Industrial Scale Production of High-Purity (-)-Huperzine A via Advanced Chiral Resolution
Introduction to Advanced Chiral Resolution for Neurological APIs
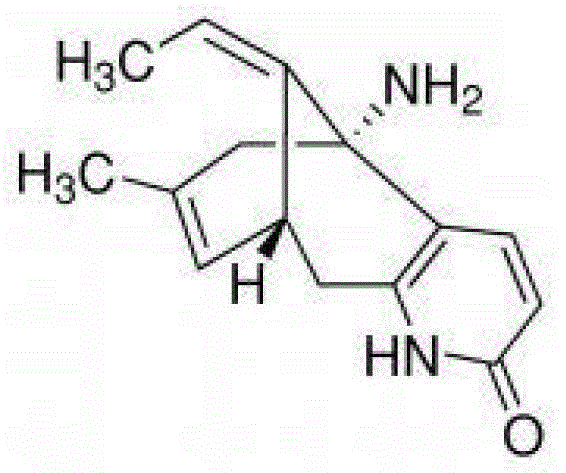
The escalating global demand for effective treatments against Alzheimer's disease and cognitive decline has placed (-)-Huperzine A at the forefront of pharmaceutical research. As a potent and reversible acetylcholinesterase inhibitor, the therapeutic efficacy of this alkaloid is strictly dependent on its stereochemical configuration, necessitating production methods that guarantee exceptional optical purity. Patent CN103224467A introduces a transformative preparation method that bypasses the traditional bottlenecks of asymmetric synthesis. By leveraging a sophisticated chiral acid resolution strategy, this technology enables the conversion of readily available racemic mixtures into highly purified (-)-Huperzine A. The process is characterized by its operational simplicity and robust scalability, achieving both chemical and optical purity levels exceeding 99.5%. This breakthrough represents a significant paradigm shift for manufacturers seeking a reliable API intermediate supplier capable of delivering consistent quality without the prohibitive costs associated with earlier synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
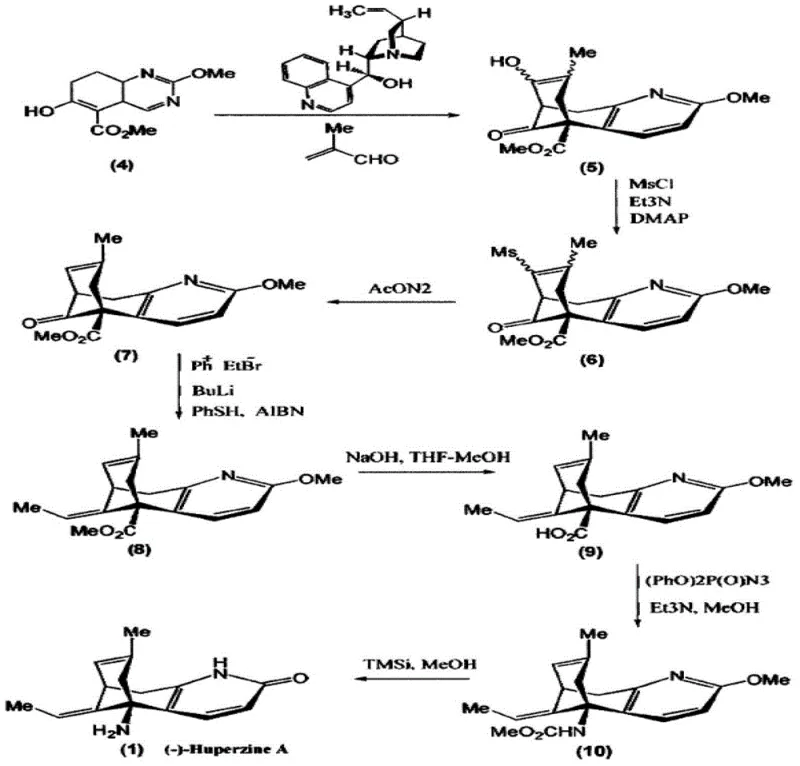
Historically, the industrial production of (-)-Huperzine A has been hindered by the inherent complexities of constructing its rigid bridged ring system with precise stereocontrol. Prior art strategies, such as those detailed in patent WO2009120774, rely on asymmetric Trost reactions which require expensive palladium catalysts and specialized chiral ligands. These multi-step sequences often suffer from cumulative yield losses, with total recoveries sometimes dropping as low as 6.8%, driving production costs to approximately 400 yuan/gram. Furthermore, alternative resolution methods described in patents like CN101130520 utilize resolving agents that result in diastereomeric salts with poor crystallization properties, leading to recovery rates as low as 16% and final costs nearing 600 yuan/gram. The reliance on column chromatography for purification in these legacy processes further exacerbates solvent consumption and processing time, rendering them economically unviable for large-scale commercial manufacturing.
