Advanced Triphenylamine Pyridine Derivatives for High-Efficiency OLED Manufacturing
Advanced Triphenylamine Pyridine Derivatives for High-Efficiency OLED Manufacturing
The rapid evolution of the organic electronics sector demands materials that offer superior luminous efficiency and color purity while maintaining manufacturability. Patent CN102276525A introduces a groundbreaking class of triphenylamine-substituted pyridine derivatives designed specifically to address these challenges in organic electroluminescent devices. These compounds leverage a unique molecular architecture combining an electron-donating triphenylamine moiety with an electron-deficient pyridine ring, facilitating efficient intramolecular charge transfer (ICT). This structural innovation results in materials capable of emitting yellow-green fluorescence with significantly enhanced quantum efficiency and stability, positioning them as critical components for next-generation display technologies.
For R&D directors and procurement specialists seeking a reliable OLED material supplier, understanding the underlying chemistry is paramount. The patent outlines a versatile synthetic route that allows for extensive functionalization, enabling the fine-tuning of electronic properties without compromising thermal stability. By integrating these derivatives into the emissive layer, either as guest materials doped into a host matrix like AlQ3 or as independent layers, manufacturers can achieve devices with lower turn-on voltages and improved operational lifetimes. The following analysis delves into the technical specifics and commercial viability of this technology.
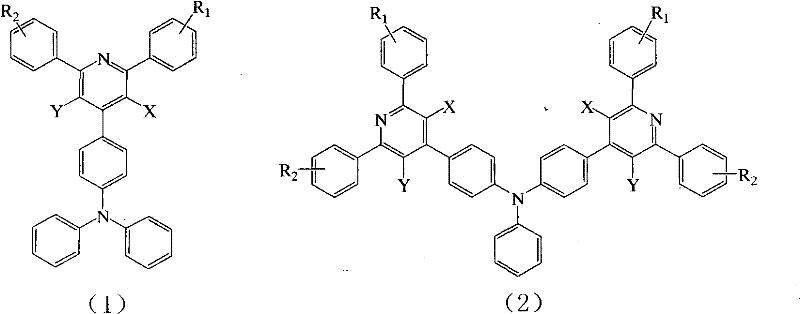
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional organic dye molecules used in electroluminescent devices often suffer from significant drawbacks when employed in solid-state applications. A primary issue is concentration quenching, where high concentrations of the emitter lead to broadened emission peaks, spectral red shifts, and a drastic reduction in fluorescence quantum efficiency. To mitigate this, conventional processes typically require doping the dye at very low concentrations into a host material, which complicates the device architecture and increases sensitivity to processing variations. Furthermore, many existing heteroaromatic compounds with large conjugated structures are prone to oxidation, generating singlet oxygen that degrades device stability over time. This instability necessitates frequent replacement and increases the total cost of ownership for display manufacturers.
The Novel Approach
The technology disclosed in CN102276525A overcomes these limitations through a strategic molecular design that incorporates intramolecular charge transfer (ICT) characteristics. By connecting an electron donor part (triphenylamine) and an electron acceptor moiety (pyridine) via a conjugated structure, the resulting derivatives exhibit a larger Stokes displacement. This effectively prevents self-absorption phenomena, a common plague in conventional fluorophores. Additionally, the chemical stability of these pyridine derivatives is markedly superior, as they are less susceptible to oxidative degradation. The ability to regulate emission wavelengths simply by changing substituting groups offers unparalleled flexibility, allowing for the creation of compounds with specific colors and required characteristics without redesigning the entire synthetic pathway.
Mechanistic Insights into Intramolecular Charge Transfer (ICT) Cyclization
The core mechanism driving the performance of these materials is the efficient intramolecular charge transfer between the donor and acceptor units. The triphenylamine group acts as a robust hole-transporting unit due to its propeller shape and electron-rich nature, while the pyridine ring serves as an electron-transporting unit. When excited, electrons move from the HOMO level localized on the triphenylamine to the LUMO level on the pyridine ring. This spatial separation of charge carriers reduces the probability of non-radiative recombination, thereby enhancing the fluorescence quantum efficiency. The presence of various substituents such as cyano groups, trifluoromethyl groups, or alkoxy groups further modulates the electron density, allowing for precise tuning of the energy gap to match the desired emission spectrum, typically in the yellow-green region for high-efficiency applications.
From a purity and impurity profile perspective, the synthesis relies on a condensation reaction that is highly selective under the specified conditions. The use of glacial acetic acid as a solvent and ammonium acetate as a catalyst promotes the formation of the pyridine ring while minimizing side reactions. The subsequent purification steps, involving silica gel column chromatography with petroleum ether and dichloromethane or recrystallization from orthodichlorobenzene, ensure that residual starting materials and by-products are removed to levels acceptable for electronic grade materials. This rigorous control over the impurity谱 is essential for preventing trap states in the OLED device that could otherwise lead to efficiency roll-off at high brightness levels.
