Scalable One-Pot Synthesis of Prednisolone Derivatives for Commercial API Manufacturing
The pharmaceutical industry continuously seeks robust manufacturing routes for corticosteroid intermediates, particularly for treating respiratory and inflammatory conditions. Patent CN1325508C introduces a transformative one-pot methodology for synthesizing prednisolone derivatives, specifically targeting the complex 16α,17-acetal-21-ester structural motif found in advanced therapeutics like ciclesonide. This innovation addresses the longstanding inefficiencies of multi-step syntheses by combining esterification and acetalization into a single operational unit. By leveraging a tandem acid-catalyzed mechanism, the process eliminates the need for isolating unstable intermediates, thereby streamlining the production workflow. For R&D directors and process chemists, this represents a significant leap forward in atom economy and operational simplicity. The ability to generate high-value anti-inflammatory intermediates with superior stereoselectivity directly impacts the commercial viability of generic and novel drug formulations. As a leading fine chemical manufacturer, we recognize this technology as a cornerstone for next-generation API intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 16,17-acetal-21-ester prednisolone derivatives has been plagued by operational complexity and poor stereochemical control. Traditional routes, such as those disclosed in German patent DE 4,129,535, typically involve a sequential approach where the C-21 hydroxyl group is first esterified, followed by isolation and purification of the intermediate before proceeding to the acetalization at the C-16 and C-17 positions. This stepwise fragmentation not only extends the overall cycle time but also exposes the sensitive steroid backbone to multiple workup procedures, each introducing potential yield losses. Furthermore, conventional methods often struggle with stereoselectivity at the C-22 position, frequently producing a near 1:1 mixture of the desired 22R-isomer and the unwanted 22S-isomer. Separating these diastereomers requires expensive and tedious chromatographic techniques or repeated crystallizations, which severely erodes profit margins and complicates supply chain logistics for large-scale manufacturing.
The Novel Approach
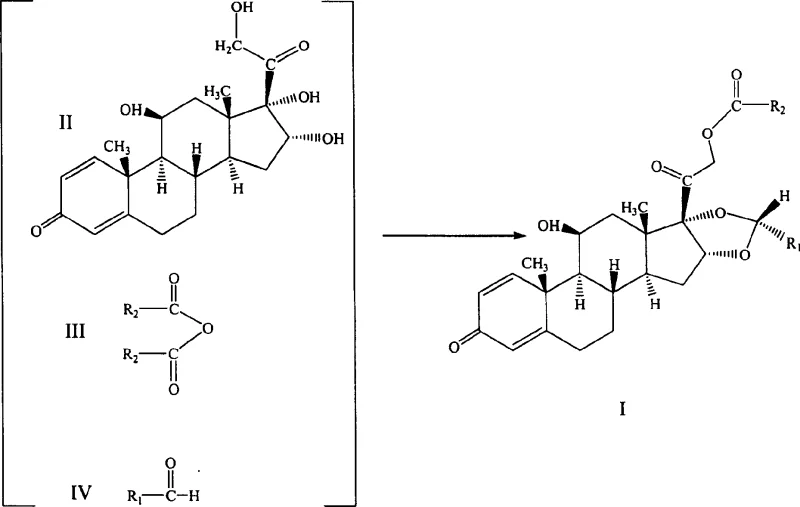
In stark contrast, the one-pot method described in CN1325508C consolidates the entire transformation into a single reactor vessel, fundamentally altering the economic landscape of production. By reacting 16α-hydroxy prednisolone simultaneously or sequentially with an acid anhydride and an aldehyde in the presence of a Bronsted acid catalyst, the process bypasses the isolation of the C-21 ester intermediate entirely. This convergence of reaction steps minimizes solvent consumption, reduces labor costs associated with filtration and drying, and significantly shortens the total processing time. Crucially, the optimized reaction conditions foster a highly favorable thermodynamic environment that preferentially drives the formation of the pharmacologically active 22R-isomer. The result is a streamlined process that delivers the target molecule with an R/S ratio exceeding 95:5, effectively solving the purity challenges that have hindered previous generations of synthesis technology.