The Novel Approach
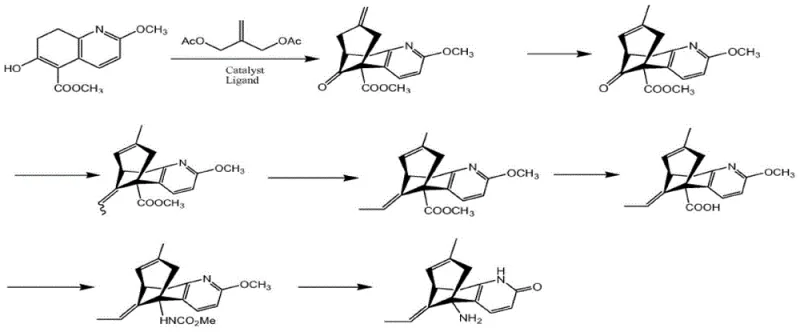
In stark contrast to these cumbersome methodologies, the novel approach disclosed in CN103224467A streamlines the production workflow into a highly efficient two-stage resolution process. Instead of attempting to build chirality from scratch through difficult catalytic cycles, this method capitalizes on the thermodynamic stability differences between diastereomeric salts formed from racemic Huperzine A and specific chiral acids. By selecting D-dibenzoyl tartaric acid as the resolving agent, the process exploits rapid precipitation kinetics where the desired (-)-isomer salt forms within minutes, while the unwanted (+)-isomer remains in solution. This fundamental shift eliminates the need for expensive transition metal catalysts and tedious chromatographic separations. The result is a drastic simplification of the supply chain, where the starting material cost is reduced to roughly 50 yuan/gram for the racemate, and the overall production cost drops to approximately 150 yuan/gram, offering a compelling value proposition for cost reduction in pharmaceutical manufacturing.
Mechanistic Insights into Diastereomeric Salt Crystallization
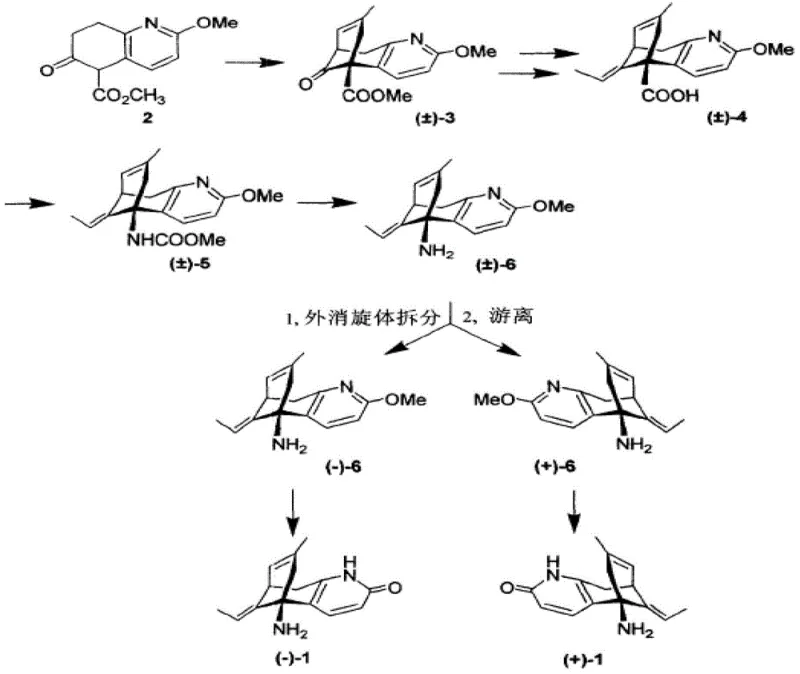
The core of this technological advancement lies in the precise manipulation of solubility equilibria during the salt formation stage. When racemic (+/-)-Huperzine A interacts with a chiral acid like D-dibenzoyl tartaric acid in a polar solvent system, two distinct diastereomeric salts are generated: the (-)-Huperzine A-(D)-dibenzoyl tartrate and the (+)-Huperzine A-(D)-dibenzoyl tartrate. The patent data reveals a critical kinetic disparity; the (-)-isomer salt precipitates almost immediately (within 1 minute) and completes formation within 1 hour, whereas the (+)-isomer salt requires significantly longer induction times (up to 3 hours) to nucleate. This kinetic window allows for the selective isolation of the desired enantiomer before the counter-enantiomer can contaminate the crystal lattice. Furthermore, the choice of solvent plays a pivotal role in enhancing this selectivity. Mixed solvent systems, particularly acetone/water or ethanol/water ratios ranging from 1:1 to 5:1, create an environment where the solubility product of the target salt is exceeded rapidly, driving high-yield crystallization while keeping impurities dissolved.
Impurity control is intrinsically built into this crystallization mechanism through the rigorous optimization of molar ratios and pH conditions. The patent specifies a molar ratio of racemic Huperzine A to chiral acid between 1:0.5 and 1:2, ensuring that there is sufficient resolving agent to drive the equilibrium towards salt formation without creating excessive mother liquor contamination. Following the isolation of the chiral salt, the free-basing step utilizes aqueous sodium hydroxide to adjust the pH to a narrow window of 9.0-9.3. This specific pH range is critical; it is high enough to fully deprotonate the amine group of the Huperzine A, causing it to precipitate as the free base, yet controlled enough to prevent the degradation of the sensitive lactam ring or the formation of emulsions that could trap impurities. The resulting solid is then subjected to recrystallization, which acts as a final polishing step to ensure the chemical purity exceeds 99.5%, effectively removing any trace amounts of the resolving acid or residual solvents.
