starting materials, such as octahydrobinaphthol or dimethylcyclohexanediamine, ensuring the final product maintains high optical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Anthracene Derivative Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from patent literature to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move from benchtop discovery to full-scale manufacturing without interruption. We are committed to delivering materials that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify chirality, purity, and optical properties.
We invite you to collaborate with us to unlock the full potential of these advanced OLED materials. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can accelerate your time-to-market and enhance your competitive edge in the global display industry.