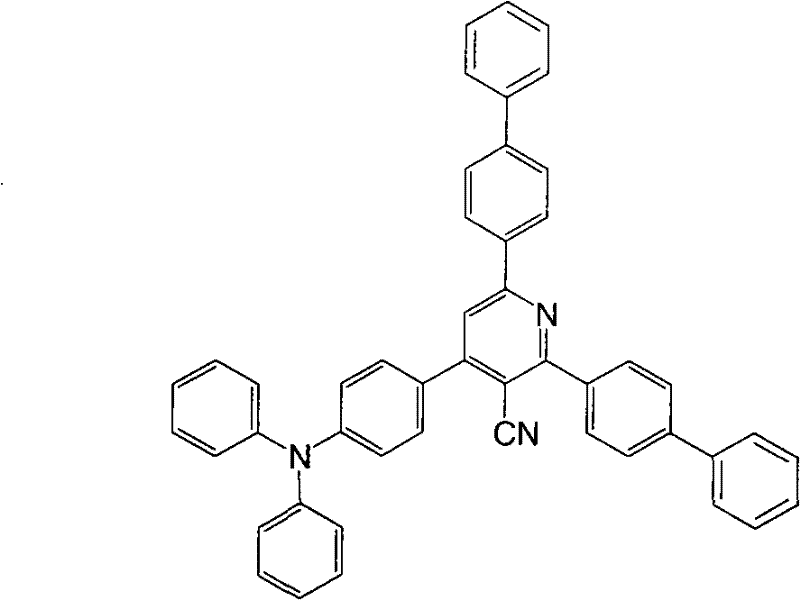
How to Synthesize Triphenylamine Pyridine Derivatives Efficiently
The synthesis protocol described in the patent offers a robust and scalable pathway suitable for industrial production. The process begins with the formation of a pyridinium tribromide salt intermediate, followed by a one-pot condensation with substituted acetophenones and triphenylamine aldehydes. This method avoids the use of expensive transition metal catalysts, relying instead on thermal activation in a acidic medium. The detailed standardized synthesis steps, including specific molar ratios and purification techniques, are outlined below to guide process engineers in replicating these high-performance materials.
- Preparation of pyridinium tribromide salt by reacting 2-bromo-1-methyl phenyl ketone with pyridine in ethanol at room temperature.
- Condensation reaction mixing the pyridinium salt, substituted acetophenone, and triphenylamine aldehyde in glacial acetic acid with ammonium acetate catalyst.
- Refluxing the mixture at 120°C for 24 hours followed by purification via silica gel column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology presents compelling economic and logistical benefits. The synthetic route is characterized by the use of readily available commodity chemicals such as acetophenones, benzaldehydes, and pyridine, which ensures a stable and diversified supply base. Unlike phosphorescent emitters that rely on scarce precious metals like iridium or platinum, these fluorescent derivatives are metal-free, eliminating the volatility associated with precious metal markets and significantly reducing raw material costs. This shift to organic-only compositions simplifies the sourcing strategy and mitigates supply chain risks related to geopolitical constraints on metal mining.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of simple reflux conditions at 120°C drastically simplify the production process. This thermal method does not require specialized high-pressure equipment or inert atmosphere gloveboxes beyond standard nitrogen protection, leading to substantial capital expenditure savings. Furthermore, the high yields reported in the embodiments, often ranging from 70% to 90%, minimize waste generation and improve overall material throughput, directly contributing to a lower cost per gram of the final active material.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the easy substitution of starting materials to create different derivatives without altering the core process infrastructure. This flexibility means that production lines can be quickly adapted to meet changing market demands for specific emission colors or efficiency profiles. The reliance on common organic solvents like ethanol and glacial acetic acid further ensures that solvent supply chains remain robust and unaffected by niche chemical shortages, guaranteeing consistent delivery schedules for downstream OLED panel manufacturers.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to heavy-metal-based synthesis routes. The primary by-products are water and ammonium salts, which are easier to treat and dispose of in compliance with environmental regulations. The scalability of the reflux condensation reaction is well-proven in the fine chemical industry, allowing for seamless scale-up from kilogram laboratory batches to multi-ton annual commercial production without the need for complex process re-engineering or safety overhauls.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of triphenylamine-substituted pyridine derivatives in organic electroluminescent devices. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy for engineering and procurement decision-making.
Q: What is the primary advantage of triphenylamine-substituted pyridine derivatives in OLEDs?
A: These derivatives exhibit strong intramolecular charge transfer (ICT) characteristics, resulting in yellow-green fluorescence with longer wavelengths, narrower absorption bands, and higher device efficiency compared to conventional dyes.
Q: How is the emission color tuned in these materials?
A: The emission wavelength can be easily regulated by modifying the substituting groups (R1, R2, X, Y) on the pyridine ring and phenyl groups, allowing for precise control over the HOMO-LUMO energy gap.
Q: Are these materials suitable for large-scale production?
A: Yes, the synthesis utilizes a straightforward one-pot condensation reaction in glacial acetic acid at 120°C, avoiding complex catalytic cycles and enabling scalable manufacturing with yields often exceeding 80%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triphenylamine Pyridine Derivative Supplier
As the demand for high-performance OLED materials continues to surge, partnering with an experienced CDMO is essential for translating patented chemistry into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our stringent purity specifications and rigorous QC labs guarantee that every batch of triphenylamine pyridine derivatives meets the exacting standards required for display and lighting applications, minimizing device failure rates and maximizing yield.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain for these advanced materials. By requesting a Customized Cost-Saving Analysis, you can gain insights into potential reductions in your Bill of Materials (BOM) and identify opportunities for process integration. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific product requirements, ensuring a seamless transition from R&D to mass production.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