How to Synthesize (-)-Huperzine A Efficiently
The synthesis of (-)-Huperzine A via this resolution pathway offers a robust framework for laboratory and pilot-scale operations, prioritizing safety and reproducibility. The process begins with the suspension of the racemic mixture in a carefully selected solvent system, followed by the controlled addition of the chiral resolving agent under mild thermal conditions. Detailed standard operating procedures regarding stirring rates, cooling profiles, and filtration techniques are essential to maximize the recovery rate, which the patent indicates can exceed 70%. For process chemists looking to implement this technology, understanding the nuances of the recrystallization solvent selection is paramount to achieving the target optical purity. The detailed standardized synthesis steps see the guide below.
- Suspend racemic (+/-)-Huperzine A in a mixed solvent system such as acetone/water or ethanol/water and add a chiral resolving agent like D-dibenzoyl tartaric acid.
- Stir the mixture to facilitate salt formation, filter the precipitate, and purify the resulting (-)-Huperzine A chiral acid salt through recrystallization.
- Treat the purified salt with aqueous sodium hydroxide to adjust pH to 9.0-9.3, inducing precipitation of the free base, followed by filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this resolution technology translates directly into enhanced margin protection and supply security. The most significant advantage is the substantial reduction in raw material and processing costs. By shifting from a multi-step asymmetric synthesis requiring precious metal catalysts to a simple salt formation and crystallization process, the cost of goods sold is dramatically lowered. The patent explicitly highlights a cost reduction from approximately 400-600 yuan/gram in prior art methods to roughly 150 yuan/gram with this new technique. This economic efficiency is achieved not only through cheaper starting materials but also by eliminating unit operations such as column chromatography, which are resource-intensive and difficult to scale. Consequently, this allows for cost reduction in pharmaceutical manufacturing that can be passed down the value chain or reinvested into further R&D initiatives.
- Cost Reduction in Manufacturing: The elimination of expensive chiral catalysts and the reduction of synthetic steps from over seven to essentially two major processing stages results in significant operational expenditure savings. The process utilizes common industrial solvents like acetone and ethanol, which are readily available and inexpensive compared to specialized reagents. Furthermore, the high recovery rate of greater than 70% ensures that raw material waste is minimized, directly improving the overall atom economy of the production line. This efficiency makes the commercial scale-up of complex alkaloids financially viable even in markets with tight pricing pressure.
- Enhanced Supply Chain Reliability: Reliance on complex asymmetric synthesis often creates single points of failure in the supply chain, particularly if specific chiral ligands or catalysts face shortages. In contrast, this resolution method uses D-dibenzoyl tartaric acid, a commodity chiral acid with a stable global supply. The robustness of the crystallization process also means that production is less susceptible to minor fluctuations in reaction conditions, ensuring consistent batch-to-batch quality. This reliability reduces lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demand spikes for Alzheimer's treatments.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this method offers a cleaner profile. The avoidance of heavy metal catalysts removes the need for costly and hazardous metal scavenging steps, simplifying waste stream management. The solvents used are primarily alcohols and ketones, which are easier to recover and recycle compared to halogenated solvents often used in older synthetic routes. The simplicity of the equipment required—primarily reactors, filters, and dryers—means that scaling from kilogram to tonnage production does not require exotic engineering solutions, facilitating a smoother technology transfer to commercial manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral resolution technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, ensuring accuracy for technical decision-makers. Understanding these specifics is crucial for evaluating the feasibility of integrating this process into existing production lines.
Q: What is the optical purity achievable with this resolution method?
A: The patented method consistently achieves an optical purity greater than 99.5%, meeting the stringent requirements for active pharmaceutical ingredients used in neurological treatments.
Q: How does this method compare to asymmetric synthesis in terms of cost?
A: Unlike complex asymmetric synthesis routes which can cost upwards of 400 yuan/gram due to expensive catalysts and low yields, this resolution method reduces production costs to approximately 150 yuan/gram by utilizing readily available racemic mixtures.
Q: Which chiral acid provides the best resolution efficiency?
A: D-dibenzoyl tartaric acid is identified as the preferred resolving agent, offering rapid salt formation kinetics and superior solubility differentiation compared to alternatives like camphorsulfonic acid or tartrate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (-)-Huperzine A Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-quality neurological agents for the global pharmaceutical market. Our technical team has extensively analyzed the pathways described in CN103224467A and possesses the expertise to optimize this resolution process for maximum efficiency. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial reality is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the >99.5% optical purity required for clinical-grade (-)-Huperzine A, guaranteeing that every batch meets the highest regulatory standards.
We invite potential partners to engage with our technical procurement team to discuss how this cost-effective manufacturing route can benefit your specific product pipeline. By leveraging our capabilities, you can secure a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical metrics rather than theoretical projections. Together, we can accelerate the availability of affordable, high-purity treatments for cognitive health disorders.