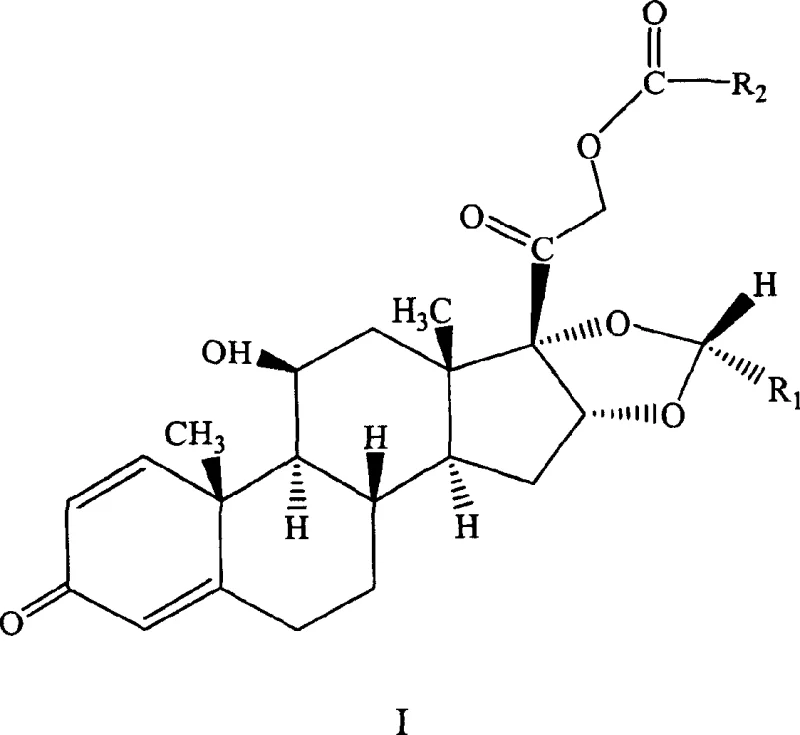
Mechanistic Insights into Acid-Catalyzed Tandem Esterification and Acetalization
The core of this technological breakthrough lies in the precise orchestration of two distinct chemical transformations occurring concurrently within the same reaction matrix. The mechanism initiates with the activation of the acid anhydride by the strong Bronsted acid catalyst, likely perchloric acid, which facilitates the nucleophilic attack by the primary C-21 hydroxyl group of the prednisolone scaffold. Simultaneously, the acid catalyst protonates the carbonyl oxygen of the aldehyde, rendering it highly electrophilic and susceptible to attack by the vicinal diol system at the C-16 and C-17 positions. This dual activation ensures that both the esterification and the cyclic acetal formation proceed with synchronized kinetics, preventing the accumulation of partially reacted species that could lead to side products. The choice of solvent plays a critical role in stabilizing the transition states; polar aprotic solvents like dioxane or nitromethane provide the necessary dielectric environment to support the ionic intermediates generated during the acid catalysis.
Furthermore, the exceptional stereoselectivity observed in this process can be attributed to the specific coordination geometry enforced by the bulky cyclohexyl or aryl groups on the aldehyde reactant during the ring-closing step. Under the controlled temperature range of 20°C to 30°C, the reaction pathway favors the formation of the thermodynamically more stable 22R-configured acetal ring. This kinetic control prevents the epimerization that often plagues high-temperature reactions, ensuring that the final product possesses the requisite biological activity without the need for downstream chiral resolution. By maintaining the acid catalyst concentration within a molar ratio of 1:1 to 10:1 relative to the steroid substrate, the system avoids excessive acidity that could degrade the sensitive enone system in the A-ring of the steroid, thus preserving the integrity of the pharmacophore throughout the synthesis.
How to Synthesize Prednisolone Derivatives Efficiently
Implementing this one-pot strategy requires careful attention to reagent addition sequences and thermal management to maximize yield and purity. The process begins by suspending the 16α-hydroxy prednisolone starting material in a suitable polar solvent, followed by the controlled addition of the acid catalyst to initiate the reaction environment. Subsequently, the acid anhydride and aldehyde are introduced, either simultaneously or in a specific sequence depending on the desired reaction kinetics, while maintaining strict temperature control to prevent exothermic runaway. The detailed standardized synthetic steps, including specific molar ratios, stirring rates, and workup procedures, are outlined in the guide below to ensure reproducibility on a commercial scale.
- Suspend 16α-hydroxy prednisolone in a polar organic solvent such as dioxane and add a catalytic amount of strong Bronsted acid, preferably 60-70% perchloric acid.
- Sequentially add the acid anhydride (e.g., isobutyric anhydride) and the aldehyde (e.g., cyclohexyl formaldehyde) to the reaction mixture while maintaining the temperature between 0°C and 50°C.
- Stir the reaction mixture for 5 to 8 hours to ensure complete conversion, followed by neutralization, extraction, and recrystallization to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this one-pot synthesis route offers profound strategic advantages that extend beyond simple chemical efficiency. By collapsing multiple unit operations into a single step, the manufacturing footprint is significantly reduced, allowing for higher throughput within existing facility constraints. The elimination of intermediate isolation steps means that there is no need for dedicated drying equipment, filtration hardware, or storage vessels for unstable intermediates, which translates directly into substantial capital expenditure savings and reduced operational overhead. Moreover, the simplified workflow minimizes the exposure of the product to environmental contaminants, thereby enhancing the overall quality consistency and reducing the risk of batch failures due to handling errors.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the drastic reduction in processing time and material usage. Since the intermediate does not need to be isolated, purified, and dried, the consumption of solvents for washing and crystallization is markedly decreased. This reduction in solvent volume not only lowers raw material costs but also diminishes the expense associated with solvent recovery and waste disposal. Additionally, the higher stereoselectivity reduces the need for costly chiral separation technologies, further driving down the cost of goods sold and improving the margin profile for the final API.
- Enhanced Supply Chain Reliability: Shortening the synthetic route inherently reduces the lead time required to produce batches of the intermediate. With fewer steps comes a lower probability of bottlenecks or equipment downtime affecting the overall schedule. The robustness of the one-pot method allows for more predictable production planning, enabling suppliers to respond more agilely to fluctuations in market demand. This reliability is crucial for securing long-term contracts with pharmaceutical companies that require guaranteed continuity of supply for their critical respiratory medications.
- Scalability and Environmental Compliance: From an environmental perspective, the process aligns well with green chemistry principles by minimizing waste generation and energy consumption. The ability to potentially run the reaction under solvent-free conditions or with recyclable solvents like dioxane simplifies the regulatory compliance burden regarding volatile organic compound emissions. Scaling this process from pilot plant to multi-ton production is straightforward because it relies on standard mixing and temperature control technologies rather than specialized reactors, making it an ideal candidate for rapid capacity expansion to meet global healthcare needs.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this patented synthesis route. Understanding these nuances is essential for process engineers looking to adapt this technology for their specific manufacturing lines. The answers provided are derived directly from the experimental data and embodiments disclosed in the patent literature, ensuring accuracy and relevance for industrial application.
Q: How does the one-pot method improve stereoselectivity compared to traditional stepwise synthesis?
A: The one-pot method utilizing specific Bronsted acid catalysts like perchloric acid significantly enhances the ratio of the desired 22R-isomer. While conventional methods often yield nearly equal proportions of R and S isomers, this optimized process achieves an R/S ratio greater than 95:5, drastically reducing the burden of chiral purification.
Q: What are the critical reaction parameters for maximizing yield in this synthesis?
A: Temperature control is paramount; the reaction should be conducted between 20°C and 30°C. Temperatures exceeding 50°C can lead to the decomposition of the C-21 ester, while temperatures below 0°C may hinder the solubility of reactants. Additionally, maintaining a molar ratio of approximately 1:6:4 for the steroid, anhydride, and aldehyde respectively ensures optimal conversion.
Q: Can this process be adapted for solvent-free conditions to reduce environmental impact?
A: Yes, the patent discloses that the reaction can proceed without additional solvents, as liquid reactants like the aldehyde or anhydride can serve as the reaction medium. However, for better heat dissipation and mixing on a large scale, polar solvents like dioxane or ethyl acetate are preferred to ensure consistent product quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prednisolone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like this one-pot prednisolone derivative synthesis are translated efficiently from the lab to the plant. Our technical team is adept at optimizing reaction parameters to meet stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the highest international standards for API intermediates. We understand that the transition from bench-scale discovery to commercial manufacturing requires not just chemical expertise but also deep engineering insight to manage heat transfer, mixing, and safety on a large scale.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits tailored to your specific volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering high-quality chemical solutions that drive your business forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
